Impact Factor ISSN: 1449-2288
Int J Biol Sci 2015; 11(9):1049-1055. doi:10.7150/ijbs.12023 This issue Cite
Research Paper
Higher Frequency of Circulating PD-1high CXCR5+CD4+ Tfh Cells in Patients with Chronic Schistosomiasis
1. National Institute of Parasitic Diseases, Chinese Center for Disease Control and Prevention; Key Laboratory of Parasite and Vector Biology, MOH, China; WHO Collaborating Center for Malaria, Schistosomiasis and Filariasis, Shanghai 200025, PR China
2. Department of Pathogenic Biology, Binzhou Medical University, Yantai, 264003, Shandong, China
Received 2015-3-2; Accepted 2015-5-23; Published 2015-7-15
Abstract
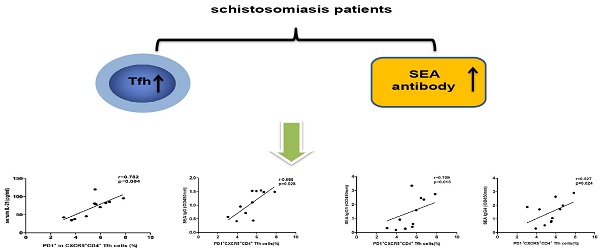
The current knowledge of immunological responses to schistosomiasis is insufficient for the development of vaccine and therapies. The role of T follicular helper (Tfh) cells in schistosome infections is not fully defined. The frequency of circulating Tfh cells and serum cytokine levels were analyzed in 11 patients with chronic schistosomiasis and 10 healthy controls (HC), who reside in an endemic area for Schistosomiasis japonicum. Significantly higher frequencies of circulating CXCR5+ CD4+ Tfh cells and higher expression levels of ICOS and PD-1 in CXCR5+ CD4+ Tfh cells were observed in patients with chronic schistosomiasis compared to HC. The levels of IL-21 in serum and the expression of IL-21 mRNA were higher in chronic schistosomiasis patients than in HC. Moreover, the frequency of circulating PD-1high CXCR5+ CD4+ Tfh cells positively correlated with the levels of IL-21 in serum from patients with chronic schistosomiasis. A positive correlation was also found between the frequency of PD-1high CXCR5+ CD4+ Tfh cells and the levels of soluble egg antigen (SEA)-specific antibodies in serum samples from the patient group. Our study is the first regarding Tfh cells in chronic human schistosomiasis and the finding indicate that PD-1high CXCR5+ CD4+Tfh cells might play an important role in the production of specific antibodies in schistosomiasis. This study contributes to the understanding of immune response to schistosomiasis and may provide helpful support in vaccine development.
Keywords: T follicular helper cells, schistosomiasis, specific antibody, blood.
Introduction
Schistosomiasis is a major neglected helminthic disease which remains a serious public health problem in many developing countries in tropical and subtropical regions. There are approximately 200 million people infected and more than 700 million people at risk of becoming infected [1,2]. Schistosomiasis japonica, caused by infection with Schistosoma japonicum, leads to severe pathology such as granuloma formation in the liver, periportal fibrosis and portal hypertension in chronic infections [3], but there is no definitive host vaccine for S. japonica. Since the mechanisms underlying the immunologic response to schistosomiasis are not well described, better understanding would be helpful in vaccine development and therapies.
CD4+ T cells play an important role in both host immune responses against Schistosoma and immunopathology in schistosomiasis [4]. Naive CD4+ T helper (Th) cells recognize diverse antigens of Schistosoma to differentiate into distinct effector subsets, including Th1, Th2, Th17, T regulatory (Treg) and Tfh cells. Hepatic granuloma formation was upregulated by Th2 and Th17 cells secreting IL-4 and IL-17 respectively [5,6], and downregulated by Th1 and Treg cells[7,8]. Tfh has recently been recognized as a separate subset of CD4+ T cells that is mainly responsible for assisting B cells to induce antibody response[9]. Tfh cells are characterized by high expression levels of the CD28 family members, PD1 and ICOS, as well as the chemokine receptor CXCR5. B-cell lymphoma 6 (BCl-6) and c-Maf are two important transcriptional factors that control the transcriptional signature of Tfh cells[10-12]. In addition, cytokine interleukin 21 (IL-21) from Tfh cells enhance humoral immune responses[13,14]. Recently, a study regarding Tfh cells has been reported that showed Tfh cells can promote liver pathology in mice during S. japonicum infection[15], but little is known about the role of circulating Tfh cells in human schistosomiasis infection.
Most Tfh cells are located in the GC zone of secondary lymph nodules and therefore hard to access[16]. However, several studies have also detected Tfh cells in peripheral blood[17] and circulating Tfh cells have some features of GC Tfh cells[18]. Circulating Tfh cells may act as a counterpart, enabling measurement of GC Tfh cells and could be used to study Tfh cells. The role of circulating Tfh cells has been explored in patients with autoimmune diseases such as primary Sjogren's syndrome (pSS) and autoimmune thyroid disease (AITD), and some infectious diseases such as HIV and chronic hepatitis B (CHB)[19-21]. In this study, we examined the frequencies of Tfh cells and associated molecules in chronic schistosomiasis patients and healthy controls (HC) to investigate the potential roles of circulating Tfh cells. This study may provide novel insights regarding the role of circulating Tfh cells in patients with chronic schistosomiasis.
Materials and Methods
Ethics statement
Ethical approval for the collection and examination of human feces and blood samples was obtained from the Ethics Committee of the National Institute of Parasitic Diseases, Chinese Center for Disease Control and Prevention, China (reference no. 2012-12). The objectives, procedures and potential risks were verbally explained to all participants. Written informed consent was obtained from all the study participants.
Collection of study subjects
Our study was carried out in Dongting Lake Basin, Hunan Province, China, where schistosomiasis is endemic. We detected Schistosoma eggs in feces, and soluble egg antigen (SEA) IgG and indirect hemagglutination antibody (IHA) in sera, in study subjects. Patients with chronic schistosomiasis are asymptomatic carriers with no fever or fatigue, but were designated as suffering chronic schistosomiasis based on positive results from fecal egg, SEA IgG and IHA analyses, past medical history of infection, and frequent contact with cercariae in water. Healthy controls were selected by negative results from fecal egg, SEA IgG and IHA analyses and had no history of schistosomiasis.
Cell isolation
Sera were collected and stored at -80°C for detecting cytokine levels and specific antibodies against S. japonicum. Peripheral blood mononuclear cells (PBMCs) were isolated by standard Ficoll-density gradient centrifugation and washed in FACS buffer (5% FBS, 0.1% NaN3 in PBS) before surface staining. CD4+ T cells were purified from PBMCs using human CD4 MicroBeads (Miltenyi Biotec, USA) according to the manufacturer's instructions.
Flow cytometric analysis
PBMCs were immunostained with fluorescein isothiocyanate (FITC)-ICOS, phycoerythrin (PE)-CD4 (eBioscience, USA), and APC-CXCR5, PE-Cy7-PD-1 (BioLegend, USA) monoclonal antibodies according to the manufacturers' protocols. The stained cells were then analyzed using a CyAn ADP analyzer (Beckman Coulter) and data were analyzed with FlowJo software.
Enzyme-linked immunosorbent assay (ELISA) analysis of cytokine and S. Japonicum-specific antibodies
The concentration of serum IL-21 was determined using a human IL-21 ELISA Ready-SET-Go kit (eBioscience, USA) according to the manufacturer's protocol. All determinations were performed in triplicate. SEA were coated on plates, then the sera from schistosomiasis patients and healthy controls were tested for the presence of anti-SEA IgG, IgG1 and IgG4 antibodies by ELISA. All samples were analyzed in triplicate using the average of the optical density (OD) 450 nm values to calculate concentrations.
Real-time polymerase chain reaction
To detect the mRNA expression of Bcl6, c-Maf and IL-21, total RNA was extracted from CD4+ T cells using TRIzol reagent and cDNA was synthesized according to the manufacturer's instructions (TaKaRa, Japan). Each gene was amplified in triplicate using SYBR Supermix (TaKaRa) and a CFX960 analysis system (Applied Biosystems, USA). The PCR amplification program was as follows: an initial denaturation at 95°C for 5 min, followed by 40 amplification cycles at 95°C for 10 s, 60°C for 30 s. The fluorescence values were collected at 60°C. Concentration differences of cDNA were normalized to β-actin. Primer sequences were: IL-21 sense, 5'-CACAGACTAACATGCCCTTCAT-3' and antisense, 5'-GAATCTTCACTTC CGTGTGTTCT-3'; Bcl-6 sense, 5'-AAGGCCAGTGAAGCAGAGA-3', and antisense, 5'-CCGATAGGCCATGATGTCT-3'; c-Maf sense, 5'- CAAGCTAGAAGCGCCCC-3', and antisense, 5'- AGTTCTGATGCCATTCTCCTG-3'; β-actin sense 5'-AGCGAGCATCCCCCAAAGTT-3' , and antisense 5'-GGGCACGAAGGCTCATCATT -3'.
Statistical analysis
All data are presented as mean ± standard deviation. The differences between two groups were analyzed by independent t-test or Mann-Whitney U-test using SPSS19.0. The correlation between groups was tested using Spearman's rank correlation. A two-sided p value of < 0.05 was considered statistically significant.
Results
Characteristics of study subjects
Eleven chronic schistosomiasis patients (aged 47 ± 5.22) and 10 healthy controls (aged 42 ± 10.05) were enrolled in the study. They were all male and had no significant difference in age (p = 0.302). Peripheral blood samples were obtained from all patients and healthy controls.
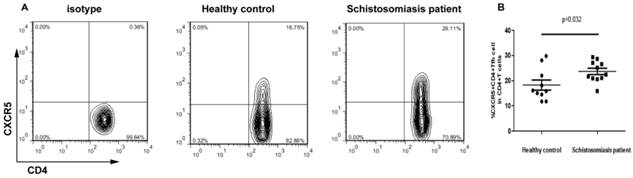
Higher frequency of CXCR5+ CD4+ Tfh cells in the peripheral blood of chronic schistosomiasis patients
To determine circulating Tfh cell immune function, 11 schistosomiasis patients and 10 healthy controls were recruited. The frequency of circulating CXCR5+CD4+Tfh cells within the CD4+ T cell population was monitored by flow cytometry (Fig. 1A). As shown in Fig. 1B, the frequency of CXCR5+CD4+Tfh cells was significantly higher in chronic schistosomiasis patients than in the HC group (p < 0.05).
Flow cytometry analysis of CXCR5+ CD4+ Tfh cells in schistosomiasis patients and healthy controls. Isotype controls were used to determine the positive cells. All of the values were gated on CD4+ cells. (A) Values in the upper right quadrant correspond to the percentage of CXCR5+ CD4+ Tfh cells in the total CD4+ T cells. (B) The correlation between the percentages of CXCR5+ CD4+ Tfh cells between two groups (HC and schistosomiasis patients) (p < 0.05). Data shown are the mean percentage of Tfh cells in total CD4+ T cells in individual subjects. The horizontal lines represent the median values.
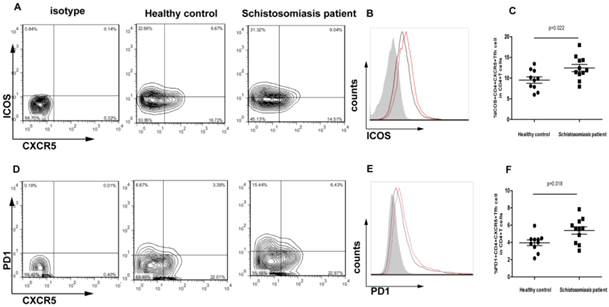
Increased expression of ICOS and PD-1 in chronic schistosomiasis patients
A significantly higher frequency of CXCR5+CD4+ Tfh cells was observed in the chronic schistosomiasis patients than in the HC group. Thus, it was important to determine whether the expression of markers typically associated with Tfh cells was altered. The function of Tfh cells is closely related to the expression of ICOS and PD-1. The phenotype of Tfh cells was assessed in this study by flow cytometry (Fig. 2A, 2D). Notably, the expression of ICOS was increased in CXCR5+CD4+ Tfh cells in chronic schistosomiasis patients compared to healthy controls (p < 0.05) (Fig. 2B-C). Likewise, high expression of PD-1 was observed in chronic schistosomiasis patients compared to healthy subjects (p < 0.05) (Fig. 2E-F).
The phenotype of CXCR5+CD4+ Tfh cells in chronic schistosomiasis patients and healthy controls. (A) ICOS+ CXCR5+CD4+ Tfh cells in total CD4+ T cells from chronic schistosomiasis patients and healthy controls. (B) The expression of ICOS in isotype (gray), chronic schistosomiasis patients (red) and healthy controls (black). Samples were gated for CXCR5+CD4+ Tfh cells. (C) Comparison of the percentages of ICOS+ CXCR5+CD4+ Tfh cells in chronic schistosomiasis patients and healthy controls (p < 0.05). (D) PD-1+ CXCR5+CD4+ Tfh cells in total CD4+ T cells of chronic schistosomiasis patients and healthy controls. (E) The expression of PD-1 in isotype (gray), chronic schistosomiasis patients (red) and healthy controls (black). Samples were gated for CXCR5+CD4+ Tfh cells. (F) Comparison of the percentages of PD-1+ CXCR5+CD4+ Tfh cells in chronic schistosomiasis patients and healthy controls (p < 0.05).
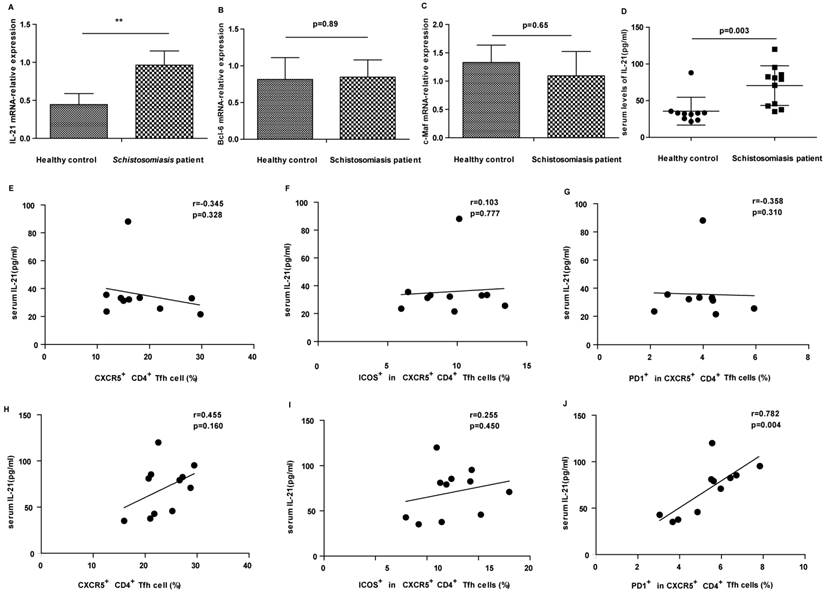
Levels of IL-21 in sera correlated with PD-1high CXCR5+CD4+ Tfh cells
IL-21 is a typical cytokine produced by Tfh cells and can enhance B cells to produce antibodies. As expected, the expression of IL-21 mRNA in CD4+ T cells of patients was increased compared with healthy controls (Fig. 3A). Moreover, the levels of IL-21 in sera were significantly higher in patients than in HC (Fig. 3D). The levels of serum IL-21 positively correlated with PD-1high CXCR5+CD4+ Tfh cells in schistosomiasis patients (Fig. 3J). However, there was no significant correlation between IL-21 levels and CXCR5+CD4+ Tfh cells or ICOShigh CXCR5+CD4+ Tfh cells in patients with schistosomiasis (Fig.3H-I). Moreover, there were no relationships between IL-21 levels and CXCR5+CD4+ Tfh cells, ICOShigh CXCR5+CD4+ Tfh cells, or PD-1high CXCR5+CD4+ Tfh cells in HC (Fig. 3E-G). A previous study showed that Bcl6 and c-Maf cooperate to ensure the differentiation of human Tfh [12] . We therefore also assessed the expression of transcription factor Bcl-6 and c-Maf in schistosomiasis patients and healthy controls. However, the results of the qPCR assays showed that Bcl-6 and c-Maf mRNA expression levles in CD4 + T cells of patients were similar to those in controls (Fig. 3B-C).
Correlation of IL-21 levels and circulating Tfh cells in schistosomiasis patients and HC. (A) Detection of IL-21 mRNA expression. (* p < 0.05). (B) Detection of Bcl-6 mRNA expression. (C) Detection of c-Maf mRNA expression. (D) Levels of IL-21 in sera from schistosomiasis patients and HC. Data shown are the mean ± SD, the horizontal lines show the median. (E-G) Relationship of IL-21 levels in sera and the percentage of CXCR5+CD4+ Tfh cells, ICOShigh CXCR5+CD4+ Tfh cells and PD-1high CXCR5+CD4+ Tfh cells in HC; (H-J) Relationship of IL-21 levels in sera and the percentage of CXCR5+CD4+ Tfh cells, ICOShigh CXCR5+CD4+ Tfh cells and PD-1high CXCR5+CD4+ Tfh cells in schistosomiasis patients.
Levels of serum SEA specific antibodies
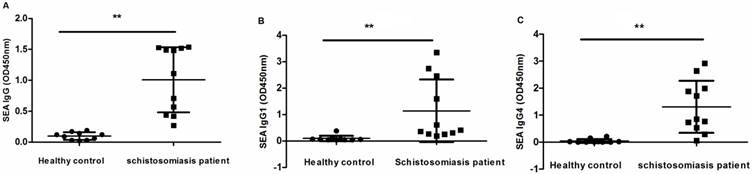
Because of the high frequency of Tfh cells and the upregulation of ICOS and PD1 in patients with chronic schistosomiasis, we evaluated whether the function of Tfh cells was changed in the patients. Tfh cells are mainly responsible for assisting B cells to induce antibodies. In chronic schistosomiasis, SEA-specific antibodies may be beneficial for the hosts and also have high value for the detection of schistosomiasis. We determined the SEA-specific IgG, IgG1, and IgG4 levels in sera of study subjects by ELISA. SEA specific IgG, IgG1 and IgG4 levels were high in chronic schistosomiasis patients but negative or low in healthy controls (Fig. 4).
Levels of SEA-antibodies in sera from schistosomiasis patients and HC. (A) Levels of serum SEA-specific IgG; (B) Levels of serum SEA-specific IgG1; (C) Levels of serum SEA-specific IgG4. Data are expressed as the mean ± SD. (** p < 0.01).
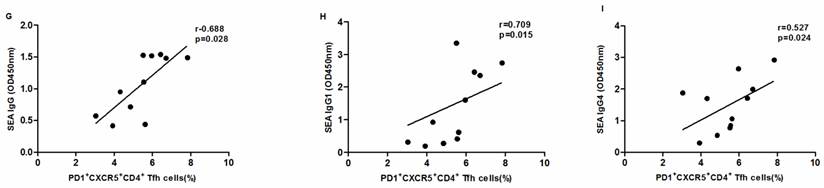
The levels of serum SEA specific antibodies correlated with the frequency of circulating PD1+ CXCR5+ CD4+ Tfh cells in schistosomiasis patients
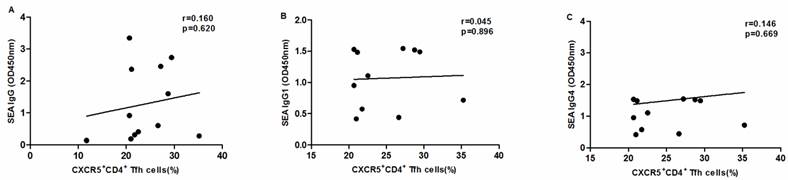
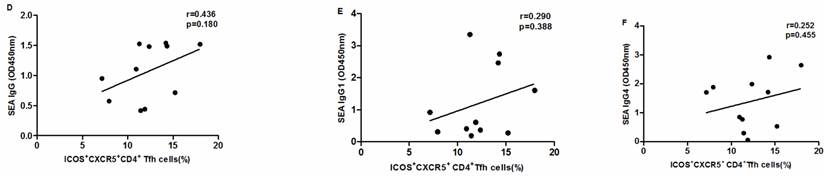
SEA specific antibody levels were significantly higher in chronic schistosomiasis patients than in healthy controls. We also analyzed whether SEA specific antibodies correlated with Tfh cells in chronic schistosomiasis patients. Positive correlation was found between the levels of SEA specific IgG, IgG1, IgG4 and the frequency of PD1+CXCR5+CD4+ Tfh cells (Fig.5G-I). However, no significant correlation was observed between the percentages of other circulating Tfh cells and SEA specific antibodies in sera (Fig. 5A-F).
Correlation analysis of the levels of SEA specific IgG, IgG1 and IgG4 with the percentages of circulating Tfh cells in schistosomiasis patients (n = 11). Relationship of the percentages of CXCR5+ CD4+Tfh cells with (A) SEA specific IgG, (B) SEA specific IgG1, and (C) SEA specific IgG4. Relationship of the percentages of ICOS+CXCR5+ CD4+Tfh cells with (D) SEA specific IgG, (E) SEA specific IgG1, and (F) SEA specific IgG4. The percentages of PD-1+CXCR5+ CD4+Tfh cells were positively associated with (G) SEA specific IgG, (H) SEA specific IgG1, and (I) SEA specific IgG4 (p < 0.05).
Discussion
More and more studies have focused on the role of Tfh cells in infectious and autoimmune diseases. Recent work indicated that Tfh cells can control antigen specific B cell immunity [22] and help the production of antibodies in response to foreign antigens [23]. However, the mechanisms by which Tfh cells and associated molecules regulate B cell production of antibodies in humans remain largely unknown. Several studies indicated that the frequency of Tfh cells in patients with autoimmune diseases such as systemic lupus erythematosus and autoimmune thyroid disease was not significantly higher than in healthy controls [24]. In contrast, paradoxical results have been obtained in that a higher frequency of Tfh cells was observed in patients with chronic HIV infection [14] and chronic HBV infection [25] than in HC groups. Our findings showed that the frequencies of CXCR5 +CD4+ Tfh cells were increased in chronic schistosomiasis patients compared to healthy controls. In addition to the increased frequency of these cells, the phenotype of CXCR5 +CD4+ Tfh cells was altered. The expression of ICOS and PD-1 in chronic schistosomiasis patients was significantly increased compared to healthy controls. This indicated that chronic schistosomiasis infection induces CXCR5+CD4+ Tfh cell differentiation.
The function of Tfh cells is closely related to the expression of markers such as CXCR5, PD-1, IL-21, ICOS and Bcl6. PD-1 plays a major role in inhibiting T cell responses. One study indicated that PD-1 was increased during a viral infection to inhibit chronically activated T cells from causing immunopathology or becoming autoreactive [26]. However, another recent study indicated that the expression of PD-1 in Tfh cells regulates the formation of GC, the survival of plasma cells, and IL-21 production [27]. In the present study, the expression of PD-1 was increased in CXCR5+CD4+ Tfh cells in patients with chronic schistosomiasis infection compared to healthy controls. IL-21 is a known critical cytokine for the formation of GC, the development of Tfh cells with ICOS or PD-1 expression, and the production of antibodies [28,29]. In some autoimmune diseases, the levels of IL-21 were significantly associated with the frequency of circulating Tfh cells with ICOS or PD-1 expression [30]. Higher expression of IL-21 mRNA and higher levels of IL-21 in serum were observed in chronic schistosomiasis patients. Moreover, the levels of IL-21 in serum positively correlated with the frequency of PD-1high CXCR5+CD4+ Tfh cells in chronic schistosomiasis patients. Tfh cells are mainly responsible for assisting B cells to induce antibody response. SEA specific antibodies are usually how schistosomiasis is diagnosed, in addition to providing protection for the host. Therefore, we further investigated the relationship between circulating Tfh cells and SEA antibodies (SEA IgG, IgG1, and IgG4) in sera from chronic schistosomiasis patients. We found that the frequencies of circulating PD-1high CXCR5+CD4+ Tfh cells positively correlated with the concentration of the SEA antibodies. However, no significant correlation was observed between the percentages of CXCR5+CD4+ Tfh cells or ICOShigh CXCR5+CD4+ Tfh cells and SEA-specific antibodies in sera. Thus, our data suggest that PD-1high CXCR5+CD4+ Tfh cells may be involved in the production of SEA antibodies by the secretion of IL-21 in chronic schistosomiasis patients. The mechanism of PD-1high CXCR5+ CD4+ Tfh in regulating antibody production in the schistosomiasis will be investigated in future studies.
High expression of ICOS was also observed in chronic schistosomiasis patients compared to healthy controls. ICOS belongs to the CD28 family and plays an important role in Tfh cell recruitment to the follicle and maintenance of Tfh cells. It has been shown that ICOS deficiency in humans and mice causes a reduction in the number of Tfh cells and B cell maturation defects [31,32]. Recent study identified that ICOS expression in Tfh cells participated in the formation of hepatic granuloma in mice infected with S. japonicum [13]. These chronic patients are asymptomatic carriers; therefore, we inferred that only PD-1high CXCR5+CD4+ Tfh cells correlate with Abs. Further studies are needed to determine whether the expression of ICOS in CXCR5+CD4+ Tfh cells was associated with liver damage in different stages of schistosomiasis.
The transcription factors, Bcl-6 and c-Maf, were selectively expressed in Tfh cells. They are essential for the differentiation of Tfh cells, but we did not observe increased mRNA levels of Bcl-6 and c-Maf in serum samples from patients with chronic schistosomiasis compared to healthy controls. A previous study demonstrated that GC Tfh cells expressed high levels of Bcl-6, while circulating CXCR5+CD4+ Tfh cells failed to express increased levels of Bcl-6 [33]. Our data consistently indicated that the expression levels of Bcl-6 and c-Maf mRNA were not raised in chronic schistosomiasis in circulating Tfh cells.
Conclusions
Our study reveals that circulating Tfh cells may be involved in immune responses in chronic schistosomiasis infection via markers and secreted cytokines. Our data indicated that frequencies of CXCR5+ CD4+ Tfh cells and the expression of ICOS and PD-1 were significantly higher in chronic schistosomiasis patients than in HC. Moreover, PD-1high CXCR5+ CD4+ Tfh cell levels positively correlated with IL-21 cytokine levels and the concentration of SEA-specific antibodies in sera from patients with chronic schistosomiasis, indicating that PD-1high CXCR5+ CD4+ Tfh cells might promote production of SEA specific antibodies by secretion of IL-21 in chronic schistosomiasis patients. The mechanism of PD-1high CXCR5+ CD4+ Tfh in regulating B-cell responses and antibody production will be investigated in future studies.
Abbreviations
Tfh, T follicular helper; HC, healthy control; SEA, soluble egg antigens; ELISA, enzyme-linked immunosorbent assay; Th, T helper; Treg, T regulatory; BCl-6, transcriptional repressor B-cell lymphoma 6; IL-21, interleukin 21; IHA, indirect hemagglutination antibody; PBMCs, peripheral blood mononuclear cells; OD, optical density.
Acknowledgements
The authors are grateful to Professor Yiwei Chu in Fudan University and Professor Shengjun Wang in Jiangsu University for technical guidance. We also thank Professor Yongkang He for providing samples at Institute of Prevention and Control of schistosomiasis in Hunan Province, China. This work was supported by grants from the National Natural Science Foundation (No. 81371841 to JC and No. 81202301 to YW).
Author Contributions
Conceived and designed the experiments: YZ YW JC. Performed the experiments: YZ YJ YW HL YS ZY YH YX. Analyzed the data: YZ YJ YW JC. Contributed reagents/materials/analysis tools: JC YW. Wrote the manuscript: YZ YJ YW JC.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Ross AG, Bartley PB, Sleigh AC. et al. Schistosomiasis. N Engl J Med. 2002;346:1212-1220
2. Gryseels B, Polman K, Clerinx J. et al. Human schistosomiasis. Lancet. 2006;368:1106-1118
3. Cai P, Piao X, Liu S. et al. MicroRNA-gene expression network in murine liver during Schistosoma japonicum infection. PLoS One. 2013;8:e67037
4. Pearce EJ, MacDonald AS. The immunobiology of schistosomiasis. Nat Rev Immunol. 2002;2:499-511
5. Kaplan MH, Whitfield JR, Boros DL. et al. Th2 cells are required for the Schistosoma mansoni egg-induced granulomatous response. J Immunol. 1998;160:1850-1856
6. Wen X, He L, Chi Y. et al. Dynamics of Th17 cells and their role in Schistosoma japonicum infection in C57BL/6 mice. PLoS Negl Trop Dis. 2011;5:e1399
7. Rutitzky LI, Stadecker MJ. Exacerbated egg-induced immunopathology in murine Schistosoma mansoni infection is primarily mediated by IL-17 and restrained by IFN-gamma. Eur J Immunol. 2011;41:2677-2687
8. Singh KP, Gerard HC, Hudson AP. et al. Retroviral Foxp3 gene transfer ameliorates liver granuloma pathology in Schistosoma mansoni infected mice. Immunology. 2005;114:410-417
9. Breitfeld D, Ohl L, Kremmer E. et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B cell follicles, and support immunoglobulin production. J Exp Med. 2000;192:1545-1552
10. Nurieva RI, Chung Y, Martinez GJ. et al. Bcl6 mediates the development of T follicular helper cells. Science. 2009;325(5943):1001-1005
11. Xie J, Cui D, Liu Y. et al. Changes in follicular helper T cells in idiopathic thrombocytopenic purpura patients. Int J Biol Sci. 2015;11:220-229
12. Kroenke MA, Eto D, Locci M. et al. Bcl6 and Maf cooperate to instruct human follicular helper CD4 T cell differentiation. J Immunol. 2012;188:3734-3744
13. Ma CS, Suryani S, Avery DT. et al. Early commitment of naive human CD4(+) T cells to the T follicular helper (T(FH)) cell lineage is induced by IL-12. Immunol Cell Biol. 2009;87:590-600
14. Johnston RJ, Poholek AC, DiToro D. et al. Bcl6 and Blimp-1 are reciprocal and antagonistic regulators of T follicular helper cell differentiation. Science. 2009;325:1006-1010
15. Chen X, Yang X, Li Y. et al. Follicular Helper T Cells Promote Liver Pathology in Mice during Schistosoma japonicum Infection. PLoS Pathog. 2014;10:e1004097
16. Strauss L, Bergmann C, Szczepanski MJ. et al. Expression of ICOS on human melanoma-infiltrating CD4+CD25high Foxp3+ T regulatory cells: implications and impact on tumor-mediated immune suppression. J Immunol. 2008;180:2967-2980
17. Feng J, Lu L, Hua C. et al. High frequency of CD4+ CXCR5+ TFH cells in patients with immune-active chronic hepatitis B. PLoS One. 2011;6:e21698
18. Crotty S. Follicular helper CD4 T cells (TFH). Annu Rev Immunol. 2011;29:621-663
19. Jin L, Yu D, Li X. et al. CD4+CXCR5+ follicular helper T cells in salivary gland promote B cells maturation in patients with primary Sjogren's syndrome. Int J Clin Exp Pathol. 2014;7:1988-1996
20. Simpson N, Gatenby PA, Wilson A. et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 2010;62:234-244
21. Lindqvist M, van Lunzen J, Soghoian DZ. et al. Expansion of HIV-specific T follicular helper cells in chronic HIV infection. J Clin Invest. 2012;122:3271-3280
22. Fazilleau N, Mark L, McHeyzer-Williams LJ. et al. Follicular helper T cells: lineage and location. Immunity. 2009;30:324-335
23. Rasheed AU, Rahn HP, Sallusto F. et al. Follicular B helper T cell activity is confined to CXCR5(hi)ICOS(hi) CD4 T cells and is independent of CD57 expression. Eur J Immunol. 2006;36:1892-1903
24. Zhu C, Ma J, Liu Y. et al. Increased frequency of follicular helper T cells in patients with autoimmune thyroid disease. J Clin Endocrinol Metab. 2012;97:943-950
25. Li Y, Ma S, Tang L. et al. Circulating chemokine (C-X-C Motif) receptor 5(+) CD4(+) T cells benefit hepatitis B e antigen seroconversion through IL-21 in patients with chronic hepatitis B virus infection. Hepatology. 2013;58:1277-1286
26. Sharpe AH, Wherry EJ, Ahmed R. et al. The function of programmed cell death 1 and itsligands in regulating autoimmunity and infection. Nat Immunol. 2007;8:239-245
27. Good-Jacobson KL, Szumilas CG, Chen L. et al. PD-1 regulates germinal center B cell survival and the formation and affinity of long-lived plasma cells. Nat Immunol. 2010;11:535-542
28. Ozaki K, Spolski R, Feng CG. et al. A critical role for IL-21 in regulating immunoglobulin production. Science. 2002;298:1630-1634
29. Zotos D, Coquet JM, Zhang Y. et al. IL-21 regulates germinal center B cell differentiation and proliferation through a B cell-intrinsic mechanism. J Exp Med. 2010;207:365-378
30. Terrier B, Costedoat-Chalumeau N, Garrido M. et al. Interleukin 21 correlates with T cell and B cell subset alterations in systemic lupus erythematosus. J Rheumatol. 2012;39:1819-1828
31. Bossaller L, Burger J, Draeger R. et al. ICOS deficiency is associated with a severe reduction of CXCR5+CD4 germinal center Th cells. J Immunol. 2006;177:4927-4932
32. Bruchfeld A, Benedek S, Hilderman M. et al. Rituximab for minimal change disease in adults: long-term follow-up. Nephrol Dial Transplant. 2014;29:851-856
33. Morita R, Schmitt N, Bentebibel SE. et al. Human blood CXCR5(+)CD4(+) T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity. 2011;34:108-121
Author contact
Corresponding authors: Mail address: National Institute of Parasitic Diseases, Chinese Center for Disease Control and Prevention, Shanghai 200025, China. Telephone: +86-21-64735258. Fax: +86-21-64376308. E-mails: caojpcom (JC) or jiangyy083com (YJ)

 Global reach, higher impact
Global reach, higher impact