ISSN: 1449-2288International Journal of Biological Sciences
Int J Biol Sci 2022; 18(4):1755-1772. doi:10.7150/ijbs.68015 This issue Cite
Research Paper
PrPC Promotes Endometriosis Progression by Reprogramming Cholesterol Metabolism and Estrogen Biosynthesis of Endometrial Stromal Cells through PPARα Pathway
1. Shanghai Key Laboratory of Maternal Fetal Medicine, Clinical and Translational Research Center, Shanghai First Maternity and Infant Hospital, School of Medicine, Tongji University, Shanghai 200092, China
2. Department of Obstetrics and Gynecology, Shanghai Ninth People' s Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200011, China
3. Department of Gynecology, Shanghai First Maternity and Infant Hospital, School of Medicine, Tongji University, Shanghai 200092, China
4. Laboratory for Reproductive Immunology, Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases, Obstetrics and Gynecology Hospital of Fudan University, Shanghai 200011, China
5. Key Laboratory of Reproduction Regulation of NPFPC, SIPPR, IRD, Fudan University, Shanghai 200032, China
Abstract
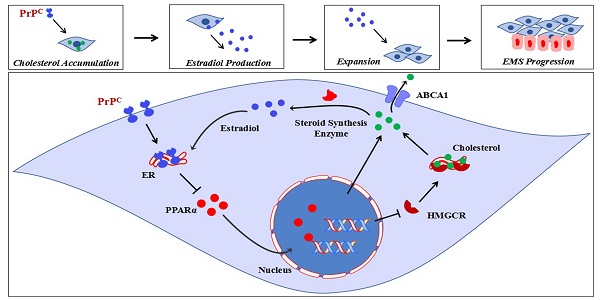
Endometriosis (EMs) is characterized as an estrogen-dependent disease. Whereas, the underlying mechanism for activated estrogen biosynthesis in EMs lesions is largely unknown. We analyzed cholesterol metabolism and estrogen biosynthesis condition of EMs lesions by biological information analysis of GEO datasets, and further verified both in vitro and in vivo by constructing EMs models with uterus fragments from donors of PRNP knockout mouse (Prnp-/-, KO119), Octapeptide repeat region of PRNP knockout mouse (KO120) and PRNP transgenic mouse (Tg20). We found that transcriptome of cholesterol metabolism and estrogen-converting enzymes were disturbed in EMs patients, and cellular cholesterol concentration and local estradiol level were substantially increased in EMs lesions, as well as the high level of prion (PrPC, encoded by PRNP). Notably, 17-β estradiol stimulation significantly up-regulated PrPC expression in endometrial stromal cells (ESC) and PrPC promoted the proliferative, migratory and invasive abilities of ESC, and was further verified to accelerate EMs progression in mouse models. More importantly, PrPC promoted cholesterol accumulation and activated estrogen biosynthesis of ESC in a PPARα pathway-dependent manner. Taken together, this study suggests that PrPC-cholesterol metabolism/estrogen biosynthesis contributes to the progression of EMs by negatively regulating PPARα pathway, and could be potential therapeutic targets for EMs intervention.
Keywords: endometriosis (EMs), prion (PrPC), cholesterol metabolsim, estrogen, PPARα
