ISSN: 1449-2288
Int J Biol Sci 2016; 12(9):1041-1051. doi:10.7150/ijbs.16134 This issue Cite
Research Paper
Multiple Growth Factors, But Not VEGF, Stimulate Glycosaminoglycan Hyperelongation in Retinal Choroidal Endothelial Cells
1. School of Health and Biomedical Sciences, RMIT University, Bundoora, VIC 3083 Australia;
2. Department of Immunology, Monash University, Melbourne 3004 VIC, Australia.
3. Faculty of Health Sciences, University of Macau, Taipa, Macau, China;
4. State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou 510006, China.
5. School of Pharmacy. The University of Queensland, Wooloongabba, QLD 4102, Australia.
*These two authors contributed equally to this work.
Abstract
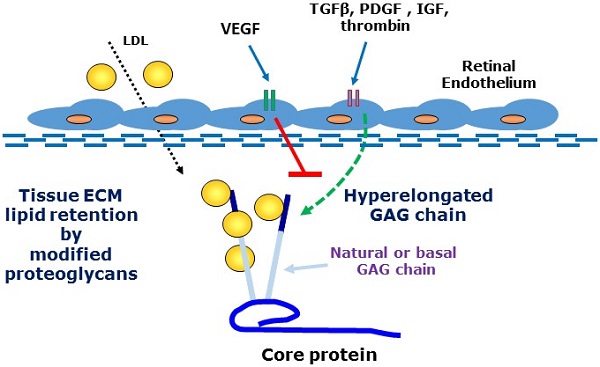
A major feature of early age-related macular degeneration (AMD) is the thickening of Bruch's membrane in the retina and an alteration in its composition with increased lipid deposition. In certain pathological conditions proteoglycans are responsible for lipid retention in tissues. Growth factors are known to increase the length of glycosaminoglycan chains and this can lead to a large increase in the interaction between proteoglycans and lipids. Using choroidal endothelial cells, we investigated the effects of a number of AMD relevant growth factors TGFβ, thrombin, PDGF, IGF and VEGF on proteoglycan synthesis. Cells were characterized as of endothelial origin using the specific cell markers endothelial nitric oxide synthesis and von Willebrand factor and imaged using confocal microscopy. Cells were treated with growth factors in the presence and absence of the appropriate inhibitors and were radiolabeled with [35S]-SO4. Proteoglycans were isolated by ion exchange chromatography and sized using SDS-PAGE. Radiosulfate incorporation was determined by the cetylpyridinium chloride (CPC) precipitation technique. To measure cellular glycosaminoglycan synthesizing capacity we added xyloside and assessed the xyloside-GAGs by SDS-PAGE. TGFβ, thrombin, PDGF & IGF dose-dependently stimulated radiosulfate incorporation and GAG elongation as well as xyloside-GAG synthesis, however VEGF treatment did not stimulate any changes in proteoglycan synthesis. VEGF did not increase pAKT but caused a large increase in pERK relative to the response to PDGF. Thus, AMD relevant agonists cause glycosaminoglycan hyperelongation of proteoglycans synthesised and secreted by retinal choroidal endothelial cells. The absence of a response to VEGF is intriguing and identifies proteoglycans as a novel potential target in AMD. Future studies will examine the relevance of these changes to enhanced lipid binding and the development of AMD.
Keywords: Age related macular degeneration, proteoglycans, growth factors, Bruch's membrane, VEGF, xyloside.

 Global reach, higher impact
Global reach, higher impact