ISSN: 1449-2288
Int J Biol Sci 2016; 12(1):63-71. doi:10.7150/ijbs.12690 This issue Cite
Research Paper
uPA Attenuated LPS-induced Inflammatory Osteoclastogenesis through the Plasmin/PAR-1/Ca2+/CaMKK/AMPK Axis
1. Department of Clinical Pathological Biochemistry, Faculty of Pharmaceutical Science, Doshisha Women's College of Liberal Arts, 97-1 Kodo Kyo-tanabe, Kyoto 610-0395, Japan.
2. Division of Cellular Biosignal Sciences, Department of Biochemistry, Iwate Medical University, 2-1-1 Nishitokuta, Yahaba-cho, Shiwa-gun, Iwate 028-3694, Japan.
3. Kinki University Faculty of Medicine 377-2 Ohnohigashi, Osaka-sayama 589-8511, Japan.
Abstract
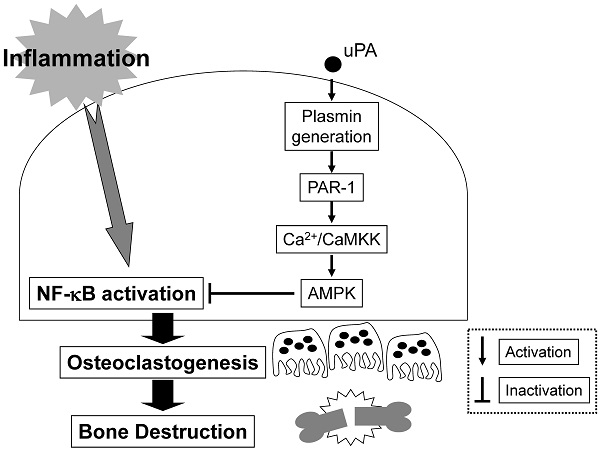
Chronic inflammatory diseases, such as rheumatoid arthritis and periodontitis-caused bone destruction, results from an increase of bone-resorbing osteoclasts (OCs) induced by inflammation. However, the detailed mechanisms underlying this disorder remain unclear. We herein investigated that the effect of urokinase-type plasminogen activator (uPA) on inflammatory osteoclastogenesis induced by lipopolysaccharide (LPS), which is a potent stimulator of bone resorption in inflammatory diseases. We found that the uPA deficiency promoted inflammatory osteoclastogenesis and bone loss induced by LPS. We also showed that LPS induced the expression of uPA, and the uPA treatment attenuated the LPS-induced inflammatory osteoclastogenesis of RAW264.7 mouse monocyte/macrophage lineage cells. Additionally, we showed that the uPA-attenuated inflammatory osteoclastgenesis is associated with the activation of plasmin/protease-activated receptor (PAR)-1 axis by uPA. Moreover, we examined the mechanism underlying the effect of uPA on inflammatory osteoclastogenesis, and found that uPA/plasmin/PAR-1 activated the adenosine monophosphate-activated protein kinase (AMPK) pathway through Ca2+/calmodulin dependent protein kinase kinase (CaMKK) activation, and attenuated inflammatory osteoclastogenesis by inactivation of NF-κB in RAW264.7 cells. These data suggest that uPA attenuated inflammatory osteoclastogenesis through the plasmin/PAR-1/Ca2+/CaMKK/AMPK axis. Our findings may provide a novel therapeutic approach to bone loss caused by inflammatory diseases.
Keywords: uPA, plasmin, AMPK, osteoclasts, inflammation

 Global reach, higher impact
Global reach, higher impact