ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2022; 18(8):3209-3222. doi:10.7150/ijbs.70289 This issue Cite
Research Paper
Renal cell carcinoma-derived exosomes deliver lncARSR to induce macrophage polarization and promote tumor progression via STAT3 pathway
1. Department of Urology, Tongde Hospital of Zhejiang Province, Hangzhou, Zhejiang 310012, China.
2. Cancer Institute of Integrated Traditional Chinese and Western Medicine, Key laboratory of cancer prevention and therapy combining traditional Chinese and Western Medicine of Zhejiang Province, Zhejiang Academy of Traditional Chinese Medicine, Tongde Hospital of Zhejiang Province, Hangzhou, Zhejiang 310012, China.
3. College of Life Sciences, Zhejiang Chinese Medical University, Hangzhou, 310053, China.
4. Department of Tumor Research Institute, Cancer Hospital of the University of Chinese Academy of Sciences, Zhejiang Cancer Hospital, Hangzhou, China.
5. Department of Urology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou 325000, China.
*These authors contributed equally to this work.
Abstract
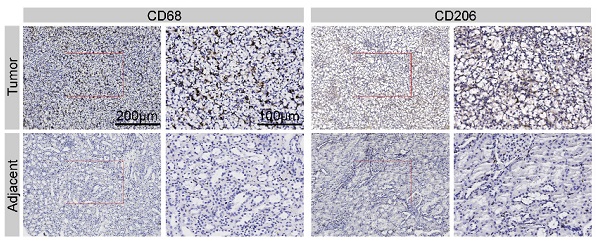
Tumor-derived exosomes play a pivotal role in regulating tumor progression by mediating crosstalk between tumor cells and immune cells such as macrophages within the tumor microenvironment. Macrophages can adopt two distinct polarization statuses and switch between M1 or M2 activation phenotypes in response to the different external stimuli. However, the role of tumor derived exosomes in the macrophage phenotypic switch and tumor development have not been elucidated in renal cell carcinoma (RCC). Here we found that high macrophage infiltration was associated with worse prognosis in RCC patients, therefore we propose our hypothesis that RCC derived exosomes might directly influence macrophage polarization and thus promote tumor progression. Both cell-based in vitro models and orthotopic transplantation in vivo tumor models were constructed and ELISA, flow cytometry, and macrophage functional studies were performed to investigate whether and how RCC-derived exosomes regulate macrophage polarization and tumor growth. The results found that these exosomes promote macrophage polarization, cytokine release, phagocytosis, angiogenesis, and tumor development. Further study revealed high amount of a recently discovered lncRNA called lncARSR in RCC-derived exosomes. Overexpression of lncARSR induced phenotypic and functional changes of macrophages in vitro and promoted tumor growth in vivo, while knockdown of lncARSR by siRNA disrupted the exosomes-mediated macrophage polarization. LncARSR interacts directly with miR-34/miR- 449 to increase STAT3 expression and mediate macrophage polarization in RCC cells. Together, RCC-derived exosomes facilitate the development of tumor through inducing macrophage polarization via transferring lncARSR, suggesting that RCC-derived exosomes, lncARSR and STAT3 are the potential therapeutic targets for treatment of RCC.
Keywords: Tumor-derived exosomes, Renal cell carcinoma (RCC), Macrophage polarization, lncARSR, STAT3
