Impact Factor ISSN: 1449-2288
Int J Biol Sci 2012; 8(6):913-934. doi:10.7150/ijbs.3697 This issue Cite
Review
Honey - A Novel Antidiabetic Agent
Department of Pharmacology, School of Medical Sciences, Universiti Sains Malaysia, 16150, Kubang Kerian, Kelantan, Malaysia.
Received 2011-10-23; Accepted 2012-1-24; Published 2012-7-7
Abstract
Diabetes mellitus remains a burden worldwide in spite of the availability of numerous antidiabetic drugs. Honey is a natural substance produced by bees from nectar. Several evidence-based health benefits have been ascribed to honey in the recent years. In this review article, we highlight findings which demonstrate the beneficial or potential effects of honey in the gastrointestinal tract (GIT), on the gut microbiota, in the liver, in the pancreas and how these effects could improve glycemic control and metabolic derangements. In healthy subjects or patients with impaired glucose tolerance or diabetes mellitus, various studies revealed that honey reduced blood glucose or was more tolerable than most common sugars or sweeteners. Pre-clinical studies provided more convincing evidence in support of honey as a potential antidiabetic agent than clinical studies did. The not-too-impressive clinical data could mainly be attributed to poor study designs or due to the fact that the clinical studies were preliminary. Based on the key constituents of honey, the possible mechanisms of action of antidiabetic effect of honey are proposed. The paper also highlights the potential impacts and future perspectives on the use of honey as an antidiabetic agent. It makes recommendations for further clinical studies on the potential antidiabetic effect of honey. This review provides insight on the potential use of honey, especially as a complementary agent, in the management of diabetes mellitus. Hence, it is very important to have well-designed, randomized controlled clinical trials that investigate the reproducibility (or otherwise) of these experimental data in diabetic human subjects.
Keywords: Diabetes mellitus, antidiabetic, hypoglycemic, glycemic control, liver, pancreas, insulin, honey.
Introduction
The prevalence of diabetes mellitus, estimated as 285 million people in 2010, is predicted to increase to 439 million people by the year 2030 [1]. The majority of this diabetic population will emerge from developing countries [1]. Despite the availability of various classes of antidiabetic agents, diabetes mellitus remains a major cause of mortality and morbidity globally [2, 3]. As a result, there has been a considerable effort to search for more effective drugs. This has resulted in a renewed interest in research that investigates the health benefits of herbs and natural products including honey in the management of diabetes mellitus. Honey is a natural substance produced by bees from nectar. It is considered one of the last untreated natural food substances [4]. The composition of honey is influenced by a number of factors such as geographical origin, botanical sources of nectar, environmental and climatic conditions as well as processing techniques [4, 5]. The various varieties of honey may be grouped into monofloral or multifloral [6]. The classification basically depends on whether a dominating pollen grain originated from only one particular plant (monofloral honey) or no dominant pollen type in the sample (multifloral honey) [6, 7].
Honey, which comprises predominantly monosaccharides and oligosaccharides, contains at least 181 constituents [8, 9]. It also contains other bioactive constituents such as phenolic compounds, flavonoids, organic acids, carotenoid-derived compounds, nitric oxide (NO) metabolites, ascorbic acid, Maillard reaction products, aromatic compounds, trace elements, vitamins, amino acids and proteins [5, 9, 10]. Evidence indicates that some varieties of honey contain kynurenic acid (a tryptophan metabolite with neuroactive activity) which may contribute to its antinociceptive and antimicrobial properties [11]. A number of enzymes such as glucose oxidase, diastase, invertase, phosphatase, catalase and peroxidase have also been documented in honey [9, 12]. The use of honey in folk medicine dates back to 2100-2000 BC [4, 12]. In the past, most of the health benefits attributed to honey were based on mere observations or generalizations without any scientific support [13]. However, in the last few years, there has been a renewed interest in research that investigates the potential health benefits of natural and unprocessed honey in the management of various diseases. This has resulted in findings that attribute several medicinal effects to honey. These include cardioprotective [14], hepatoprotective [15], hypoglycemic [16], antioxidant [16-21] and antihypertensive effects [22, 23]. Other effects such as antibacterial [24], anti-fungal [25], anti-viral [26], anti-inflammatory [27] and antitumor [28] have also been documented and attributed to honey. In this review, the first of its kind, we summarize both experimental and clinical findings which demonstrate that the effects of honey in the GIT, on the gut microbiota, in the liver as well as in the pancreas may result in improved glycemic control and metabolic derangements. Based on the not-too-impressive clinical findings (due to improper study designs), recommendations on how to ensure that these compelling data obtained in animal studies are successfully translated to the benefits of diabetic patients are made.
Effects of honey in the GIT
Fructose and glucose, the main carbohydrates in honey, have the same molecular formula but different structural formula (figure 1) [29-30]. These monosaccharides do not need to be hydrolyzed by GIT enzymes and thus are ready for absorption. Fructose and glucose have different transporters, GLUT5 (and/or GLUT2) and SGLT1, respectively [31]. Studies have shown that fructose reduces hyperglycemia or glucose levels in rodent models of diabetes, healthy subjects and diabetic patients [32-35]. Evidence suggests that fructose consumption prolongs gastric emptying [36], which may slow down the rate of intestinal absorption [37]. In addition to fructose, oligosaccharides such as palatinose (isomaltulose) present in honey have been reported to delay digestion and intestinal absorption of glucose resulting in reduced glycemia [38, 39]. Besides delaying absorption, evidence suggests that fructose consumption lowers food intake [40]. Reduced food intake is attributed to delayed gastric emptying [41]. Reduced food intake due to fructose ingestion has been reported to influence the selection of macronutrients [40, 42]. Even though the findings on satiety are inconclusive [42], a recent study also corroborates this suppressant effect of fructose on food intake [43]. It is suggested that the slow absorption of fructose in the intestine might elongate the duration of contact and interaction between fructose and intestinal receptors that play a key role in satiety [44, 45]. This might allow more macronutrients (including carbohydrates) to be passed into the large intestine, thereby preventing/limiting their intestinal absorption. Moreover, with the evidence suggesting that fructose reduces food intake, there is a possibility for reduced weight gain. A recent study reported that supplementation with a low or moderate fructose diet (together with natural fruit supplements) resulted in weight loss in obese subjects [46]. The study further showed that obese subjects on the moderate-fructose diet lost more weight than those on the low-fructose diet [46]. However, some studies have found that fructose feeding or consumption at high doses is associated with increased weight gain [43, 47].
Molecular structures of glucose and fructose.
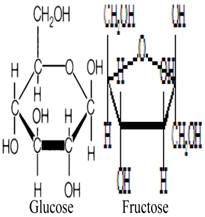
After fructose, glucose is the second major constituent in most varieties of honey [4]. Compelling evidence indicates that the intestinal absorption of fructose is enhanced in the presence of glucose [48]. Although it remains unclear how glucose enhances fructose absorption, the recruitment of GLUT2 carrier to the brush border membrane caused by increased intestinal fructose may contribute to the synergistic effect of glucose on the absorption of fructose [48, 49]. In addition, increased expression levels of GLUT5 mRNA after ingestion of fructose but not glucose has been documented [50]. It is also suggested that there may be a disaccharidase-related transport system which considers both fructose and glucose as products of the enzymatic hydrolysis of sucrose [51]. Evidence also suggests that fructose is absorbed via a saturable carrier in the absence of glucose [52]. In contrast, in the presence of glucose, fructose is absorbed via a disaccharidase-related transport system [52]. The presence of this disaccharidase-related transport system appears to be corroborated by a study which showed that the greatest absorption of fructose occurred when equal amounts of fructose and glucose were given simultaneously [53]. Passive diffusion across the intestinal epithelium is also a possible mechanism [48, 54]. These studies have generally shown that glucose enhances the transportation and absorption of fructose but not vice versa. This enhancing effect of glucose on fructose absorption may further increase the amounts of fructose that reach the liver, a primary target of fructose.
The absorption of some macromolecules, nutrients and drugs may be influenced by the concurrent intake or ingestion of certain food constituents. Although little is known about the role of honey in facilitating the absorption and uptake of food constituents or drugs, however, evidence suggests a potential role of honey in modulating such an effect [55]. This differential absorption of macronutrients and drugs, due to concomitant ingestion of food components may also influence considerably their systemic effects. Studies have documented a number of changes or alterations in the brush border membrane (BBM) fluidity in diabetes [56]. These alterations (such as increased number and size of cells, increased number and area of microvilli per cell, enhanced activities of carbohydrate-hydrolyzing enzymes such as maltase, sucrase and trehalase as well as facilitated glucose absorption) may aggravate diabetes and its related complications [57-59]. Besides alterations in the BBM fluidity, Bhor and Sivakami reported increased oxidative damage and non-enzymatic glycation in duodenum and jejunum of diabetic rats [56]. The authors also found a significant correlation between reduced BBM fluidity and increased oxidative stress in the duodenum and jejunum of diabetic rats [56]. Vitamin E, an antioxidant, has been shown to ameliorate intestinal oxidative stress [60]. Even though there is no evidence to suggest that honey ameliorates oxidative stress in the intestine, there is overwhelming evidence in support of such an effect in pancreas, kidney and liver [16, 17, 19, 61-63]. Considering that the antioxidant effect of honey is not likely to be limited to these tissues alone, therefore, amelioration of intestinal oxidative stress might restore alterations associated with BBM fluidity, promote healing and enhance intestinal health during diabetes. Besides, studies have also implicated the role of oxidation-reduction reactions in the differential transport of molecules [64]. Additional evidence also showed that intestinal oxidative state can influence the bioavailability of nutrients and drugs [65]. Considering the reported antioxidant effect of honey [16, 17, 19, 61-63], this might be one of the reasons co-administration of glibenclamide or metformin with honey improved glycemic control better than either hypoglycemic drug in streptozotocin (STZ)-induced diabetic rats [66].
Other effects of honey in the GIT
Studies have shown that diabetes mellitus impairs gastric ulcer healing in rodents [67]. Evidence also suggests diabetes may be a predictor of peptic ulcer disease or aggravate associated complications or mortality in patients with gastric and duodenal ulcers [68, 69]. In view of findings which demonstrated that honey prevented chemical-induced gastric mucosal injury in rodents [70], this gastroprotective effect of honey might be beneficial in diabetes mellitus. Evidence that monosaccharides (fructose and glucose) and disaccharides (sucrose and maltose) present in honey may contribute to the gastroprotective effect of honey is provided by Gharzouli and co-workers [70]. The authors showed that a mixture comprising glucose, fructose, sucrose and maltose exerted gastroprotective effect, which was similar to that produced by honey [70]. Additional evidence in support of the potential role of honey in ameliorating gastric ulcer is provided by studies which showed that honey inhibited the growth of Helicobacter pylori, a main causative pathogen in gastric and duodenal ulcers [71]. As demonstrated by Osato and colleagues, fructose and glucose in honey might contribute to the inhibitory effect of honey on Helicobacter pylori [71]. Other beneficial effects of honey supplementation in the GIT have also been documented. These effects include improved intestinal morphology and protection against intra-abdominal adhesions and anastomotic dehiscence in rats after colonic resection and anastomosis [72]. In rats with obstructive jaundice, honey supplementation also improved morphology of the ileum [73] or anastomotic wound healing [74].
These potential effects of honey or its constituents on gastric emptying [36], rate of intestinal absorption [37-39, 42, 55], fructose transporter [48-49, 51-52], food intake [40, 43, 46] and perhaps the synergistic effect of glucose on fructose [48, 53] may contribute to the glucose-lowering effect of honey in systemic circulation. Some yet-to-be-delineated preabsorptive signals elicited by honey or its constituents may also contribute to the glucose-lowering effect of honey. Other potential beneficial GIT effects of honey include the amelioration of intestinal oxidative stress (with a resultant effect on the restoration of BBM fluidity) which may enhance bioavailability of vital macronutrients or drugs [64, 65]. By and large, these studies indicate that honey or its constituents may modulate several physiological and pathophysiological conditions in the GIT. Together with its potential to improve intestinal morphology [72-74] or its gastroprotective effect [70, 71], these GIT effects of honey might promote healing and enhance GIT health in diabetes mellitus. This might impact positively on glycemic control.
Effect of honey on gut microbiota
The gut microbiota is recognized to play an important role in health and disease [75]. Recent evidence now indicates that the role of gut microbiota extends beyond the gut [76, 77]. As demonstrated in recent studies, these gut microorganisms can influence several functions in systemic organs such as liver [76] and brain [77]. Even though the gut microbiota has not been linked directly to the pathogenesis of diabetes mellitus, these latest findings on the systemic effects of gut microbiota suggest a possibility [76, 77]. These data are important in view of the fact that an imbalance or alteration in the intestinal composition of microbiota is implicated in the development of insulin resistance and obesity [78]. A study by Cani et al. provides evidence in support of a potential role of the gut microbiota in diabetes [79]. The authors reported that mice fed high fat had markedly declined levels of intestinal bifidobacteria. Treatment of these high-fat-fed mice with oligofructose totally restored the intestinal contents of bifidobacteria [79]. The study also indicated that in high-fat-fed mice treated with oligofructose, the levels of bifidobacteria considerably and positively correlated with improved glucose-induced insulin secretion and glucose tolerance [79].
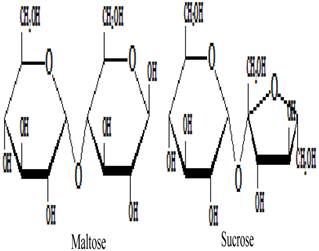
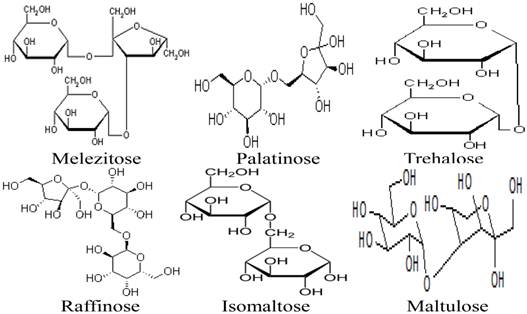
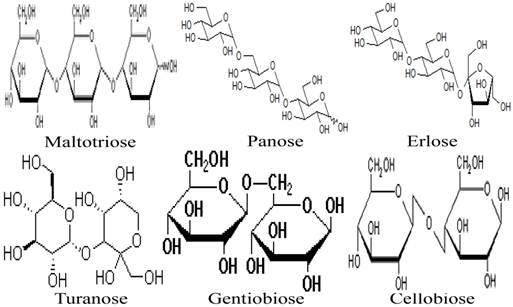
Besides the monosaccharides, honey contains a variety of oligosaccharides which constitute about 5-10 % of the total carbohydrates in honey [9, 80]. Oligosaccharides are sugars formed from the condensation of monosaccharides (usually 2 to 6). These monosaccharide monomers are linked together by a glycosidic bond [42]. Based on the number of their monosaccharide units, oligosaccharides are grouped into disaccharides, trisaccharides, tetrasaccharides, pentasaccharides and hexasaccharides. Some of the oligosaccharides which are present in honey include maltose, sucrose, melezitose, palatinose, trehalose, raffinose, isomaltose, maltulose, maltotriose, panose, erlose, turanose, gentiobiose and cellobiose [80-83]. The molecular structures of these oligosaccharides are presented in figures 2, 3 and 4 [29, 84-87]. The effect of honey on beneficial or non-pathogenic gut microorganisms is well documented [80]. Evidence from in vitro and in vivo studies has shown that honey markedly increased the number of Lactobacillus (L. acidophilus and L. plantarum) counts [88]. Honey was also reported to enhance the growth of Lactobacillus acidophilus, Bifidobacterium bifidum, Streptococcus thermophilus and Lactobacillus delbrukeii sub. sp. bulgaricus [89]. The study also showed that honey enhanced lactic acid production [89]. The fact that the effect of honey on these microbes was comparable to that of fructose or sucrose implies that the sugar constituents of honey might contribute to the prebiotic effect of honey. Similarly, a study that investigated the effect of honey (5% w/v) on five different species of human intestinal Bifidobacterium (B. longum, B. adolescentis, B. breve, B. bifidum and B. infantis) revealed that honey considerably augmented the growth of these bacteria [90]. The study further showed that the effect of honey on these microbes, as measured by production of lactic and acetic acid, was comparable to that of fructooligosaccharide, galactooligosaccharide or inulin [90]. As reported by Sanz et al., fractions of honey oligosaccharides exhibited prebiotic activity and increased the populations of beneficial or non-pathogenic bacteria, lactobacilli and bifidobacteria [91]. A number of other studies have also documented the beneficial effect of different varieties of honey on the growth and activity of intestinal microorganisms [92, 93].
Molecular structures of maltose and sucrose.
Molecular structures of melezitose, palatinose, trehalose, raffinose, isomaltose and maltulose.
Molecular structures of maltotriose, panose, erlose, turanose, gentiobiose, and cellobiose.
Additional in vivo evidence in support of prebiotic effect of honey is provided by Ezz El-Arab and colleagues [94]. The authors showed that honey feeding inhibited the harmful and genotoxic effects of mycotoxins in Swiss albino mice and considerably increased the colonic bifidobacteria and lactobacilli counts [94]. Similarly, a recent study also demonstrated the prebiotic effect of honey [95]. The authors reported that the growth, activity and viability of microorganisms were not influenced by the type of sweetener, oligosaccharide content or floral source of the different varieties of honey [95]. In addition to the oligosaccharides in honey, evidence suggests that the large quantities of monosaccharides (fructose and glucose) in honey may also augment the growth of intestinal microflora [89, 91, 95]. Therefore, considering that honey is enriched in oligosaccharides, fructose, glucose and other sugars [9, 90, 91], honey supplementation in diabetes mellitus may enhance intestinal health and growth of gut microbiota; and thus may contribute to glucose management by yet-to-be-identified mechanisms [66].
Evidence implicates the role of oligosaccharides in the antidiabetic effect of honey [80]. Considering that honey is enriched in oligosaccharides, even though data are still lacking, the pharmacokinetic parameters of honey are likely to be similar to those of oligosaccharides such as palatinose and isomaltose. These include delayed gastric emptying, slower rate of digestion, delayed intestinal absorption, reduced intestinal absorption of calorie resulting from increased intestinal calorie extraction by pathogenic gut microbiota, urinary excretion of honey metabolites [38, 39, 79, 96-98]. Besides, some of these pharmacokinetic properties have also been reported for fructose, a major constituent of honey [36, 37, 40-42]. These effects will result in lower elevations in blood glucose which may as well stimulate minimal secretion of insulin [39, 79, 98].
Potential effect of honey in the liver
The liver is recognized for its role in the pathophysiology and control of glycemia in diabetes mellitus [99]. Honey comprises predominantly carbohydrates which can be classified into monosaccharides and oligosaccharides. Monosaccharides are grouped into trioses, tetroses, pentoses, hexoses and heptoses based on their number of carbon atoms (usually 3 to 7). The two monosaccharides in honey, glucose and fructose, are basically hexoses. The molecular structures of these monosaccharides are shown in figure 1 [29-30]. In the liver, the uptake and metabolism of these monosaccharides in honey (glucose and fructose) are different [42]. Evidence suggests that less quantities of glucose than fructose are metabolized in the liver [42]. Available data also indicate that the uptake of glucose into the liver is impaired in the absence or insufficiency of insulin. Likewise, the metabolism of glucose is impaired by inadequate amounts of insulin [42, 100]. In contrast, neither the uptake nor metabolism of fructose is impaired or affected by the levels of insulin [42, 100]. Earlier authors have described the metabolism of fructose and glucose in more details [42, 100].
Based on the literature, fructose (the major constituent in honey) may contribute to the antidiabetic or hypoglycemic effect of honey [35]. Fructose has been shown to increase hepatic glucose phosphorylation via activation of glucokinase [101]; and inhibit glycogenolysis via suppression of phosphorylase [102]. Increased hepatic glycogen synthesis via activation of glycogen synthase produced by fructose administration in diabetic and non-diabetic rats has been reported [101, 103]. Evidence has also implicated the complementary and synergistic role of fructose and glucose on key enzymes involved in the metabolism of glucose and glycogen in the liver [104, 105]. Evidence indicates that these monosaccharides produce a synergistic effect in the liver only when present or administered at a low or moderate dose or concentration [106]. It has been reported that the percentage of the absorbed fructose phosphorylated in the liver is two-fold or more that of the absorbed glucose [100, 107].
With greater amounts of fructose compared to glucose (which undergoes less extensive metabolism) being extensively metabolized in the liver [108], more quantities of glucose than fructose pass through the liver unmetabolized [108]. With the activation of glucokinase by fructose, more of the unmetabolized glucose might be taken up from the circulation into the liver. This might further contribute to improved glycemic control. With honey supplementation, the fructose in honey might enhance hepatic glucose uptake and glycogen synthesis and storage resulting in improved glycemic control in diabetes mellitus. Compelling evidence indicates that diabetes mellitus is commonly associated with hepatic dysfunction or abnormalities such as elevations in serum alkaline phosphatase, aspartate aminotransferase and alanine aminotransferase [109]. Honey supplementation has been shown to exert a protective effect against streptozotocin-induced, carbon tetrachloride-induced or trichlorfon-induced liver damage [15, 63, 110]. In view of the important role of the liver in mediating glycemic control and the hypoglycemic effect of honey, the hepatoprotective effect of honey might be beneficial in diabetes mellitus. Considering that increased hepatic oxidative damage is documented in diabetes mellitus [111], honey supplementation might protect the liver against oxidative stress and damage [63, 112]. This hepatoprotective effect of honey might retard deterioration and improve hepatic functions and thus contribute to improved glucose homeostasis [99].
Recent findings indicate that fructose consumption is associated with a number of deleterious effects such as increased weight gain, hepatic de novo lipogenesis, hypertriglyceridemia, reduced insulin sensitivity, increased visceral adiposity and hepatic steatosis [43, 113-117]. Considering that these detrimental effects do not occur with acute use of fructose, it may suggest that the glycemic index is not directly important in evaluating the effects of foods or drinks containing fructose. Instead, the medium or long term effects of fructose-containing foods or drinks on plasma lipids may be more important than the acute effect in glycemia. It is also important to note that fructose is not the same as honey. Honey is quite unique in the sense that it contains more than 181 constituents including free radical scavenging and antioxidant compounds [8, 9]. Fructose is just one of these numerous constituents. As evidence has shown, antioxidants do not cause weight gain and hepatic deleterious effects associated with fructose [118]. Instead, antioxidants reduce weight gain and ameliorate these abnormalities of lipids [46]. Similarly, reduced weight gain and anti-lipidemic effect has been demonstrated for honey in rats [119], diabetic human subjects [120] or no change in body weight in overweight or obese subjects [121]. It is also worth mentioning that these detrimental effects of fructose in the liver are not associated with low or moderate doses of fructose but with high doses of fructose consumption [117, 122, 123]. The lack of deleterious effects of honey in the liver, in spite of its fructose content, may be due to the health beneficial effects of its numerous bioactive constituents which counter potential deleterious effects of fructose. It may also indicate that the amount of fructose in honey is relatively low compared to the levels in many sugar- or fructose-sweetened beverages.
Effect of honey on glycemic control (glucose, fructosamine and glycosylated hemoglobin)
The measurements of blood glucose, fructosamine and glycosylated hemoglobin are commonly employed to assess the extent of glycemic control in diabetes mellitus [124]. Unlike fasting blood glucose, fructosamine and glycosylated hemoglobin measure glycemic control over a period of two to three weeks and eight to twelve weeks, respectively [66]. Some of the studies which have investigated the effect of honey on glycemic control are highlighted below.
Evidence from animal studies
Honey was reported to produce lower glycemic response in both diabetic and non-diabetic rabbits [125]. We and others have shown that honey supplementation significantly reduced blood glucose concentrations in alloxan-induced diabetic rats [126] and STZ-induced diabetic rats [16, 61]. Our data and those of other authors also indicated that the hypoglycemic or health beneficial effects of honey might be dose-dependent [9, 16]. As regards the effect of honey on the levels of fructosamine or glycosylated hemoglobin, limited data are available. Chepulis and Starkey reported that chronic honey supplementation reduced glycosylated hemoglobin in non-diabetic rats [127]. In one of our published articles, we reported that administration of honey to STZ-induced diabetic rats reduced significantly serum concentrations of fructosamine [66]. The study also showed that the combination of antidiabetic drugs, glibenclamide or metformin, with honey resulted in further reductions in serum concentrations of both glucose and fructosamine in STZ-induced diabetic rats [66]. Even though studies demonstrating the effect of honey on glycemic control are still limited, these available data indicate that honey has a potential to lower elevated blood glucose, fructosamine or glycosylated hemoglobin.
Evidence from clinical studies
Honey, compared with dextrose, sucrose or other sweeteners, was reported to attenuate postprandial glycemic response in non-diabetic volunteers [128]. In healthy human subjects, natural honey produced minimal increment (20%) compared to simulated honey and D-glucose which elevated the blood glucose levels by 47% and 52%, respectively after 60 minutes [129]. The study further showed that after 180 minutes, the reduction in blood glucose levels following D-glucose consumption was 20%, whereas it was twice lower (9.75%) following natural honey consumption [129]. Another related study also showed that honey supplementation in healthy subjects resulted in lower serum glucose concentrations and glycemic response than honey-comparable glucose-fructose solution did [81]. Other authors also reported reduced serum glucose levels in non-diabetic subjects administered honey [121, 130].
In patients with diabetes mellitus, studies showed that honey supplementation considerably reduced postprandial glycemic response or elicited much lower rise in plasma glucose than other sugars or sweeteners did [128, 131]. Similarly, honey administered orally or via inhalation was reported to reduce considerably the concentrations of blood glucose in patients with type 2 diabetes mellitus [22, 120]. Compared to sucrose, honey was shown to produce lower glycemic and peak incremental indices in type 1 diabetic patients [130]. A recent study also showed that honey reduced hyperglycemia in children with type 1 diabetes mellitus [132]. A similar glucose-lowering effect of honey was also reported in subjects with impaired glucose tolerance or patients with mild diabetes [133]. With regard to the effect of honey on serum concentrations of fructosamine or glycosylated hemoglobin, there is a dearth of data. Conversely, as reported by Bahrami and co-workers, honey supplementation for 8 weeks caused increased glycosylated hemoglobin levels in patients with type 2 diabetes mellitus [120]. The elevated levels of glycosylated hemoglobin may be due to the high dose of honey administered to the diabetic patients [134].
However, in spite of the evidence which demonstrates the hypoglycemic effect of honey, some studies found no beneficial effect of honey on hyperglycemia in type 2 diabetic patients [135, 136]. Similarly, in rodents, Nemoseck et al. found no significant difference in the concentrations of glucose and fructosamine in non-diabetic rats fed honey, a honey-based diet or sucrose [119]. Likewise, we reported that honey supplementation in non-diabetic rats did not alter the serum concentrations of glucose and fructosamine [66]. The lack of significant beneficial effect of honey on glucose, fructosamine and glycosylated hemoglobin in non-diabetic rats may be due to the short duration of honey supplementation or feeding. A study by Chepulis [137] showed that honey feeding in non-diabetic rats for 6 weeks reduced % weight gain only. In contrast, in another related follow-up study, honey feeding in non-diabetic rats for 52 weeks resulted in significantly reduced % weight gain, body fat and glycosylated hemoglobin while HDL cholesterol level was increased [127].
Similar to the data obtained in animal studies, honey supplementation in diabetic patients or in subjects with impaired glucose tolerance lowers hyperglycemia. Even though honey consumption causes short-term hyperglycemia, compelling evidence indicates that the hyperglycemia resulting from honey consumption is significantly less than that following the consumption of other common sugars. Thus, honey consumption or its addition to carbohydrate diets will still be beneficial in individuals with diabetes. With the exception of data published by Bahrami et al. [120], most other clinical studies did not measure fructosamine or glycosylated hemoglobin in diabetic patients. Thus, it is difficult to ascertain the actual effect of honey on elevated levels of fructosamine or glycosylated hemoglobin in diabetic patients. Therefore, there is a need for clinical studies that investigate both the short and long term effect of honey on fructosamine and/or glycosylated hemoglobin in diabetic patients.
Effect of honey on glucose-regulating hormones and pancreas
A number of substances (both natural and synthetic) with antidiabetic effect are known to modulate key glucose-regulating hormones especially insulin [138]. In healthy subjects, compared to dextrose, honey supplementation has been shown to elicit lower increments in serum insulin and C-peptide levels [139]. A study by Munstedt et al. showed that honey, compared to honey-comparable glucose-fructose solution, produced significantly lower serum insulin and C-peptide concentrations in healthy men [82]. In diabetic patients, honey supplementation was shown to increase insulin concentrations more than sucrose did [139]. In another study, it was reported that type 2 diabetic patients administered honey had reduced insulin resistance [136]. Similarly, in STZ-induced diabetic rats, honey supplementation was associated with considerable improvement in pancreatic islets as well as increased serum insulin levels [18, 66]. In general, as these studies have revealed, when there is insulin resistance, honey supplementation reduces or ameliorates insulin resistance [136]. On the other hand, with impaired pancreatic function and low levels of insulin, honey improves islets and increases insulin concentrations [18, 66]. These findings suggest that the effect of honey on insulin and/or C-peptide is dependent on their circulating concentrations.
Effect of honey on lipid metabolism
Diabetes mellitus is frequently associated with impaired lipid metabolism such as elevated cholesterol and triglycerides (TG) as well as impaired lipoprotein synthesis or metabolism [2]. In healthy sheep, inhalation or intrapulmonary administration of honey was reported to ameliorate lipid abnormalities, though most of the parameters except TG were not statistically significant compared with dextrose [110]. In rats fed honey, reduced TG [140] or increased high density lipoprotein (HDL) cholesterol [127] was reported. A study found that rats fed honey had lower epididymal fat weight and TG while non-HDL cholesterol was higher [119]. We also reported that honey supplementation significantly increased HDL cholesterol while it reduced TG and very low-density lipoprotein (VLDL) cholesterol in STZ-induced diabetic rats [66]. Additionally, the study showed that combination of glibenclamide with honey resulted in further reductions in TG and VLDL cholesterol while HDL cholesterol, total cholesterol (TC) and low-density lipoprotein (LDL) cholesterol were slightly elevated in STZ-induced diabetic rats. On the other hand, the combination of metformin with honey resulted in further reductions in TG, TC, LDL cholesterol and VLDL cholesterol whereas HDL cholesterol was slightly reduced in STZ-induced diabetic rats [66].
In healthy subjects as well as patients with hyperlipidemia, honey consumption reduced TC, LDL cholesterol and C-reactive proteins while it increased HDL cholesterol [139]. However, the study found significant reductions in some of the elevated lipid parameters in hyperlipidemic subjects only [139]. A study by Yaghoobi et al. found that honey supplementation produced only minimal reductions in TC, LDL cholesterol and triacylglycerole in subjects with normal or elevated parameters but significantly reduced triacylglycerole in subjects with elevated values [121]. Likewise in subjects with elevated cholesterol, the beneficial effect of honey on TC or TG was not observed except that it prevented the rise in LDL cholesterol in women [141]. In type 2 diabetic patients, honey was reported to reduce TG [136]. Similarly, a recent study showed that honey supplementation in type 2 diabetic patients for 8 weeks significantly reduced TG, TC, LDL cholesterol, LDL/HDL ratio and increased HDL cholesterol compared with baseline data [120]. However, compared with the untreated diabetic patients, no significant effect of honey was found except for TC [120]. Taken together, these data suggest that honey supplementation could ameliorate lipid abnormalities in both animal and human diabetes mellitus.
Effect of honey on renal and hepatic functions as well as other biochemical parameters
In addition to impaired lipid metabolism, abnormalities caused by reduced or impaired functions of liver and kidney have been documented in diabetes mellitus [109, 142]. Findings from a study performed in healthy sheep showed that administration of honey increased serum protein, albumin, hemoglobin, white blood cell counts and neutrophil percentage, while it decreased blood urea nitrogen [110]. We also reported that honey supplementation in diabetic rats considerably increased serum albumin and albumin/globulin ratio while serum bilirubin was markedly reduced [66]. The study showed that honey tended to lower serum creatinine and urea in diabetic rats. Further analysis of some of the parameters showed that combination of antidiabetic drugs with honey resulted in synergism [66]. This synergistic effect was evident from the significantly reduced serum creatinine and bilirubin in the honey + glibenclamide-treated or honey + metformin-treated diabetic rats [66]. Similarly, we reported that honey supplementation in diabetic rats significantly reduced elevated levels of hepatic enzymes including aspartate transaminase (AST), alanine transaminase (ALT) and alkaline phosphatase (ALP), suggestive of hepatoprotective effect of honey in diabetes mellitus [15]. A study by Al-Waili showed that the activities of several enzymes such as AST, ALT, ALP, lactic acid dehydrogenase (LDH), creatinine kinase (CK) and glutathione reductase (GR) were markedly or moderately reduced in healthy individuals administered honey [143]. The author also reported that honey consumption in healthy subjects increased considerably the serum concentrations of copper and iron while the levels or percentages of zinc, magnesium, vitamin C, eosinophils and monocytes were moderately increased [143]. Calcium, phosphorus, ferritin, blood sugars and immunoglobulin E concentrations were either markedly or mildly reduced following honey supplementation [143]. Similarly, honey was shown to increase urinary creatinine and nitrite excretion while it reduced renal excretion of prostaglandin E2, prostaglandin F2 alpha and thromboxane B2 in healthy volunteers [144]. Findings from a recent study suggest that honey may exert an inhibitory effect on platelet aggregation as well as blood coagulation [145]. These findings suggest that, similar to the anti-lipidemic effect of honey, honey supplementation in both rodents and human subjects has the potential to ameliorate or restore renal and hepatic functions as well as other biochemical parameters.
Effect of honey on appetite-regulating hormones, body weight and food/energy intake
Certain hormones such as leptin, ghrelin and peptide YY are recognized for their role in modulating satiety, appetite, calorie intake, energy expenditure and body weight [146, 147]. At the moment, only very few studies have investigated the effect of honey on appetite-regulating hormones - leptin, ghrelin and peptide YY. In rats, a recent study reported that the levels of leptin were considerably lower in rats administered honey than in those fed sucrose [119]. Similarly, compared with sucrose-containing diet, honey was reported to delay postprandial ghrelin release and enhance total peptide YY response in healthy human subjects [148]. Even though the data are still limited, fructose in honey may contribute to the modulating effect of honey on appetite-regulating hormones [35, 149].
As regards the effect of honey on body weight, available data (especially in experimental studies) are inconsistent. Weight gain or percentage weight gain was reported to be significantly lower in honey-fed healthy rats than in sucrose-fed rats [127, 137]. Another study also reported a considerably lower weight gain and food/energy intake in honey-supplemented non-diabetic rats [119]. In contrast, in most of our previous studies, no significant effect of honey on body weight in non-diabetic rats was observed [16, 18, 62, 66, 150]. However, Fasanmade and Alabi found that honey supplementation significantly increased body weight in non-diabetic and fructose-fed rats [126]. In alloxan-induced or STZ-induced diabetic rats (commonly characterized by weight loss), honey administration considerably prevented weight loss or improved body weight [16, 18, 62, 66, 126, 150]. As previously reported, we did not find any significant effect of honey on food intake in both diabetic and non-diabetic rats [16, 19, 66]. Although there are limited data in clinical studies, available evidence suggests that honey supplementation in human subjects is associated with reduced weight gain. In overweight or obese human subjects, Yaghoobi et al. reported that honey consumption produced a mild reduction in body weight and body fat [121]. Bahrami and colleagues showed that honey supplementation in type 2 diabetic patients significantly reduced body weight [120].
Although the mechanisms by which honey decreases weight gain are still not fully understood, findings from some recent studies suggest that honey might reduce weight gain via modulation of appetite-regulating hormones such as leptin, ghrelin and peptide YY [119, 148]. Furthermore, based on findings which showed lack of significant difference in food efficiency ratio (FER) in sucrose- and honey-fed rats, reduced food intake might contribute considerably to reduced weight gain in honey-fed rats [119]. Fructose and oligosaccharides which are present in honey might also contribute to reduced body weight and food intake in honey-fed rats [35, 43, 46, 151]. Besides, available evidence indicates that honey might reduce body weight through reduced digestion and absorption of protein and increased fecal nitrogen output [137]. Considering the overwhelming evidence which indicates that honey increases plasma antioxidants and ameliorates oxidative stress in tissues [16, 18, 19, 143, 150, 152, 153], the antioxidant effect of honey might also contribute to reduced weight gain [118].
Relative strengths and weaknesses of some of these studies
It seems important to briefly highlight the relative strengths and weaknesses of studies that investigated the hypoglycemic or antidiabetic effect of honey. Some of these studies investigated the acute effect of honey [82, 83, 129, 133, 135, 136, 154, 155] whereas only very few studies evaluated the effect of honey over a long duration [120]. This may influence the outcome, leading to significant findings in some studies and lack of significant results in others. Investigating the acute effect of honey in blood glucose in subjects with diabetes mellitus does not provide detailed and valid findings because diabetes mellitus is a chronic disease. Some studies could not compare and differentiate if honey samples, despite differences in their fructose content and fructose to glucose ratios, were better than common sugars due to lack of controls [156]. In contrast, others could differentiate the differences between various honey samples as well in comparison with common sugars or artificial honey due to inclusion of controls [82, 83, 121, 129, 130, 135, 139, 155]. Some studies were performed in healthy and non-diabetic subjects [82, 83, 139, 155, 156] while others used impaired glucose tolerant or diabetic subjects [130, 133, 135, 136, 139]. As we reported earlier, in the majority of our studies, honey did not alter or affect the blood glucose in non-diabetic rats but only in diabetic rats [16, 18, 62, 66, 151]. Therefore, the inclusion of different subjects with or without diabetes may influence the findings. Among the diabetic subjects, some authors recruited type 1 diabetic patients [130] while others enlisted type 2 diabetic subjects [120]. As a result of the differences in the etiologies of these two types of diabetes, compared to type 1 diabetic subjects, type 2 diabetic patients are more likely to respond to the antidiabetic effect of honey [120]. This difference in study participants may influence the data obtained as well as the deductions from those data.
Besides, some studies used doses of honey that ranged from low [135, 136, 154], moderate [155] to high doses [82, 83, 120, 121, 129, 130]. The fructose proportion in relation to glucose in the honey samples also differed. While the fructose content and fructose to glucose ratios in some honeys were within the stipulated or normal range [66, 82, 83, 156], some studies used honeys with low fructose and high glucose levels as well as unusually low fructose to glucose ratio [120]. Fructose is a potential hypoglycemic constituent of honey [35]. Hence, diabetic patients administered honey containing usual fructose amounts are likely to demonstrate better improvements in glycemic control than those administered honey with unusually low fructose content [120]. The sample size also differed. While some studies recruited small or moderate number of subjects or patients [82, 83, 130, 135, 136, 139, 154, 155], only very few studies recruited larger or adequate number [120, 121]. Improper study design was another major problem. Most studies were not randomized control trials and/or did not have definite criteria for enlisting participants, whereas some (few) studies were randomized control trials and/or had well defined inclusion and exclusion criteria [82, 83, 120]. Additionally, the measurement of glycemic control (fructosamine or glycosylated hemoglobin), an important parameter, was not measured in most of these studies with the exception of few [66, 120]. Those are some of the weaknesses and strengths observed in these studies. These differences in study designs including study participants and the variations among honeys lead to difficulties in comparing the data obtained in a study with those of other studies. For instance, despite the fact that some of the studies were properly randomized with controls and had well defined inclusion and exclusion criteria, findings from such studies cannot be compared due to differences in the doses of honey used, honey constituents, types or nature of recruited subjects or patients, and other study design related issues [82, 120, 130]. Therefore, it is important to consider all these factors while making an inference on the ineffectiveness (or otherwise) of honey as an antidiabetic agent. Any of these factors may influence and give inaccurate data, either false positive or negative, resulting in misinterpretation of data.
Potential toxic effects of honey: honey intoxication and infant botulism
Before the main highlights of this review are summarized, two important toxic effects that may result from honey consumption - honey intoxication and infant botulism, will be discussed briefly. Honey intoxication (also known as mad honey disease, Rhododendrons poisoning or grayanotoxin poisoning) refers to a poisoning that results from the consumption of honey produced from the nectar of Rhododendrons [157]. Rhododendron plants can be found in many countries and regions such as Turkey, Japan, Nepal, North America and Brazil [158]. The first written account of honey intoxication dates back to the 4th Century BC when thousands of soldiers were poisoned after the consumption of honey produced from the nectar of Rhododendron luteum (Azalea pontica) in Greece [157, 158]. Several cases of honey poisoning have also been reported in Turkey, Germany and Austria [157, 159, 160]. The easy and rapid food transport, together with a renewed interest in consumption of natural foods all over the world may increase cases of honey poisoning [158]. It is suggested that while the general population may be vulnerable to honey intoxication, individuals who consume honey from (local) beekeepers who have only a few hives are at higher risk [160]. Commercially produced honey is considered safe or safer. Commercial processing of massive quantities of honey obtained from different farmers helps to dilute any toxic substances that may be present in honey [160]. This toxic substance is identified as a poisonous compound, grayanotoxin, found in rhododendron nectar [159]. Grayanotoxins belong to a group of toxins known as diterpenes which are polyhydroxylated cyclic hydrocarbons that do not have nitrogen [158, 159]. Consumption of between 5 and 30 g of mad honey may cause honey poisoning [158]. In most reported cases, honey intoxication is hardly critical and the patients usually recover within 24 hours without any intervention [159]. In most cases, discontinuation of mad honey intake is sufficient for patient recovery [160]. Some of the symptoms of honey intoxication, which are dose-dependent, include salivation, nausea, vomiting, sharp burning sensation in the throat, dizziness, excessive perspiration, abnormal sensations, breathing problems and progressive muscular weakness [157, 159, 160]. However, severe honey intoxication may lead to life threatening complications such as atrioventricular block, cardiac arrhythmia, bradycardia, low blood pressure and shock [157, 159]. This may necessitate fluid supplementation and treatment of bradycardia with atropine to restore severe low blood pressure. The use of a pacemaker or treatment with vasopressors may be required but only rarely [157, 159].
Infant botulism is a neuroparalytic disease characterized by a blockade of voluntary motor and autonomic functions [161]. It was first recognized in the United States in the late 1976 [161]. Several cases have since been reported in USA, Argentina, Australia, Italy, Canada, Japan, China and India [161]. Infant botulism affects mostly healthy children less than one year old. It is caused by a heat-labile neurotoxin produced by Clostridium botulinum [161, 162]. It occurs when infants ingest Clostridium botulinum spores which grow and produce toxin after it has colonized the infant's gut [161]. The most common symptoms include constipation, poor sucking reflex, weak cry, irritability, lack of facial expression, hypotonia, hyporeflexia, progressive to generalized weakness, respiratory failure and loss of head control [162]. The Clostridium botulinum spores are commonly found in the environment, in the dust, inside or outside the house and in the soil [161, 163]. The exposure of infants to spores of Clostridium botulinum is increased by factors such as staying in a windy site, soil disruption caused by farming activities and disturbing the soil, as in a building site [163, 164]. More than 1,000 cases have been reported and intake of honey has been linked to infant botulism [161, 163, 165, 166]. Clostridium botulinum spores and toxins have also been detected in many honey samples [163, 165]. Although it remains unclear how Clostridium botulinum finds its way into the honey, the honeybee may carry and bring the Clostridium botulinum spores to the hive [161, 165]. Also being ubiquitous, the Clostridium botulinum spores may easily be found around the hive as well. At the moment, even though the exposure to botulinum spores is widespread and several host factors may contribute to the cause of infant botulism, consumption of honey is the only food-borne related case. Honey, therefore, is considered an avoidable source of Clostridium botulinum spores [161, 163, 165, 167]. This has prompted the American Academy of Pediatrics (AAP), the Centers for Disease Control (CDC) and the Food and Drug Administration (FDA) to recommend that honey not be given to infants who are less than 1 year old [163, 165, 166].
Antidiabetic effect of honey: Potential mechanisms of action based on the non-antioxidant constituents in honey
Considering that honey is reported to contain at least 181 substances [8], the exact mechanism of its antidiabetic effect is complex and therefore will necessitate detailed investigation [16]. Even though the mechanisms by which honey improves glycemic control and other diabetic profiles remain unclear, available evidence suggests that fructose in honey may modulate the hypoglycemic or antidiabetic effect of honey [35]. Certain varieties of honey have been reported to increase plasma concentrations of fructose in healthy humans [83]. Similarly, evidence indicates that the fructose content of honey negatively correlates with glycemic index [4, 9, 154]. Small amounts of fructose have been reported to reduce blood glucose through increased hepatic glucose uptake by activating glucokinase [168]. The beneficial effect of fructose administration on glycemic control has also been documented in patients with type 2 diabetes mellitus [169].
In view of the fact that sucrose and honey contain similar proportions of fructose and glucose, data which indicate that honey improves glycemic control much better than sucrose does suggest that the hypoglycemic or antidiabetic effect of honey cannot be attributed entirely to fructose. Honey is enriched in oligosaccharides such as sucrose, maltose, trehalose, turanose, panose,1-kestose, 6-kestose and palatinose [9, 80]. Evidence suggests that a number of oligosaccharides present in honey might play a role in the antidiabetic effect of honey [80]. Palatinose-based balanced formula has been shown to improve glycemic response in patients with impaired glucose tolerance [170] or produce a lower blood glucose levels in healthy subjects [96]. Similar beneficial effects of palatinose on glucose and lipd metabolism in rodents have also been reported [171, 172]. The oligosaccharides in honey may contribute to the antidiabetic effect of honey either via modulation of gut microbiota [80, 88, 90, 92] or through the systemic effects of oligosaccharides [80, 173].
Honey contains a number of mineral elements such as zinc, selenium, copper, calcium, potassium, chromium, manganese and so on [4, 9]. Some of these minerals such as chromium are recognized for their role in the reduction of elevated blood glucose, maintenance of normal glucose tolerance and insulin secretion from the pancreatic β-cells [174, 175]. Other studies have also shown that copper and zinc can improve insulin sensitivity thereby decreasing blood glucose levels [176, 177]. Even though the amounts of these minerals in honey may be low [4, 9], it is suggested that daily ingestion of honey might achieve adequate concentrations of these minerals and thus exert pharmacological responses [66]. Coupled with the evidence which demonstrates increased serum concentrations of these minerals after honey supplementation [143], these ions might also contribute to the antidiabetic effect of honey [176, 178]. Increased C-peptide and insulin secretion [66, 132] and modulation of appetite-regulating hormones such as leptin, ghrelin and peptide YY [119, 148, 179] as a result of honey supplementation may also contribute to improved glycemic control. With these proposed mechanisms of action, we have not excluded the prospect of a yet to be identified substance or substances in honey contributing to or mediating the antidiabetic effect of honey.
Antidiabetic effect of honey: Potential mechanisms of action based on the free radical scavenging constituents and antioxidant effect of honey
Compelling evidence implicates the role of oxidative stress in the pathogenesis and/or deterioration of glycemic control in diabetes mellitus [180, 181]. Increased glucose uptake in both skeletal muscle and adipose tissue enhances reactive oxygen species (ROS) generation and oxidative stress which in turn impairs glucose uptake and glycogen synthesis [182]. Oxidative stress causes insulin resistance through impaired insulin signaling pathways such as interference with insulin receptor, insulin receptor substrate-1 and protein kinase B/Akt [181, 183-186]. Thus, through its antioxidant effect [153], honey might enhance insulin sensitivity in the liver and muscle thereby increase glucose uptake resulting in reduced hyperglycemia. The pancreas expresses low level of antioxidant enzymes, which makes it vulnerable to oxidative stress [187]. The toxic effect of hyperglycemia in the β-cells, a condition referred to as glucotoxicity, is mediated via oxidative stress [188]. The role of glucotoxicity is implicated in the declined β-cell function [188]. A number of studies have demonstrated that ROS or oxidative products impair insulin secretion in pancreatic β-cells, inhibit glucose-induced insulin secretion and biosynthesis, deplete insulin content of the pancreatic β-cells and increase β-cell apoptosis [189-191]. The beneficial effect of antioxidants in β-cells has been reported [190, 192]. A number of studies have shown that honey can scavenge free radicals [20, 21, 153, 190]. Thus, honey supplementation may ameliorate oxidative stress in the pancreas, protect the pancreas against oxidative damage and thus enhance insulin secretion resulting in improved glycemic control [17-19, 62].
Diabetes mellitus is characterized by impairments in lipid metabolism leading to lipid abnormalities such as elevated levels of low-density lipoproteins (LDLs). The increased oxidative environment in diabetes further enhances the oxidation and glycation of LDLs to form oxLDLs, which is implicated in endothelial dysfunction [193]. The antioxidant and free radical scavenging effects of honey may help to reduce the oxidative milieu and enhance antioxidant defenses in diabetes [16, 19, 20, 153, 194-196]. This may reduce vascular ROS, inhibit inactivation and increase bioavailability of NO, suppress the formation of oxLDLs and thus reduce or prevent deterioration of endothelial function [20, 197-199]. These potential antioxidant effect and free radical scavenging activities of honey on LDL oxidation [16, 19, 20, 194-196] and its potential anti-lipidemic effect [66, 120, 121, 127, 139] may delay or prevent the development of atherosclerosis. The beneficial effects of antioxidants including honey in glycemic control have also been demonstrated [16, 66, 199-203].
Evidence also indicates antioxidants may reduce protein glycosylation in diabetic subjects independently of changes in plasma glucose [204, 205]. Antioxidants have been shown to ameliorate oxidative stress and increase the levels of C-peptide and insulin as well as improve insulin resistance in diabetes mellitus [66, 199-201, 206-208]. Antioxidants may ameliorate intestinal oxidative stress and enhance BBM fluidity thereby promote GIT health which may impact positively on glycemic control in diabetes mellitus [56, 65]. In view of the role of liver in glucose homeostasis, the antioxidant effect of honey may ameliorate hepatic oxidative stress and damage [15, 111]. These hepatic effects may enhance liver functions and contribute to improved glycemic control.
Antidiabetic effect of honey: Additional beneficial effect in preventing diabetic complications via its antioxidant effect
Recent evidence indicates that hyperglycemia-induced mitochondrial oxidative stress is the underlying mechanism by which hyperglycemia induces cellular damage [209, 210]. This mechanism is now known to activate all the different pathogenic pathways implicated in the pathogenesis of diabetic complications [209, 210]. These pathogenic or mechanistic pathways include (1) polyol pathway; (2) formation of advanced glycation endproducts (AGEs); (3) protein kinase C (PKC) pathway; (4) hexosamine pathway; and (5) Poly-ADP ribose polymerase (PARP) pathway [209-211]. These pathways increase the consumption or utilization of NADPH and inhibit glucose 6-phosphate dehydrogenase (GAPDH) [212]. NADPH is an obligate co-factor for several intracellular enzymes and metabolic processes. For instance, GR requires NADPH for the regeneration of reduced glutathione (GSH), a potent endogenous scavenger of ROS. GAPDH is the rate-limiting enzyme of the pentose phosphate pathway necessary for generating reducing equivalents to the antioxidant defense system [212, 213]. These pathways (via intracellular NADPH depletion and intracellular GAPDH inhibition) play a major role in impairing antioxidant defenses and increasing the cellular susceptibility to oxidative stress and damage.
Nishikawa et al. classified these mechanisms of hyperglycemia-induced cellular damage into two groups [211]. The first group involves constant acute fluctuations in cellular metabolism which are temporary and reversible following restoration of normoglycemia [211]. This is where most of the anti-diabetic drugs are beneficial. The second group of mechanisms entails cumulative changes in long-lived macromolecules which are irreversible even after normoglycemia is restored [211]. Here, the anti-diabetic drugs have no or limited role to play [214-217]. This second group of mechanisms-induced irreversible changes or alterations, despite improved glycemic control, is explained by a phenomenon known as glycemic or metabolic memory [211, 218]. Oxidative stress is identified or recognized as the major mechanism by which glycemic or metabolic memory induces tissue damage [211, 215, 216, 218-220]. This is where the antioxidant effect of honey may complement its antidiabetic effect [16, 18-20, 62, 66, 153 194-196]. This might result in synergistic and beneficial effects in diabetic patients. Honey, via its antioxidant effect, may prevent depletion of intracellular NADPH and inhibition of intracellular GAPDH. These two effects may enhance antioxidant defenses in tissues or organs that are susceptible to oxidative stress-mediated diabetic complications. These include kidney (reducing diabetic nephropathy), retina (reducing diabetic retinopathy), nerve (reducing diabetic neuropathy) and heart (reducing diabetic cardiomyopathy). This assertion is further supported by findings in honey-supplemented diabetic rats [17-19, 62, 66, 153]. Honey supplementation may also increase the expression of cytoprotective genes through up-regulation of Nrf2 activity and expression [23, 221]. Diabetic patients are also likely to benefit more if honey is used as a complementary agent or combined with the conventional or standard anti-diabetic therapy [17-19, 62, 66].
The effects of honey in pre-clinical and clinical studies are summarized in tables 1 and 2, respectively. Figure 5 shows the proposed mechanisms of antidiabetic effect of honey.
Antidiabetic effect of honey: summary of key findings from pre-clinical studies.
| Study design | Summary of key findings | Ref. |
|---|---|---|
| Rats fed diets containing 65 g/100g combined fructose and glucose or a honey-based diet for 2 weeks | Statistically significant parameters: ↔ Glucose, ↑ Fructose, ↓ TG, ↑ Serum vitamin E, ↑ Serum vitamin E/TG, | [140] |
| Healthy sheep or sheep with CCl4-induced liver injury given honey infusion | Statistically significant parameters (honey-treated healthy sheep vs control): ↓ Glucose, ↓ GGT, ↓ AST, ↓ ALT, ↓ TG, ↑ Serum albumin, ↑ Serum protein, ↑ Hemoglobin, ↑ White blood cell, ↓ Uric acid, ↓ Creatinine Statistically insignificant parameters (honey-treated healthy sheep vs control): ↓ AST, ↓ ALT, ↓ ALP, ↓ Bilirubin, ↓ TC, ↑ Percentage of eosinophils and neutrophil Statistically significant parameters (honey-pretreated CCl4-injected sheep vs control): ↓ GGT, ↓ AST, ↓ ALT | [110] |
| Healthy, fructose-fed, alloxan-induced diabetic rats given 10ml honey/kg/5ml for 3 weeks | Statistically significant parameters ↓ Glucose in alloxan-induced diabetic rats, ↑ Body weight gain in healthy and fructose-fed rats Statistically insignificant parameters ↓ Glucose in fructose-fed rats | [126] |
| Non-diabetic rats fed a diet containing 10 % honey for 6 weeks or 52 weeks | Honey feeding for 6 weeks: Statistically significant parameters ↓ % weight gain, Honey feeding for 6 weeks: Statistically insignificant parameters ↓ Food intake, ↓ Glycosylated hemoglobin, Honey feeding for 52 weeks: Statistically significant parameters ↓ Glycosylated hemoglobin, ↑ HDL cholesterol, ↓ % weight gain, ↓ body fat | [127, 137] |
| Non-diabetic & diabetic rats treated with honey (1.0g/kg/BW) orally once daily for 28 days | Statistically insignificant parameters (Honey versus non-diabetic control) ↔ All parameters Statistically significant parameters (Honey versus diabetic control) ↓ Glucose, ↓ Fructosamine, ↓ TG, ↓ VLDL cholesterol, ↓ Bilirubin, ↓ AST, ↓ ALT, ↓ ALP, ↑ Insulin, ↑ HDL, ↑ Albumin, ↑ Albumin/Globulin, ↑ body weight gain Diabetic + honey: Statistically insignificant parameters ↓ Creatinine, ↓ Urea, ↓ TC | [15, 66] |
| Healthy rats fed diets containing 20% carbohydrate from honey for about 33 days | Statistically significant parameters ↓ TG, ↑ Non-HDL cholesterol, ↓ Leptin, ↓ Body weight gain, ↓ Food/energy intake, ↓ Epididymal fat weight Statistically insignificant parameters ↓ Glucose, ↓ TC, ↑ HDL cholesterol, ↓ Adiponectin, ↓ CRP, ↓ MCP-1 | [119] |
↑ = increased, ↓ = reduced, ↔ = no significant effect, TC = total cholesterol, TG = triglycerides, HDL = high-density lipoprotein, LDL = low-density lipoprotein, VLDL = very low-density lipoprotein, AST = aspartate transaminase, ALT = alanine transaminase, ALP = alkaline phosphatase, GGT = Gamma-glutamyltransferase, CRP = C-reactive proteins, MCP - monocyte chemoattractant protein-1.
Antidiabetic effect of honey: summary of key findings from clinical studies.
| Study design | Summary of key findings | Ref. |
|---|---|---|
| Healthy subjects given a regular diet supplemented with honey (1.2 g/kg/BW once daily) dissolved in 250 ml of water for a period of 2-week | Statistically significant parameters ↑ Copper, ↑ Iron, ↓ Calcium, ↓ CK, ↓ AST, ↓ LDH, ↓ GR, Statistically insignificant parameters ↓ Glucose, ↑ Vitamin C, ↑ Zinc, ↑ Magnesium, ↓ Phosphorus, ↓ Ferritin, ↓ ALT, ↓ ALP | [143] |
| Healthy subjects given solutions containing dextrose (70 g), artificial honey (30 g glucose + 40 g fructose) or natural honey (80 g) for 15 days | Statistically significant parameters (Honey versus dextrose within the first 2 hours) ↓ Glucose, ↓ insulin, ↓C-peptide Statistically insignificant parameters (15-day daily honey consumption versus baseline) ↓ Glucose, ↓ TC, ↓ LDL cholesterol, ↑ HDL cholesterol, ↓ TG, ↓ CRP, ↓ Homocysteine | [139] |
| Healthy men with no history of metabolic disorders given 75 g of honey | Statistically significant parameters ↓ Glucose, ↓ Insulin, ↓ C-peptide | [82] |
| Healthy, nonobese women given 450 kcal honey- or sucrose-containing breakfasts | Statistically significant parameters ↓ Glucose, ↓ Ghrelin, ↑Peptide YY | [148] |
| Overweight or obese subjects given honey (70 g in 250 mL tap water) orally once daily for 30 days | Statistically significant parameters ↓ Glucose, ↓ Triacylglycerole, ↓ Hs-CRP, ↓ BMI, ↓ Body weight Statistically insignificant parameters ↓ body fat %, ↓ LDL cholesterol, ↓ TC, ↑ HDL | [121] |
| Patients with lipid abnormalities given solutions containing artificial honey or natural honey for 15 days | Statistically significant parameters (15-day daily honey consumption versus baseline) Natural honey: ↓ TC, ↓ LDL cholesterol, ↓ CRP Statistically insignificant parameters (Honey versus baseline within the first 3 hours) Artificial honey: ↑ TC, ↑ LDL cholesterol, ↑ TG Natural honey: ↓ TC, ↓ LDL cholesterol, ↓ TG | [139] |
| Type 2 diabetic patients dextrose (70 g), sucrose (30 g) or natural honey (60%, 30 or 90 g) | Statistically significant parameter (Honey versus dextrose within the first 3 hours) ↓ Glucose Statistically insignificant parameters (Honey versus sucrose within the first 3 hours) ↓ Glucose, ↑ Insulin | [22, 139] |
| Type 2 diabetic patients administered honey (first 2 weeks, 1 g/kg/day; second 2 weeks, 1.5 g/kg/day; third 2 weeks, 2 g/kg/day; and last 2 weeks, 2.5 g/kg/day) orally for 8 weeks | Statistically significant parameters (versus baseline) ↓ Glucose, ↑ Glycosylated hemoglobin, ↓ TC, ↓ LDL cholesterol, ↑ HDL cholesterol, ↓ TG, ↓ LDL/HDL ratio, ↓ Body weight Statistically significant parameters (versus control) ↓ Glucose, ↓ LDL/HDL ratio, ↓ Body weight | [120] |
↑ = increased, ↓ = reduced, ↔ = no significant effect, TC = total cholesterol, TG = triglycerides, HDL = high-density lipoprotein, LDL = low-density lipoprotein, VLDL = very low-density lipoprotein, AST = aspartate transaminase, ALT = alanine transaminase, ALP = alkaline phosphatase, LDH = lactic acid dehydrogenase, CK = creatinine kinase, GR = glutathione reductase, CRP = C-reactive proteins, MCP-1 = monocyte chemoattractant protein-1, Hs-CRP = high-sensitivity C-reactive protein.
Proposed mechanisms of action of antidiabetic effect of honey.
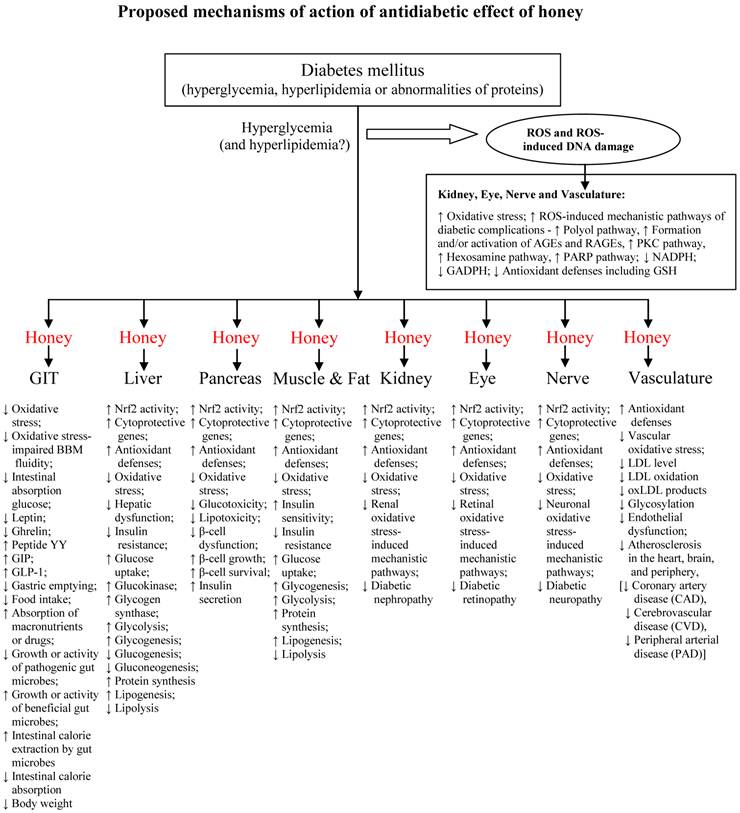
Impacts and future perspectives on the use of honey as an antidiabetic agent
A closer look at the proposed mechanisms of action of antidiabetic effect of honey (figure 5) suggests the following:
- Honey, through its GIT effects, would possess characteristic effects of α-glucosidase inhibitors such as acarbose.
- Honey, through its hepatic and/or pancreatic effects, would possess characteristic effects of insulin secretagogues such as sulfonylureas (glibenclamide) as well as repaglinide and nateglinide.
- Honey, through its hepatic and muscular amelioration of oxidative stress-induced insulin resistance, would possess characteristic effects of thiazolidinediones and biguanides such as metformin.
- Honey, through its effects on incretin and appetite-regulating hormones, would possess characteristic effects of dipeptidyl peptidase-IV inhibitors such as sitagliptin and GLP-1 mimetic such as exenatide.
- Honey, through its anti-lipidemic effects, would possess characteristic effects of anti-obesity drugs.
All these clearly indicate that honey possesses characteristics of most of the currently prescribed antidiabetic drugs and suggest it is a novel antidiabetic agent.
At the moment, the underlying pathogenic mechanism remains unknown. However, evidence suggests that insulin resistance precedes impaired glucose tolerance and onset of type 2 diabetes mellitus in type 2 diabetic patients [180, 181]. In view of the compelling evidence which implicates the role of oxidative stress in the pathogenesis of insulin resistance [181, 183-186]. Honey supplementation in individuals with impaired glucose tolerance or who are at pre-diabetic stage may delay or prevent the development of diabetes mellitus. In individuals at the early stages of or with mild type 2 diabetes mellitus, honey supplementation in combination with regular exercise and dietary management may suffice to maintain glycemic control. Honey as an antidiabetic agent is likely to be more effective and beneficial if combined with conventional antidiabetic therapy. Therefore, in patients with moderate or severe type 2 diabetes mellitus, honey supplementation in combination with one or two (instead of multiple) antidiabetic drugs may be sufficient to achieve glycemic control [17, 18, 62, 66]. Considering that oxidative stress-induced diabetic complications are similar in both type 1 and type 2 diabetic patients, the use of honey in combination with insulin therapy would also be beneficial in type 1 diabetic patients. If combined with conventional antidiabetic drugs or insulin, the use of honey may necessitate lower doses of these synthetic drugs or insulin to achieve similar glycemic control. Supplementation with honey may also help to counteract and/or minimize the adverse effects such as hypoglycemia and weight gain associated with some of these drugs. Its anti-lipidemic effect will also be beneficial in diabetic patients [66, 119-121, 139]. In addition to its antidiabetic effect, honey in combination with antidiabetic drugs may also help to delay or prevent the development of hypertension in diabetic patients or reduce elevated blood pressure in diabetic hypertensive patients [23, 150, 203, 222].
The potential of honey to be used in the prevention (prophylactic effect) and treatment (antidiabetic effect) of diabetes mellitus holds much promise in view of the rising prevalence and complications of this disorder. The goal of conventional antidiabetic therapy is to achieve glycosylated hemoglobin target of ≤ 7.0% [223, 224]. While this is beneficial [223, 224], evidence indicates that it is difficult to achieve and maintain this glycemic goal [225]. In many patients, this will necessitate the use of multiple drugs [226]. This further exposes patients to toxicities of these drugs [225, 226]. The latest findings reveal that the intensive therapy of hyperglycemia, which benefits are limited to only microvascular complications, is associated with increased weight gain, severe hypoglycemia and increased mortality [227, 228]. In light of these limitations and adverse effects, combination of these drugs with honey might be a new and more effective therapeutic option in the management of diabetes mellitus in the near future. The recent statistics have shown that the majority of the newly diagnosed diabetes cases will emerge from developing countries [1]. The vast majority of whom cannot afford the high cost of these synthetic drugs [229, 230]. In such developing countries, considering the global availability of honey, honey may be one of the few sources of succor or relief among diabetics in the near future. Due to its sweetness, combination of antidiabetic therapy with honey may also help to improve patients' compliance. There is no doubt that as more compelling data become available in the future, the potential of honey to be used as a prophylactic and antidiabetic agent (especially in combination with conventional antidiabetic drugs) holds much promise. In the future, honey might become a valued commodity that will be adopted as self-medication by the general public. As research interest into the potential prophylactic and antidiabetic effects of honey intensifies, the discovery of biologically active antidiabetic constituents in honey is anticipated. With the aid of biotechnology or medicinal chemistry synthesis, there is a possibility to develop these identified bioactive principles into a pharmacologically active molecule or a drug candidate. In the future, honey or its identified drug candidate might become one of the lead antidiabetic agents to be added to the classes of antidiabetic drugs.
Conclusions and recommendations
There is no doubt that studies investigating the potential role of honey in the management of diabetes mellitus are at a relatively early stage. In spite of a dearth of data, these studies have overwhelmingly shown that honey is more tolerable than most common sugars or sweeteners in healthy subjects or patients with impaired glucose tolerance or diabetes mellitus. It is worth mentioning that honey consumption is associated with short-term hyperglycemia. However, due to its minimal incremental effect on blood glucose compared to other sweeteners or common sugars, consumption of honey or its addition to other carbohydrate diets is highly encouraged in diabetic patients. This is very important because it is well known that the majority of diabetic patients do not always adhere to the dietary guidelines such as the exclusion of simple sugars in the diabetic diets.
In animal models of diabetes, there is more compelling evidence in support of honey as a novel antidiabetic agent. However, at the moment, this does not seem to be the case in clinical diabetes mellitus. This is because, unlike in animal studies, most of the data obtained on the antidiabetic effect of honey in human subjects were significant or showed improved trends in comparison with baseline data only. The majority of the clinical studies lacked control groups. Among the few studies that did include controls, most of the data obtained in the honey-supplemented subjects or diabetic patients were not significant in comparison with the controls. The lack of significant data in some of the clinical studies with controls could largely be attributed to poor study designs or due to the fact that the studies were too preliminary. Considering that honey has a potential to be used as an antidiabetic agent, the following recommendations are made: there is a need for (1) well designed clinical studies that enlist a large sample size; (2) randomized clinical trials comprising diabetic patients (if possible, should be grouped into mild, moderate or severe) treated with graded doses of honey as well as controls; and (3) clinical studies investigating and comparing both the short-term and long-term effects of honey supplementation in diabetic patients.
Besides, the experimental findings which indicate that glibenclamide or metformin, the two most commonly prescribed antidiabetic drugs, combined with honey improves glycemic control and other metabolic abnormalities merit further studies in type 2 diabetic patients. In view of the similarities and differences between animal and human diabetes mellitus, it will be interesting and worthwhile to explore whether these experimental data can be reproduced and substantiated or perhaps refuted in diabetic human subjects. Besides, considering that most of the studies that investigated the antidiabetic effect of honey did not pay much attention to possible mechanisms of action, it will be desirable to ensure that future studies emphasize mechanisms by which honey improves glycemic control and ameliorates metabolic derangements in diabetes mellitus. These mechanism studies might also help our understanding on how a chemically bioactive substance or substances in honey could bypass various epithelial membrane barriers including GIT, undergo (extensive) hepatic metabolism, and still be able to elicit an antidiabetic effect including increased insulin secretion. Therefore, in order to ensure that these novel findings obtained in animal studies are successfully reproduced in clinical studies and translated to the benefits of diabetic patients, the significance of having rigorous and well-designed randomized controlled clinical trials cannot be overemphasized.
Acknowledgements
The studies on the effects of tualang honey were supported by grants from Universiti Sains Malaysia.
Competing Interests
The authors have declared that they have no personal or financial conflict of interest..
References
1. Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4-14
2. Kokil GR, Rewatkar PV, Verma A. et al. Pharmacology and chemistry of diabetes mellitus and antidiabetic drugs: a critical review. Curr Med Chem. 2010;17:4405-23
3. Roglic G, Unwin N. Mortality attributable to diabetes: estimates for the year 2010. Diabetes Res Clin Pract. 2010;87:15-9
4. Honey as nutrient and functional food. Bogdanov S. http://www.bee-hexagon.net
5. Wang J, Li QX. Chemical composition, characterization, and differentiation of honey botanical and geographical origins. Adv Food Nutr Res. 2011;62:89-137
6. Molan P. The limitations of the methods of identifying the floral source of honeys. Bee World. 1998;97:59-68
7. Ramírez-Arriaga E, Navarro-Calvo AL, Díaz-Carbajal E. Botanical characterisation of Mexican honeys from a subtropical region (Oaxaca) based on pollen analysis. Grana. 2011;50:40-5
8. Gheldof N, Wang XH, Engeseth NJ. Identification and quantification of antioxidant components of honeys from various floral sources. J Agric Food Chem. 2002;50:5870-7
9. Bogdanov S, Jurendic T, Sieber R. et al. Honey for nutrition and health: a review. J Am Coll Nutr. 2008;27:677-89
10. Beretta G, Gelmini F, Lodi V. et al. Profile of nitric oxide (NO) metabolites (nitrate, nitrite and N-nitroso groups) in honeys of different botanical origin: nitrate accumulation as index of origin, quality and of therapeutic opportunities. J Pharm Biomed Anal. 2010;53:343-9
11. Beretta G, Caneva E, Facino RM. Kynurenic acid in honey from arboreal plants: MS and NMR evidence. Planta Med. 2007;73:1592-5
12. Crane E. History of honey. In: (ed.) Crane E. Honey, a comprehensive survey. London: William Heinemann. 1975:439-88
13. Heitkamp K, Busch-Stockfisch M. [The pros and cons of honey--are statements about the effects of honey "scientifically verified"?]. Z Lebensm Unters Forsch. 1986;182:279-86
14. Rakha MK, Nabil ZI, Hussein AA. Cardioactive and vasoactive effects of natural wild honey against cardiac malperformance induced by hyperadrenergic activity. J Med Food. 2008;11:91-8
15. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Hepatoprotective effect of tualang honey supplementation in streptozotocin-induced diabetic rats. Int J Appl Res Nat Prod. 2012;4:37-41
16. Erejuwa OO, Gurtu S, Sulaiman SA. et al. Hypoglycemic and antioxidant effects of honey supplementation in streptozotocin-induced diabetic rats. Int J Vitam Nutr Res. 2010;80:74-82
17. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Antioxidant protective effect of glibenclamide and metformin in combination with honey in pancreas of streptozotocin-induced diabetic rats. Int J Mol Sci. 2010;11:2056-66
18. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Comparison of antioxidant effects of honey, glibenclamide, metformin, and their combinations in the kidneys of streptozotocin-induced diabetic rats. Int J Mol Sci. 2011;12:829-43
19. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Antioxidant protection of Malaysian tualang honey in pancreas of normal and streptozotocin-induced diabetic rats. Ann Endocrinol (Paris). 2010;71:291-6
20. Beretta G, Orioli M, Facino RM. Antioxidant and radical scavenging activity of honey in endothelial cell cultures (EA.hy926). Planta Med. 2007;73:1182-9
21. Beretta G, Granata P, Ferrero M. et al. Standardization of antioxidant properties of honey by a combination of spectrophotometric/fluorimetric assays and chemometrics. Anal Chim Acta. 2005;533:185-91
22. Al-Waili N. Intrapulmonary administration of natural honey solution, hyperosmolar dextrose or hypoosmolar distill water to normal individuals and to patients with type-2 diabetes mellitus or hypertension: their effects on blood glucose level, plasma insulin and C-peptide, blood pressure and peaked expiratory flow rate. Eur J Med Res. 2003;8:295-303
23. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Honey supplementation in spontaneously hypertensive rats elicits antihypertensive effect via amelioration of renal oxidative stress. Oxid Med Cell Longev. 2012;2012:1-14
24. Tan HT, Rahman RA, Gan SH. et al. The antibacterial properties of Malaysian tualang honey against wound and enteric microorganisms in comparison to manuka honey. BMC Complement Altern Med. 2009;9:1-8
25. Feas X, Estevinho ML. A survey of the in vitro antifungal activity of heather (erica sp.) organic honey. J Med Food. 2011;14:1284-8
26. Zeina B, Othman O, al-Assad S. Effect of honey versus thyme on Rubella virus survival in vitro. J Altern Complement Med. 1996;2:345-8
27. Kassim M, Achoui M, Mustafa MR. et al. Ellagic acid, phenolic acids, and flavonoids in Malaysian honey extracts demonstrate in vitro anti-inflammatory activity. Nutr Res. 2010;30:650-9
28. Fukuda M, Kobayashi K, Hirono Y. et al. Jungle honey enhances immune function and antitumor activity. Evid Based Complement Alternat Med. 2011;2011:1-8
29. The chemistry of carbohydrates found in food. http://www.medbio.info
30. Carbohydrates. http://www.reboundhealth.com/cms/topic-carbohydrates.html
31. Wright EM, Hirayama BA, Loo DF. Active sugar transport in health and disease. J Intern Med. 2007;261:32-43
32. Vaisman N, Niv E, Izkhakov Y. Catalytic amounts of fructose may improve glucose tolerance in subjects with uncontrolled non-insulin-dependent diabetes. Clin Nutr. 2006;25:617-21
33. Stanhope KL, Griffen SC, Bremer AA. et al. Metabolic responses to prolonged consumption of glucose- and fructose-sweetened beverages are not associated with postprandial or 24-h glucose and insulin excursions. Am J Clin Nutr. 2011;94:112-9
34. Kwon S, Kim YJ, Kim MK. Effect of fructose or sucrose feeding with different levels on oral glucose tolerance test in normal and type 2 diabetic rats. Nutr Res Pract. 2008;2:252-8
35. Erejuwa OO, Sulaiman SA, Wahab MS. Fructose might contribute to the hypoglycemic effect of honey. Molecules. 2012;17:1900-15
36. Moran TH, McHugh PR. Distinctions among three sugars in their effects on gastric emptying and satiety. Am J Physiol. 1981;241:R25-30
37. Kellett GL, Brot-Laroche E, Mace OJ. et al. Sugar absorption in the intestine: the role of GLUT2. Annu Rev Nutr. 2008;28:35-54
38. Kashimura J, Nagai Y. Inhibitory effect of palatinose on glucose absorption in everted rat gut. J Nutr Sci Vitaminol (Tokyo). 2007;53:87-9
39. Lina BA, Jonker D, Kozianowski G. Isomaltulose (Palatinose): a review of biological and toxicological studies. Food Chem Toxicol. 2002;40:1375-81
40. Thibault L. Dietary carbohydrates: effects on self-selection, plasma glucose and insulin, and brain indoleaminergic systems in rat. Appetite. 1994;23:275-86
41. Gregory PC, McFadyen M, Rayner DV. Relation between gastric emptying and short-term regulation of food intake in the pig. Physiol Behav. 1989;45:677-83
42. Henry RR, Crapo PA, Thorburn AW. Current issues in fructose metabolism. Annu Rev Nutr. 1991;11:21-39
43. Meirelles CJ, Oliveira LA, Jordao AA. et al. Metabolic effects of the ingestion of different fructose sources in rats. Exp Clin Endocrinol Diabetes. 2011;119:218-20
44. Lavin JH, Wittert GA, Andrews J. et al. Interaction of insulin, glucagon-like peptide 1, gastric inhibitory polypeptide, and appetite in response to intraduodenal carbohydrate. Am J Clin Nutr. 1998;68:591-8
45. Anderson GH, Woodend D. Effect of glycemic carbohydrates on short-term satiety and food intake. Nutr Rev. 2003;61:S17-26
46. Madero M, Arriaga JC, Jalal D. et al. The effect of two energy-restricted diets, a low-fructose diet versus a moderate natural fructose diet, on weight loss and metabolic syndrome parameters: a randomized controlled trial. Metabolism. 2011;60:1551-9
47. Bocarsly ME, Powell ES, Avena NM. et al. High-fructose corn syrup causes characteristics of obesity in rats: increased body weight, body fat and triglyceride levels. Pharmacol Biochem Behav. 2010;97:101-6
48. Jones HF, Butler RN, Brooks DA. Intestinal fructose transport and malabsorption in humans. Am J Physiol Gastrointest Liver Physiol. 2011;300:G202-6
49. Gouyon F, Caillaud L, Carriere V. et al. Simple-sugar meals target GLUT2 at enterocyte apical membranes to improve sugar absorption: a study in GLUT2-null mice. J Physiol. 2003;552:823-32
50. Miyamoto K, Hase K, Takagi T. et al. Differential responses of intestinal glucose transporter mRNA transcripts to levels of dietary sugars. Biochem J. 1993;295:211-5
51. Riby JE, Fujisawa T, Kretchmer N. Fructose absorption. Am J Clin Nutr. 1993;58:748S-53S
52. Ushijima K, Riby JE, Fujisawa T. et al. Absorption of fructose by isolated small intestine of rats is via a specific saturable carrier in the absence of glucose and by the disaccharidase-related transport system in the presence of glucose. J Nutr. 1995;125:2156-64
53. Fujisawa T, Riby J, Kretchmer N. Intestinal absorption of fructose in the rat. Gastroenterology. 1991;101:360-7
54. Fine KD, Santa Ana CA, Porter JL. et al. Mechanism by which glucose stimulates the passive absorption of small solutes by the human jejunum in vivo. Gastroenterology. 1994;107:389-95
55. Ariefdjohan MW, Martin BR, Lachcik PJ. et al. Acute and chronic effects of honey and its carbohydrate constituents on calcium absorption in rats. J Agric Food Chem. 2008;56:2649-54
56. Bhor VM, Sivakami S. Regional variations in intestinal brush border membrane fluidity and function during diabetes and the role of oxidative stress and non-enzymatic glycation. Mol Cell Biochem. 2003;252:125-32
57. Zoubi SA, Williams MD, Mayhew TM. et al. Number and ultrastructure of epithelial cells in crypts and villi along the streptozotocin-diabetic small intestine: a quantitative study on the effects of insulin and aldose reductase inhibition. Virchows Arch. 1995;427:187-93
58. Fedorak RN, Gershon MD, Field M. Induction of intestinal glucose carriers in streptozocin-treated chronically diabetic rats. Gastroenterology. 1989;96:37-44
59. Sharma SD, Sivakami S. Responses of intestinal and renal alpha-glycosidases to alloxan and streptozotocin-induced diabetes: a comparative study. Biochem Mol Biol Int. 1998;44:647-56
60. Shirpoor A, Ansari MH, Salami S. et al. Effect of vitamin E on oxidative stress status in small intestine of diabetic rat. World J Gastroenterol. 2007;13:4340-4
61. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Effects of Malaysian tualang honey supplementation on glycemia, free radical scavenging enzymes and markers of oxidative stress in kidneys of normal and streptozotocin-induced diabetic rats. Int J Cardiol. 2009;137:S45
62. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Effect of glibenclamide alone versus glibenclamide and honey on oxidative stress in pancreas of streptozotocin-induced diabetic rats. Int J Appl Res Nat Prod. 2011;4:1-10
63. Eraslan G, Kanbur M, Silici S. et al. Beneficial effect of pine honey on trichlorfon induced some biochemical alterations in mice. Ecotoxicol Environ Saf. 2010;73:1084-91
64. Faria A, Mateus N, de Freitas V. et al. Modulation of MPP+ uptake by procyanidins in Caco-2 cells: involvement of oxidation/reduction reactions. FEBS Lett. 2006;580:155-60
65. Faria A, Monteiro R, Pestana D. et al. Intestinal oxidative state can alter nutrient and drug bioavailability. Oxid Med Cell Longev. 2009;2:322-7
66. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Glibenclamide or metformin combined with honey improves glycemic control in streptozotocin-induced diabetic rats. Int J Biol Sci. 2011;7:244-52
67. Naito Y, Takagi T, Oya-Ito T. et al. Impaired gastric ulcer healing in diabetic mice: role of methylglyoxal. J Physiol Pharmacol. 2009;60(Suppl 7):123-30
68. Boehme MW, Autschbach F, Ell C. et al. Prevalence of silent gastric ulcer, erosions or severe acute gastritis in patients with type 2 diabetes mellitus - a cross-sectional study. Hepatogastroenterology. 2007;54:643-8
69. Thomsen RW, Riis A, Christensen S. et al. Diabetes and 30-day mortality from peptic ulcer bleeding and perforation: a Danish population-based cohort study. Diabetes Care. 2006;29:805-10
70. Gharzouli K, Amira S, Gharzouli A. et al. Gastroprotective effects of honey and glucose-fructose-sucrose-maltose mixture against ethanol-, indomethacin-, and acidified aspirin-induced lesions in the rat. Exp Toxicol Pathol. 2002;54:217-21
71. Osato MS, Reddy SG, Graham DY. Osmotic effect of honey on growth and viability of Helicobacter pylori. Dig Dis Sci. 1999;44:462-4
72. Gollu A, Kismet K, Kilicoglu B. et al. Effect of honey on intestinal morphology, intraabdominal adhesions and anastomotic healing. Phytother Res. 2008;22:1243-7
73. Gencay C, Kilicoglu SS, Kismet K. et al. Effect of honey on bacterial translocation and intestinal morphology in obstructive jaundice. World J Gastroenterol. 2008;14:3410-5
74. Ergul E, Ergul S. The effect of honey on the intestinal anastomotic wound healing in rats with obstructive jaundice. Bratisl Lek Listy. 2010;111:265-70
75. Quigley EM. Prebiotics and probiotics; modifying and mining the microbiota. Pharmacol Res. 2010;61:213-8
76. Bjorkholm B, Bok CM, Lundin A. et al. Intestinal microbiota regulate xenobiotic metabolism in the liver. PLoS One. 2009;4(e6958):1-8
77. Heijtz RD, Wang S, Anuar F. et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci U S A. 2011;108:3047-52
78. Cani PD, Delzenne NM, Amar J. et al. Role of gut microflora in the development of obesity and insulin resistance following high-fat diet feeding. Pathol Biol (Paris). 2008;56:305-9
79. Cani PD, Neyrinck AM, Fava F. et al. Selective increases of bifidobacteria in gut microflora improve high-fat-diet-induced diabetes in mice through a mechanism associated with endotoxaemia. Diabetologia. 2007;50:2374-83
80. Erejuwa OO, Sulaiman SA, Wahab MS. Oligosaccharides might contribute to the antidiabetic effect of honey: a review of the literature. Molecules. 2012;17:248-66
81. Sanz ML, Gonzalez M, Lorenzo C. et al. Carbohydrate composition and physicochemical properties of artisanal honeys from Madrid (Spain): occurrence of Echium sp. honey. J Sci Food Agric. 2004;84:1577-84
82. Munstedt K, Sheybani B, Hauenschild A. et al. Effects of basswood honey, honey-comparable glucose-fructose solution, and oral glucose tolerance test solution on serum insulin, glucose, and C-peptide concentrations in healthy subjects. J Med Food. 2008;11:424-8
83. Munstedt K, Bohme M, Hauenschild A. et al. Consumption of rapeseed honey leads to higher serum fructose levels compared with analogue glucose/fructose solutions. Eur J Clin Nutr. 2011;65:77-80
84. Gentiobiose. http://www.biosite.dk
85. Raffinose, palatinose and turanose. http://www.chemblink.com
86. Isomaltose, cellobiose, melezitose, trehalose and maltotriose. http://www.extrasynthese.com
87. Maltulose, panose and erlose. http://www.carbosynth.com
88. Shamala TR, Shri Jyothi Y, Saibaba P. Stimulatory effect of honey on multiplication of lactic acid bacteria under in vitro and in vivo conditions. Lett Appl Microbiol. 2000;30:453-5
89. Chick H, Shin HS, Ustunol Z. Growth and acid production by lactic acid bacteria and bifidobacteria grown in skim milk containing honey. J Food Sci. 2001;66:478-81
90. Kajiwara S, Gandhi H, Ustunol Z. Effect of honey on the growth of and acid production by human intestinal Bifidobacterium spp.: an in vitro comparison with commercial oligosaccharides and inulin. J Food Prot. 2002;65:214-8
91. Sanz ML, Polemis N, Morales V. et al. In vitro investigation into the potential prebiotic activity of honey oligosaccharides. J Agric Food Chem. 2005;53:2914-21
92. Shin HS, Ustunol Z. Carbohydrate composition of honey from different floral sources and their influence on growth of selected intestinal bacteria: An in vitro comparison. Food Res Int. 2005;38:721-8
93. Riazi A, Zia H. Growth and viability of yogurt starter organisms inhoney-sweetened skimmed mil. Afr J Biotech. 2008;7:2055-63
94. Ezz El-Arab AM, Girgis SM, Hegazy EM. et al. Effect of dietary honey on intestinal microflora and toxicity of mycotoxins in mice. BMC Complement Altern Med. 2006;6:1-13
95. Popa D, Ustunol Z. Influence of sucrose, high fructose corn syrup and honey from different floral sources on growth and acidproduction by lactic acid bacteria and bifidobacteria. Int J Dairy Technol. 2011;46:247-53
96. Kawai K, Okuda Y, Yamashita K. Changes in blood glucose and insulin after an oral palatinose administration in normal subjects. Endocrinol Jpn. 1985;32:933-6
97. Tonouchi H, Yamaji T, Uchida M. et al. Studies on absorption and metabolism of palatinose (isomaltulose) in rats. Br J Nutr. 2011;105:10-4
98. Gentilcore D, Vanis L, Teng JC. et al. The oligosaccharide alpha-cyclodextrin has modest effects to slow gastric emptying and modify the glycaemic response to sucrose in healthy older adults. Br J Nutr. 2011;106:583-7
99. Klip A, Vranic M. Muscle, liver, and pancreas: Three Musketeers fighting to control glycemia. Am J Physiol Endocrinol Metab. 2006;291:E1141-3
100. Mayes PA. Intermediary metabolism of fructose. Am J Clin Nutr. 1993;58:754S-65S
101. Van Schaftingen E, Davies DR. Fructose administration stimulates glucose phosphorylation in the livers of anesthetized rats. FASEB J. 1991;5:326-30
102. Youn JH, Kaslow HR, Bergman RN. Fructose effect to suppress hepatic glycogen degradation. J Biol Chem. 1987;262:11470-7
103. Ciudad CJ, Carabaza A, Guinovart JJ. Glycogen synthesis from glucose and fructose in hepatocytes from diabetic rats. Arch Biochem Biophys. 1988;267:437-47
104. Regan JJ Jr, Doorneweerd DD, Gilboe DP. et al. Influence of fructose on the glycogen synthase and phosphorylase systems in rat liver. Metabolism. 1980;29:965-9
105. Shiota M, Galassetti P, Igawa K. et al. Inclusion of low amounts of fructose with an intraportal glucose load increases net hepatic glucose uptake in the presence of relative insulin deficiency in dog. Am J Physiol Endocrinol Metab. 2005;288:E1160-7
106. Wei Y, Bizeau ME, Pagliassotti MJ. An acute increase in fructose concentration increases hepatic glucose-6-phosphatase mRNA via mechanisms that are independent of glycogen synthase kinase-3 in rats. J Nutr. 2004;134:545-51
107. Cherrington AD. Banting Lecture 1997. Control of glucose uptake and release by the liver in vivo. Diabetes. 1999;48:1198-214
108. Wright EM, Martin MG, Turk E. Intestinal absorption in health and disease--sugars. Best Pract Res Clin Gastroenterol. 2003;17:943-56
109. Leeds JS, Forman EM, Morley S. et al. Abnormal liver function tests in patients with type 1 diabetes mellitus: prevalence, clinical correlations and underlying pathologies. Diabet Med. 2009;26:1235-41
110. Al-Waili NS. Intravenous and intrapulmonary administration of honey solution to healthy sheep: effects on blood sugar, renal and liver function tests, bone marrow function, lipid profile, and carbon tetrachloride-induced liver injury. J Med Food. 2003;6:231-47
111. Dias AS, Porawski M, Alonso M. et al. Quercetin decreases oxidative stress, NF-kappaB activation, and iNOS overexpression in liver of streptozotocin-induced diabetic rats. J Nutr. 2005;135:2299-304
112. Kilicoglu B, Gencay C, Kismet K. et al. The ultrastructural research of liver in experimental obstructive jaundice and effect of honey. Am J Surg. 2008;195:249-56
113. Malik VS, Schulze MB, Hu FB. Intake of sugar-sweetened beverages and weight gain: a systematic review. Am J Clin Nutr. 2006;84:274-88
114. Huynh M, Luiken JJ, Coumans W. et al. Dietary fructose during the suckling period increases body weight and fatty acid uptake into skeletal muscle in adult rats. Obesity (Silver Spring). 2008;16:1755-62
115. Bocarsly ME, Powell ES, Avena NM. et al. High-fructose corn syrup causes characteristics of obesity in rats: increased body weight, body fat and triglyceride levels. Pharmacol Biochem Behav. 2010;97:101-6
116. Stanhope KL, Schwarz JM, Keim NL. et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. J Clin Invest. 2009;119:1322-34
117. Tappy L, Le KA. Metabolic effects of fructose and the worldwide increase in obesity. Physiol Rev. 2010;90:23-46
118. Razquin C, Martinez JA, Martinez-Gonzalez MA. et al. 3 years follow-up of a Mediterranean diet rich in virgin olive oil is associated with high plasma antioxidant capacity and reduced body weight gain. Eur J Clin Nutr. 2009;63:1387-93
119. Nemoseck TM, Carmody EG, Furchner-Evanson A. et al. Honey promotes lower weight gain, adiposity, and triglycerides than sucrose in rats. Nutr Res. 2011;31:55-60
120. Bahrami M, Ataie-Jafari A, Hosseini S, Foruzanfar MH, Rahmani M, Pajouhi M. Effects of natural honey consumption in diabetic patients: an 8-week randomized clinical trial. Int J Food Sci Nutr. 2009;60:618-26
121. Yaghoobi N, Al-Waili N, Ghayour-Mobarhan M. et al. Natural honey and cardiovascular risk factors; effects on blood glucose, cholesterol, triacylglycerole, CRP, and body weight compared with sucrose. ScientificWorldJournal. 2008;8:463-9
122. Dolan LC, Potter SM, Burdock GA. Evidence-based review on the effect of normal dietary consumption of fructose on blood lipids and body weight of overweight and obese individuals. Crit Rev Food Sci Nutr. 2010;50:889-918
123. Perez-Pozo SE, Schold J, Nakagawa T. et al. Excessive fructose intake induces the features of metabolic syndrome in healthy adult men: role of uric acid in the hypertensive response. Int J Obes (Lond). 2009;34:454-61
124. Rohlfing CL, Wiedmeyer HM, Little RR. et al. Defining the relationship between plasma glucose and HbA(1c): analysis of glucose profiles and HbA(1c) in the Diabetes Control and Complications Trial. Diabetes Care. 2002;25:275-8
125. Akhtar MS, Khan MS. Glycaemic responses to three different honeys given to normal and alloxan-diabetic rabbits. J Pak Med Assoc. 1989;39:107-13
126. Fasanmade AA, Alabi OT. Differential effect of honey on selected variables in alloxan-induced and fructose-induced diabetic rats. Afr J Biomed Res. 2008;11:191-6
127. Chepulis L, Starkey N. The long-term effects of feeding honey compared with sucrose and a sugar-free diet on weight gain, lipid profiles, and DEXA measurements in rats. J Food Sci. 2008;73:H1-7
128. Shambaugh P, Worthington V, Herbert JH. Differential effects of honey, sucrose, and fructose on blood sugar levels. J Manipulative Physiol Ther. 1990;13:322-5
129. Ahmad A, Azim MK, Mesaik MA. et al. Natural honey modulates physiological glycemic response compared to simulated honey and D-glucose. J Food Sci. 2008;73:H165-7
130. Abdulrhman M, El-Hefnawy M, Hussein R. et al. The glycemic and peak incremental indices of honey, sucrose and glucose in patients with type 1 diabetes mellitus: effects on C-peptide level-a pilot study. Acta Diabetol. 2011;48:89-94
131. Samanta A, Burden AC, Jones GR. Plasma glucose responses to glucose, sucrose, and honey in patients with diabetes mellitus: an analysis of glycaemic and peak incremental indices. Diabet Med. 1985;2:371-3
132. Abdulrhman M, El-Hefnawy M, Ali R. et al. Honey and type 1 diabetes mellitus. In: (ed.) Liu CP. Type 1 diabetes - complications, pathogenesis, and alternative treatments. Croatia: InTech. 2011:228-33
133. Agrawal OP, Pachauri A, Yadav H. et al. Subjects with impaired glucose tolerance exhibit a high degree of tolerance to honey. J Med Food. 2007;10:473-8
134. Erejuwa OO. The use of honey in diabetes mellitus: is it beneficial or detrimental? Int J Endocrinol Metab. 2012;10:444-45
135. Bornet F, Haardt MJ, Costagliola D. et al. Sucrose or honey at breakfast have no additional acute hyperglycaemic effect over an isoglucidic amount of bread in type 2 diabetic patients. Diabetologia. 1985;28:213-7
136. Katsilambros NL, Philippides P, Touliatou A. et al. Metabolic effects of honey (alone or combined with other foods) in type II diabetics. Acta Diabetol Lat. 1988;25:197-203
137. Chepulis LM. The effect of honey compared to sucrose, mixed sugars, and a sugar-free diet on weight gain in young rats. J Food Sci. 2007;72:S224-9
138. Aronoff SL, Berkowitz K, Shreiner B. et al. Glucose metabolism and regulation: beyond insulin and glucagon. Diabetes Spectrum. 2004;17:183-90
139. Al-Waili NS. Natural honey lowers plasma glucose, C-reactive protein, homocysteine, and blood lipids in healthy, diabetic, and hyperlipidemic subjects: comparison with dextrose and sucrose. J Med Food. 2004;7:100-7
140. Busserolles J, Gueux E, Rock E. et al. Substituting honey for refined carbohydrates protects rats from hypertriglyceridemic and prooxidative effects of fructose. J Nutr. 2002;132:3379-82
141. Munstedt K, Hoffmann S, Hauenschild A. et al. Effect of honey on serum cholesterol and lipid values. J Med Food. 2009;12:624-8
142. Erbey JR, Silberman C, Lydick E. Prevalence of abnormal serum alanine aminotransferase levels in obese patients and patients with type 2 diabetes. Am J Med. 2000;109:588-90
143. Al-Waili NS. Effects of daily consumption of honey solution on hematological indices and blood levels of minerals and enzymes in normal individuals. J Med Food. 2003;6:135-40
144. Al-Waili NS. Effects of honey on the urinary total nitrite and prostaglandins concentration. Int Urol Nephrol. 2005;37:107-11
145. Ahmed A, Khan RA, Azim MK. et al. Effect of natural honey on human platelets and blood coagulation proteins. Pak J Pharm Sci. 2011;24:389-97
146. Batterham RL, Heffron H, Kapoor S. et al. Critical role for peptide YY in protein-mediated satiation and body-weight regulation. Cell Metab. 2006;4:223-33
147. Klok MD, Jakobsdottir S, Drent ML. The role of leptin and ghrelin in the regulation of food intake and body weight in humans: a review. Obes Rev. 2007;8:21-34
148. Larson-Meyer DE, Willis KS, Willis LM. et al. Effect of honey versus sucrose on appetite, appetite-regulating hormones, and postmeal thermogenesis. J Am Coll Nutr. 2010;29:482-93
149. Teff KL, Elliott SS, Tschop M. et al. Dietary fructose reduces circulating insulin and leptin, attenuates postprandial suppression of ghrelin, and increases triglycerides in women. J Clin Endocrinol Metab. 2004;89:2963-72
150. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Differential responses to blood pressure and oxidative stress in streptozotocin-induced diabetic wistar-kyoto rats and spontaneously hypertensive rats: effects of antioxidant (honey) treatment. Int J Mol Sci. 2011;12:1888-907
151. Parnell JA, Reimer RA. Weight loss during oligofructose supplementation is associated with decreased ghrelin and increased peptide YY in overweight and obese adults. Am J Clin Nutr. 2009;89:1751-9
152. Gheldof N, Wang XH, Engeseth NJ. Buckwheat honey increases serum antioxidant capacity in humans. J Agric Food Chem. 2003;51:1500-5
153. Erejuwa OO, Sulaiman SA, Wahab MS. Honey: a novel antioxidant. Molecules. 2012;17:4400-23
154. Deibert P, Konig D, Kloock B. et al. Glycaemic and insulinaemic properties of some German honey varieties. Eur J Clin Nutr. 2010;64:762-4
155. Robert SD, Ismail AA. Two varieties of honey that are available in Malaysia gave intermediate glycemic index values when tested among healthy individuals. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2009;153:145-7
156. Ischayek JI, Kern M. US honeys varying in glucose and fructose content elicit similar glycemic indexes. J Am Diet Assoc. 2006;106:1260-2
157. Koca I, Koca AF. Poisoning by mad honey: a brief review. Food Chem Toxicol. 2007;45:1315-8
158. Yilmaz O, Eser M, Sahiner A. et al. Hypotension, bradycardia and syncope caused by honey poisoning. Resuscitation. 2006;68:405-8
159. Demir H, Denizbasi A, Onur O. Mad honey intoxication: A case series of 21 patients. ISRN Toxicology. 2011;2011:1-3
160. Aliyev F, Turkoglu C, Celiker C. et al. Chronic mad honey intoxication syndrome: a new form of an old disease? Europace. 2009;11:954-6
161. Fenicia L, Anniballi F. Infant botulism. Ann Ist Super Sanita. 2009;45:134-46
162. Arriagada SD, Wilhelm BJ, Donoso FA. Infant botulism: case report and review. Rev Chilena Infectol. 2009;26:162-7
163. Arnon SS, Midura TF, Damus K. et al. Honey and other environmental risk factors for infant botulism. J Pediatr. 1979;94:331-6
164. Thompson JA, Glasgow LA, Warpinski JR. et al. Infant botulism: clinical spectrum and epidemiology. Pediatrics. 1980;66:936-42
165. Tanzi MG, Gabay MP. Association between honey consumption and infant botulism. Pharmacotherapy. 2002;22:1479-83
166. Fenicia L, Ferrini AM, Aureli P. et al. A case of infant botulism associated with honey feeding in Italy. Eur J Epidemiol. 1993;9:671-3
167. van der Vorsta MMJ, Jamal W, Rotimi VO. et al. Infant botulism due to consumption of contaminated commercially prepared honey. Med Princ Pract. 2006;15:456-8
168. Watford M. Small amounts of dietary fructose dramatically increase hepatic glucose uptake through a novel mechanism of glucokinase activation. Nutr Rev. 2002;60:253-7
169. Vozzo R, Baker B, Wittert GA. et al. Glycemic, hormone, and appetite responses to monosaccharide ingestion in patients with type 2 diabetes. Metabolism. 2002;51:949-57
170. Oizumi T, Daimon M, Jimbu Y. et al. A palatinose-based balanced formula improves glucose tolerance, serum free fatty acid levels and body fat composition. Tohoku J Exp Med. 2007;212:91-9
171. Häberer D, Thibault L, Langhans W. et al. Beneficial effects on glucose metabolism of chronic feeding of isomaltulose versus sucrose in rats. Ann Nutr Metab. 2009;54:75-82
172. Sato K, Arai H, Mizuno A. et al. Dietary palatinose and oleic acid ameliorate disorders of glucose and lipid metabolism in Zucker fatty rats. J Nutr. 2007;137:1908-15
173. Cani PD, Knauf C, Iglesias MA. et al. Improvement of glucose tolerance and hepatic insulin sensitivity by oligofructose requires a functional glucagon-like peptide 1 receptor. Diabetes. 2006;55:1484-90
174. Anderson RA, Cheng N, Bryden NA. et al. Elevated intakes of supplemental chromium improve glucose and insulin variables in individuals with type 2 diabetes. Diabetes. 1997;46:1786-91
175. Kar A, Choudhary BK, Bandyopadhyay NG. Preliminary studies on the inorganic constituents of some indigenous hypoglycaemic herbs on oral glucose tolerance test. J Ethnopharmacol. 1999;64:179-84
176. Sitasawad S, Deshpande M, Katdare M. et al. Beneficial effect of supplementation with copper sulfate on STZ-diabetic mice (IDDM). Diabetes Res Clin Pract. 2001;52:77-84
177. Song MK, Hwang IK, Rosenthal MJ. et al. Antidiabetic actions of arachidonic acid and zinc in genetically diabetic Goto-Kakizaki rats. Metabolism. 2003;52:7-12
178. Oh HM, Yoon JS. Glycemic control of type 2 diabetic patients after short-term zinc supplementation. Nutr Res Pract. 2008;2:283-8
179. Li F, Zhang G, Liang J. et al. Sleeve gastrectomy provides a better control of diabetes by decreasing ghrelin in the diabetic Goto-Kakizaki rats. J Gastrointest Surg. 2009;13:2302-8
180. Folli F, Corradi D, Fanti P. et al. The role of oxidative stress in the pathogenesis of type 2 diabetes mellitus micro- and macrovascular complications:avenues for a mechanistic-based therapeutic approach. Curr Diabetes Rev. 2011;7:313-24
181. Chang YC, Chuang LM. The role of oxidative stress in the pathogenesis of type 2 diabetes: from molecular mechanism to clinical implication. Am J Transl Res. 2010;2:316-31
182. Kim JS, Saengsirisuwan V, Sloniger JA. et al. Oxidant stress and skeletal muscle glucose transport: roles of insulin signaling and p38 MAPK. Free Radic Biol Med. 2006;41:818-24
183. Talior I, Yarkoni M, Bashan N. et al. Increased glucose uptake promotes oxidative stress and PKC-delta activation in adipocytes of obese, insulin-resistant mice. Am J Physiol Endocrinol Metab. 2003;285:E295-302
184. Rudich A, Tirosh A, Potashnik R. et al. Prolonged oxidative stress impairs insulin-induced GLUT4 translocation in 3T3-L1 adipocytes. Diabetes. 1998;47:1562-9
185. Dokken BB, Saengsirisuwan V, Kim JS. et al. Oxidative stress-induced insulin resistance in rat skeletal muscle: role of glycogen synthase kinase-3. Am J Physiol Endocrinol Metab. 2008;294:E615-21
186. Henriksen EJ. Dysregulation of glycogen synthase kinase-3 in skeletal muscle and the etiology of insulin resistance and type 2 diabetes. Curr Diabetes Rev. 2010;6:285-93
187. Tiedge M, Lortz S, Drinkgern J. et al. Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes. 1997;46:1733-42
188. Drews G, Krippeit-Drews P, Dufer M. Oxidative stress and beta-cell dysfunction. Pflugers Arch. 2010;460:703-18
189. Miwa I, Ichimura N, Sugiura M. et al. Inhibition of glucose-induced insulin secretion by 4-hydroxy-2-nonenal and other lipid peroxidation products. Endocrinology. 2000;141:2767-72
190. Tanaka Y, Gleason CE, Tran PO. et al. Prevention of glucose toxicity in HIT-T15 cells and Zucker diabetic fatty rats by antioxidants. Proc Natl Acad Sci U S A. 1999;96:10857-62
191. Marchetti P, Dotta F, Lauro D. et al. An overview of pancreatic beta-cell defects in human type 2 diabetes: implications for treatment. Regul Pept. 2008;146:4-11
192. Janjic D, Andereggen E, Deng S. et al. Improved insulin secretion of cryopreserved human islets by antioxidant treatment. Pancreas. 1996;13:166-72
193. Penckofer S, Schwertz D, Florczak K. Oxidative stress and cardiovascular disease in type 2 diabetes: the role of antioxidants and pro-oxidants. J Cardiovasc Nurs. 2002;16:68-85
194. Mohamed M, Sirajudeen K, Swamy M. et al. Studies on the antioxidant properties of Tualang honey of Malaysia. Afr J Tradit Complement Altern Med. 2010;7:59-63
195. Khalil MI, Alam N, Moniruzzaman M. et al. Phenolic acid composition and antioxidant properties of Malaysian honeys. J Food Sci. 2011;76:C921-8
196. Kishore RK, Halim AS, Syazana MS. et al. Tualang honey has higher phenolic content and greater radical scavenging activity compared with other honey sources. Nutr Res. 2011;31:322-5
197. Gheldof N, Engeseth NJ. Antioxidant capacity of honeys from various floral sources based on the determination of oxygen radical absorbance capacity and inhibition of in vitro lipoprotein oxidation in human serum samples. J Agric Food Chem. 2002;50:3050-5
198. Fava D, Cassone-Faldetta M, Laurenti O. et al. Gliclazide improves anti-oxidant status and nitric oxide-mediated vasodilation in type 2 diabetes. Diabet Med. 2002;19:752-7
199. Rahimi R, Nikfar S, Larijani B. et al. A review on the role of antioxidants in the management of diabetes and its complications. Biomed Pharmacother. 2005;59:365-73
200. Lai MH. Antioxidant effects and insulin resistance improvement of chromium combined with vitamin C and E supplementation for type 2 diabetes mellitus. J Clin Biochem Nutr. 2008;43:191-8
201. Ihara Y, Yamada Y, Toyokuni S. et al. Antioxidant alpha-tocopherol ameliorates glycemic control of GK rats, a model of type 2 diabetes. FEBS Lett. 2000;473:24-6
202. Opara EC. Role of oxidative stress in the etiology of type 2 diabetes and the effect of antioxidant supplementation on glycemic control. J Investig Med. 2004;52:19-23
203. Erejuwa OO. Management of diabetes mellitus: could simultaneous targeting of hyperglycemia and oxidative stress be a better panacea? IJMS. 2012;13:2965-72
204. Ceriello A, Giugliano D, Quatraro A. et al. Vitamin E reduction of protein glycosylation in diabetes. New prospect for prevention of diabetic complications? Diabetes Care. 1991;14:68-72
205. Vinson JA, Howard TB. Inhibition of protein glycation and advanced glycation end products by ascorbic acid and other vitamins and nutrients. J Nutr Biochem. 1996;7:659-63
206. Lautt WW, Ming Z, Legare DJ. Attenuation of age- and sucrose-induced insulin resistance and syndrome X by a synergistic antioxidant cocktail: the AMIS syndrome and HISS hypothesis. Can J Physiol Pharmacol. 2010;88:313-23
207. Tong X, Dong J, Wu Z. et al. [Whey protein improves insulin resistance via the increase of antioxidant capacity in model rats]. Wei Sheng Yan Jiu. 2011;40:617-9
208. Wright D, Sutherland L. Antioxidant supplemention in the treatment of skeletal muscle insulin resistance: potential mechanisms and clinical relevance. Appl Physiol Nutr Metab. 2008;33:21-31
209. Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615-25
210. Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res. 2010;107:1058-70
211. Nishikawa T, Edelstein D, Brownlee M. The missing link: a single unifying mechanism for diabetic complications. Kidney Int Suppl. 2000;77:S26-30
212. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813-20
213. Brand MD. The sites and topology of mitochondrial superoxide production. Exp Gerontol. 2010;45:466-72
214. Genuth S, Sun W, Cleary P. et al. Glycation and carboxymethyllysine levels in skin collagen predict the risk of future 10-year progression of diabetic retinopathy and nephropathy in the diabetes control and complications trial and epidemiology of diabetes interventions and complications participants with type 1 diabetes. Diabetes. 2005;54:3103-11
215. Kowluru RA. Effect of reinstitution of good glycemic control on retinal oxidative stress and nitrative stress in diabetic rats. Diabetes. 2003;52:818-23
216. Kowluru RA, Kanwar M, Kennedy A. Metabolic memory phenomenon and accumulation of peroxynitrite in retinal capillaries. Exp Diabetes Res. 2007;2007:1-7
217. Seghrouchni I, Drai J, Bannier E. et al. Oxidative stress parameters in type I, type II and insulin-treated type 2 diabetes mellitus; insulin treatment efficiency. Clin Chim Acta. 2002;321:89-96
218. Ceriello A. Hypothesis: the "metabolic memory", the new challenge of diabetes. Diabetes Res Clin Pract. 2009;86(Suppl 1):S2-6
219. Ihnat MA, Thorpe JE, Kamat CD. et al. Reactive oxygen species mediate a cellular 'memory' of high glucose stress signalling. Diabetologia. 2007;50:1523-31
220. Ceriello A, Ihnat MA, Thorpe JE. Clinical review 2: The "metabolic memory": is more than just tight glucose control necessary to prevent diabetic complications? J Clin Endocrinol Metab. 2009;94:410-5
221. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Impaired Nrf2-ARE pathway contributes to increased oxidative damage in kidney of spontaneously hypertensive rats: Effect of antioxidant (honey). Int J Cardiol. 2011;152:S45
222. Erejuwa OO, Sulaiman SA, Wahab MS. et al. Influence of rat strains and/or severity of hyperglycemia on systolic blood pressure and antioxidant enzymes in kidney of rats with hypertension and/or diabetes: role of honey. Int J Cardiol. 2011;152:S29
223. DCCT. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. Diabetes Control and Complications Trial Research Group. N Engl J Med. 1993;329:977-86
224. UKPDS. UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837-53
225. Cook MN, Girman CJ, Stein PP. et al. Glycemic control continues to deteriorate after sulfonylureas are added to metformin among patients with type 2 diabetes. Diabetes Care. 2005;28:995-1000
226. Turner RC, Cull CA, Frighi V. et al. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA. 1999;281:2005-12
227. Ismail-Beigi F, Craven T, Banerji MA. et al. Effect of intensive treatment of hyperglycaemia on microvascular outcomes in type 2 diabetes: an analysis of the ACCORD randomised trial. Lancet. 2010;376:419-30
228. Simpson SH, Corabian P, Jacobs P. et al. The cost of major comorbidity in people with diabetes mellitus. CMAJ. 2003;168:1661-7
229. Patel H, Srishanmuganathan J, Car J. et al. Trends in the prescription and cost of diabetic medications and monitoring equipment in England 1991-2004. J Public Health (Oxf). 2007;29:48-52
230. Justin-Temu M, Nondo RS, Wiedenmayer K. et al. Anti-diabetic drugs in the private and public sector in Dar es Salaam, Tanzania. East Afr Med J. 2009;86:110-4
Author contact
Corresponding author: E-Mail: erejuwacom; Tel.: +609-7666-877; Fax: +609-7653-370.

 Global reach, higher impact
Global reach, higher impact