Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2016; 12(8):1032-1040. doi:10.7150/ijbs.15624 This issue Cite
Research Paper
Rapamycin Reduced Ischemic Brain Damage in Diabetic Animals Is Associated with Suppressions of mTOR and ERK1/2 Signaling
1. Department of Endocrinology, General Hospital of Ningxia Medical University, Yinchuan 750004, China
2. Department of Pharmaceutical Sciences, Biomanufacturing Research Institute Biotechnology Enterprise (BRITE), North Carolina Central University, 1801 Fayetteville Street, Durham, NC 27707, USA
3. Neuroscience Center, General Hospital of Ningcia Medical University, and Key Laboratory of Craniocerebral Diseases of Ningxia Hui Autonomous Region, Yinchuan 750004, China
4. Department of Human Anatomy, Histology and Embryology, Ningxia Medical University, Yinchuan 75004, China
* These two authors contributed equally to this work.
Abstract
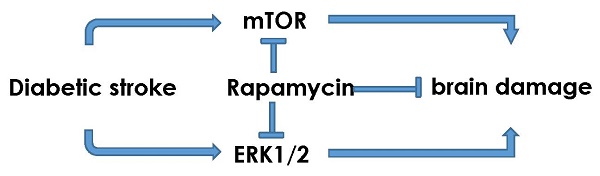
The objectives of the present study are to investigate the activation of mTOR and ERK1/2 signaling after cerebral ischemia in diabetic rats and to examine the neuroprotective effects of rapamycin. Ten minutes transient global cerebral ischemia was induced in straptozotocin-induced diabetic hyperglycemic rats and non-diabetic, euglycemic rats. Brain samples were harvested after 16 h of reperfusion. Rapamycin or vehicle was injected 1 month prior to the induction of ischemia. The results showed that diabetes increased ischemic neuronal cell death and associated with elevations of p-P70S6K and Ras/ERK1/2 and suppression of p-AMPKα. Rapamycin ameliorated diabetes-enhanced ischemic brain damage and suppressed phosphorylation of P70S6K and ERK1/2. It is concluded that diabetes activates mTOR and ERK1/2 signaling pathways in rats subjected to transient cerebral ischemia and inhibition of mTOR by rapamycin reduces ischemic brain damage and suppresses the mTOR and ERK1/2 signaling in diabetic settings.
Keywords: Cerebral ischemia, Diabetes, ERK1/2, mTOR, Rapamycin

 Global reach, higher impact
Global reach, higher impact