Impact Factor ISSN: 1449-2288
Int J Biol Sci 2017; 13(11):1387-1397. doi:10.7150/ijbs.21635 This issue Cite
Review
Risk Factors and Preventions of Breast Cancer
1. Key Lab of Vaccine against Hemorrhagic Fever with Renal Syndrome, Zhejiang Provincial Center for Disease Control and Prevention, Hangzhou, China;
2. Centre of Laboratory Medicine, Zhejiang Provincial People's Hospital.
Received 2017-6-26; Accepted 2017-8-28; Published 2017-11-1
Abstract
Breast cancer is the second leading cause of cancer deaths among women. The development of breast cancer is a multi-step process involving multiple cell types, and its prevention remains challenging in the world. Early diagnosis of breast cancer is one of the best approaches to prevent this disease. In some developed countries, the 5-year relative survival rate of breast cancer patients is above 80% due to early prevention. In the recent decade, great progress has been made in the understanding of breast cancer as well as in the development of preventative methods. The pathogenesis and tumor drug-resistant mechanisms are revealed by discovering breast cancer stem cells, and many genes are found related to breast cancer. Currently, people have more drug options for the chemoprevention of breast cancer, while biological prevention has been recently developed to improve patients' quality of life. In this review, we will summarize key studies of pathogenesis, related genes, risk factors and preventative methods on breast cancer over the past years. These findings represent a small step in the long fight against breast cancer.
Keywords: breast cancer, pathogenesis, risk factor, prevention.
Introduction
Breast cancer is one of the most common cancers in women worldwide, accounting for approximately 570,000 deaths in 2015. Over 1.5 million women (25% of all women with cancer) are diagnosed with breast cancer every year throughout the world [1,2]. In America, it is estimated that 30% of all new cancer cases (252,710) among women are breast cancer in 2017 [3]. Breast cancer is a metastatic cancer and can commonly transfer to distant organs such as the bone, liver, lung and brain, which mainly accounts for its incurability. Early diagnosis of the disease can lead to a good prognosis and a high survival rate. In North American, the 5-year relative survival rate of breast cancer patients is above 80% due to the timely detection of this disease [4]. Mammography is a widely used screening approach in the detecting of breast cancer and proved to help reduce the mortality effectively. Other screening methods, such as Magnetic Resonance Imaging (MRI), which is more sensitive than mammography, have also been implemented and studied during the last decade [5]. There're numerous risk factors such as sex, aging, estrogen, family history, gene mutations and unhealthy lifestyle, which can increase the possibility of developing breast cancer [6]. Most breast cancer occur in women and the number of cases is 100 times higher in women than that in men [3]. Although the incidence rate of breast cancer in America increases year after year, the mortality rate decreases due to the widespread early screenings and advanced medical therapies. Biological therapies have been developed in recent years and proved to be beneficial for breast cancer. Here, we will focus on studies of the pathogenesis, related genes, risk factors and preventions of breast cancer over the past years.
Pathogenesis
Breast tumors usually start from the ductal hyperproliferation, and then develop into benign tumors or even metastatic carcinomas after constantly stimulation by various carcinogenic factors. Tumor microenvironments such as the stromal influences or macrophages play vital roles in breast cancer initiation and progression. The mammary gland of rats could be induced to neoplasms when only the stroma was exposed to carcinogens, not the extracellular matrix or the epithelium [7,8]. Macrophages can generate a mutagenic inflammatory microenvironment, which can promote angiogenesis and enable cancer cells to escape immune rejection [9,10]. Different DNA methylation patterns have been observed between the normal and tumor-associated microenvironments, indicating that epigenetic modifications in the tumor microenvironment can promote the carcinogenesis [11,12]. Recently, a new subclass of malignant cells within tumors called the cancer stem cells (CSCs) are observed and associated with tumor initiation, escape and recurrence. This small population of cells, which may develop from stem cells or progenitor cells in normal tissues, have self-renewal abilities and are resistant to conventional therapies such as chemotherapy and radiotherapy [13-15]. Breast cancer stem cells (bCSCs) were first identified by Ai Hajj and even as few as 100 bCSCs could form new tumors in the immunocompromised mice [16]. bCSCs are more likely to originate from luminal epithelial progenitors rather than from basal stem cells [17]. Signaling pathways including Wnt, Notch, Hedgehog, p53, PI3K and HIF are involved in the self-renewal, proliferation and invasion of bCSCs [18-21]. However, more studies are needed to understand bCSCs and to develop novel strategies to directly eliminate the bCSCs.
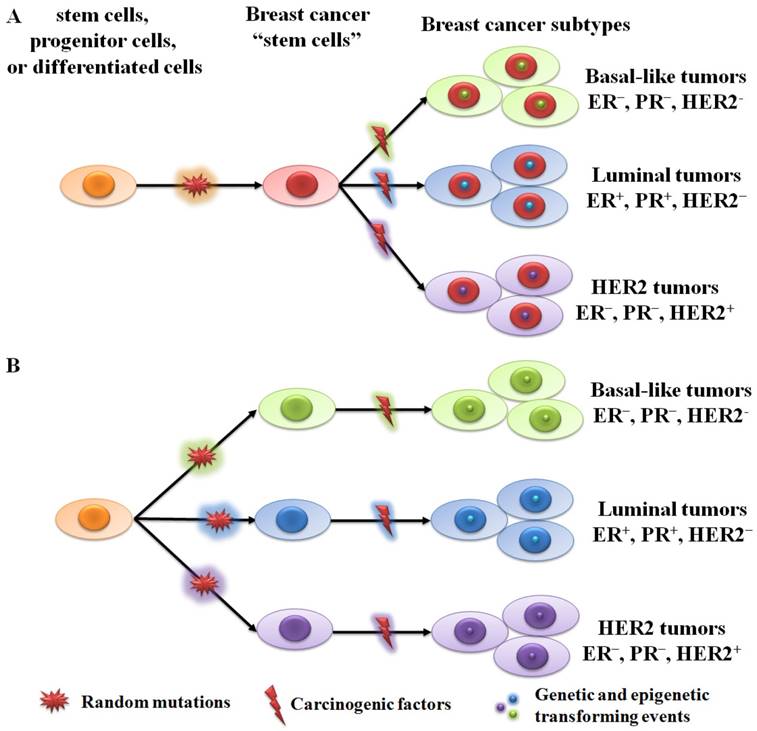
There're two hypothetical theories for breast cancer initiation and progression: the cancer stem cell theory and the stochastic theory [11,22]. The cancer stem cell theory suggests that all tumor subtypes are derived from the same stem cells or transit-amplifying cells (progenitor cells). Acquired genetic and epigenetic mutations in stem cells or progenitor cells will lead to different tumor phenotypes (Figure 1A). The stochastic theory is that each tumor subtype is initiated from a single cell type (stem cell, progenitor cell, or differentiated cell) (Figure 1B). Random mutations can gradually accumulate in any breast cells, leading to their transformation into tumor cells when adequate mutations have accumulated. Although both theories are supported by plenty of data, neither can fully explain the origin of human breast cancer.
Two hypothetical theories of breast cancer initiation and progression. (A) All subtypes of tumor are derived from the same stem cells or progenitor cells. Different tumor phenotypes are then determined by subtype-specific transforming events. (B) Each tumor subtype is initiated from a single cell type (stem cell, progenitor cell, or differentiated cell). Random mutations can gradually accumulate in any breast cells, leading to their transformation into tumor cells when an adequate number of mutations have accumulated.
Genes related to breast cancer
Lots of genes have been identified in relation to breast cancer. Mutations and abnormal amplification of both oncogenes and anti-oncogenes play key roles in the processes of tumor initiation and progression.
BRCA1/2
Breast cancer associated gene 1 and 2 (BRCA1 and BRCA2) are two famous anti-oncogenes for breast cancer risk. BRCA1 and BRCA2 are located on chromosome 17q21 and 13q12, respectively. They both encode tumor suppressor proteins. BRCA1 deficiency leads to the dysregulation of cell cycle checkpoint, abnormal centrosome duplication, genetic instability and eventually apoptosis [23,24]. BRCA1 expression is repressed by “pocket proteins” such as p130, p107 and the retinoblastoma protein in an E2F-dependent manner. The BRCA1 gene has been shown to form a loop between the promoter, introns, and terminator regions, which regulates the expression of this gene via interactions with its own promoter [25,26]. BRCA2 protein regulates recombinational repair in DNA double-strand breaks by interacting with RAD51 and DMC1 [27,28]. BRCA2-associated breast cancers are more likely to be high-grade invasive ductal carcinomas, but with a luminal phenotype [29]. The risk of breast cancer could be increased greatly if an individual inherits deleterious mutations in either BRCA1 or BRCA2 genes. BRCA1/2 mutations are inherited in an autosomal dominant manner even though the second allele is normal. Totally, about 20-25% of hereditary breast cancers and 5-10% of all breast cancers are caused by BRCA1/2 mutations [30,31]. A meta-analysis by Chen showed that the breast cancer risk ratio in women older than 70 years carrying BRCA1 or BRCA2 mutations was 57% and 49%, respectively [32].
HER2
Human epidermal growth factor receptor 2, also known as c-erbB-2, is an important oncogene in breast cancer and located on the long arm of human chromosome 17 (17q12). The homologene in mice is Neu, which was first identified in 3-methylcholanthrene induced rat neuroblastoma cells [33]. The expression of HER2 gene is activated mainly through the gene amplification and re-arrangement. HER2 protein is an epidermal growth factor receptor (EGFR) of tyrosine kinase family and form heterodimers with other ligand-bound EGFR family members such as Her3 and Her4, thus to activate downstream signaling pathways [34]. Knockout of HER2 in mouse models disrupts normal mammary duct formation. Overexpression of HER2, which is detected in about 20% of primary breast cancers, increases the number of cancer stem cells by PTEN/Akt/mTORC1 signaling, and indicates poor clinical outcomes [35,36].
Epidermal Growth Factor Receptor (EGFR)
EGFR, also known as c-erbB-1 or Her1 in humans, is located on the short arm of chromosome 7 (7p12). The EGFR protein is a cell surface glycoprotein of tyrosine kinase family and is activated by binding to EGF, TGF-α, amphiregulin, betacellulin and so on. The downstream signaling pathways of EGFR including PI3K, Ras-Raf-MAPK and JNK are triggered to promote cell proliferation, cell invasion, angiogenesis and to protect cells against apoptosis [37, 38]. Overexpression of EGFR is found in more than 30% of cases of the inflammatory breast cancer (IBC), a very aggressive subtype of breast cancer. Patients with EGFR-positive IBC have a poorer prognosis than those with EGFR-negative tumors [39,40]. More than half of triple-negative breast cancer (TNBC) cases, characterized by the absence of estrogen receptor (ER), progesterone receptor (PR) expression and HER2 amplification, also have EGFR overexpression [41]. Therefore, targeting the EGFR pathway might be a promising therapy for these malignant tumors.
c-Myc
This gene is located on the long arm of chromosome 8 (8q24) and encodes for the Myc protein, a transcription factor containing the bHLH/LZ (basic Helix-Loop-Helix Leucine Zipper) domain. Genome-wide screening shows that 15% of all genes are regulated by the Myc protein mainly through binding on the E-box consensus (CACGTG) and recruiting histone acetyltransferases (HATs) or DNA methyltransferases [42,43]. Some of the Myc-regulated genes such as MTA1, hTERT and PEG10 play vital roles in breast cancer initiation and progression. The overexpression of c-Myc is predominantly observed in the high-grade, invasive stage of breast carcinomas, while no c-Myc amplification is detected in the benign tissues [44, 45].
Other related genes
Ras
There're three members in the Ras gene family: H-ras, K-ras and N-ras, located on the chromosome of 11 (11p15), 12 (12p12) and 1 (1p22) respectively. The proteins encoded by these genes are extremely homologous, and they belong to the small guanosine triphosphate (GTP)-binding protein (G protein) superfamily [46]. Point mutations are commonly associated with the overexpression of these three human Ras genes, and most are missense mutations located at the coding domain for GTP binding. Though mutations of Ras proteins are infrequently in breast cancer (<5%), the abnormality of Ras signal transduction pathway are observed in both benign and malignant mammary tissues [47]. H-ras can cooperate with B lymphoma moloney murine leukaemia virus insertion region-1 (BMI1) to promote proliferation, invasion, and to inhibit apoptosis in breast cancer cells [48]. H-ras overexpression is detected in both primary and advanced breast cancer patients, indicating a poor prognosis [49, 50].
Risk factors
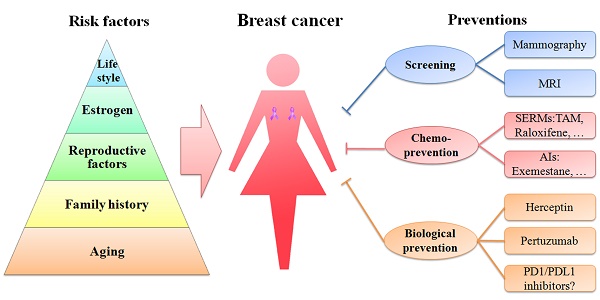
A schematic diagram of risk factors is depicted in a pyramid-style structure (Figure 2).
Schematic diagram of risk factors and preventions of breast cancer. Age, family history, reproductive factors, estrogen and life style are five important risk factors of breast cancer, represented in the pyramid chart. Screening (mammography and MRI), chemoprevention (with SERMs and AIs) and biological prevention (using Herceptin and pertuzumab) are currently being used to prevent breast cancer. PD1/PDL1 inhibitors are immunotherapy drugs and might be promising strategies in treating TNBC.
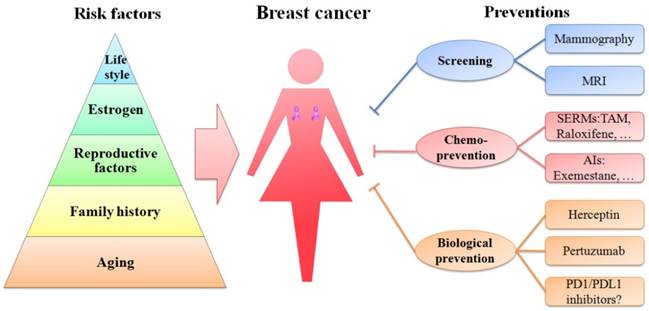
Aging
Besides sex, aging is one of the most important risk factors of breast cancer, because the incidence of breast cancer is highly related to the increasing age. In 2016, approximately 99.3% and 71.2% of all breast cancer-associated deaths in America were reported in women over the age of 40 and 60, respectively [3]. Therefore, it is necessary to have a mammography screening ahead of time in women aged 40 or older.
Family history
Nearly a quarter of all breast cancer cases are related to family history [65]. Women, whose mother or sister has a breast cancer, are prone to this disease. A cohort study of over 113,000 women in UK demonstrated that women with one first-degree relative with breast cancer have a 1.75-fold higher risk of developing this disease than women without any affected relatives. Moreover, the risk becomes 2.5-fold or higher in women with two or more first-degree relatives with breast cancer [65]. The inherited susceptibility to breast cancer is partially attributed to the mutations of breast cancer related genes such as BRCA1 and BRCA2.
Additional genes associated with breast cancer
| Gene | Location | Function | Abnormality in Breast cancer | Ref. |
|---|---|---|---|---|
| P53 | 17p13.1 | tumor suppressor gene | Mutations in 30% of breast cancers | [51,52] |
| NME1 | 17q21.3 | metastasis-suppressor gene | SNP of NME1 gene associated with higher breast cancer-specific mortality (HR=1.4) and patients with an early-stage cancer (HR=1.7) | [53,54] |
| RB1 | 13q14.2 | tumor suppressor gene | Rb1 inactivation in 20-35% of breast cancers | [55,56] |
| PTEN | 10q23.3 | tumor suppressor gene | Loss of PTEN protein expression in up to 33% of breast cancers | [57,58] |
| ATM | 11q22-q23 | tumor suppressor gene | Mutation of ATM increases the risk of 2- to 3-fold in general, and 5- to 9-fold in women under age 50 | [59] |
| CDH1 (E-cadherin) | 16q22.1 | tumor suppressor gene | Inactivation of CDH1 in 85% of lobular breast carcinomas | [60] |
| FHIT | 3p14.2 | putative tumor suppressor gene | The rate of FHIT hypermethylation in breast cancer was 8.4-fold higher than that in normal breast tissues | [61] |
| Maspin | 18q21.33 | tumor suppressor gene | Expression of Maspin in 20-80% invasive breast cancer | [62,63] |
| PIK3CA | 3q26.3 | oncogene | Mutations in 37% of the HR+/HER2- metastatic and 40% of early breast cancer | [56] |
| CCND1 (Cyclin D1) | 11q13 | oncogene | Overexpression in 50% of breast tumors | [64] |
Notes: SNP-Single Nucleotide Polymorphisms; HR-Hazard ratio
Reproductive factors
Reproductive factors such as early menarche, late menopause, late age at first pregnancy and low parity can increase the breast cancer risk. Each 1-year delay in menopause increases the risk of breast cancer by 3%. Each 1-year delay in menarche or each additional birth decreases the risk of breast cancer by 5% or 10%, respectively [66-68]. A recent Norwegian cohort study showed that a hazard ratio (HR) is 1.54 between late (≥35 years) and early (<20 years) age at first birth [69]. Reproductive factors are strongly associated with the ER status, with differences in the odds ratios (OR) between ER+ and ER- breast cancer for parity (OR: 0.7 vs. 0.9 for ≥3 births vs. nulliparae) and age at the first birth (OR: 1.6 vs. 1.2 for age ≥30 vs. <25 years) [70].
Estrogen
Both endogenous and exogenous estrogens are associated with the risk of breast cancer. The endogenous estrogen is usually produced by the ovary in premenopausal women and ovariectomy can reduce the risk of breast cancer [71]. The main sources of exogenous estrogen are the oral contraceptives and the hormone replacement therapy (HRT). The oral contraceptives have been widely used since 1960s and the formulations have been upgraded to reduce side-effects. However, the OR is still higher than 1.5 for African American women and Iranian populations [72,73]. Nevertheless, oral contraceptives do not increase the risk of breast cancer in women who stop to use them for more than 10 years [66]. HRT involves the administration of exogenous estrogen or other hormones for the menopausal or postmenopausal women. A number of studies have shown that the use of HRT can increase the breast cancer risk. The Million Women Study in UK reported a relative risk (RR) of 1.66 between current users of HRT and those who never used it [74]. A cohort study of 22,929 women in Asia demonstrated HRs of 1.48 and 1.95 after HRT use for 4 and 8 years, respectively [75]. However, the risk of breast cancer has been shown to significantly decrease after two years of stopping HRT [76]. The recurrence rate is also high among breast cancer survivors who take HRT, and the HR for a new breast tumor is 3.6 [77]. Since the adverse effects of HRT were published in 2003 based on the Women's Health Initiative randomized controlled trial, the incidence rate of breast cancer in America has decreased by approximately 7% due to the reduction in the use of HRT [78].
Lifestyle
Modern lifestyles such as excessive alcohol consumption and too much dietary fat intake can increase the risk of breast cancer. Alcohol consumption can elevate the level of estrogen-related hormones in the blood and trigger the estrogen receptor pathways. A meta-analysis based on 53 epidemiological studies indicated that an intake of 35-44 grams of alcohol per day can increase the risk of breast cancer by 32%, with a 7.1% increase in the RR for each additional 10 grams of alcohol per day [79,80]. Modern western diet contains too much fat and excess intake of fat, especially the saturated fat, is associated with mortality (RR=1.3) and poor prognosis in breast cancer patients [81]. Although the relationship between smoking and breast cancer risk remains controversial, mutagens from cigarette smoke have been detected in the breast fluid from non-lactating women. The risk of breast cancer is also elevated in women who both smoke and drink (RR=1.54) [82]. Up to now, accumulating evidences demonstrate that smoking, especially at an early age, has a higher risk on breast cancer occurrence [83-86].
Preventions
Thus far, great advances have been made in clinical and theoretical studies of breast cancer (Figure 2). The current prevention methods including screening, chemoprevention and biological prevention are more direct and effective than those in the past (Figure 2). The mortality of breast cancer has decreased. However, breast cancer is still the first leading cause of cancer death among females aged 20-59 years.
Screening
Not primary tumors but the tumor metastasis causes over 90% of cancer deaths [87]. However, if breast cancer is diagnosed as a primary tumor or at an early stage of metastasis, the breast tumor could be removed by surgery and the chemotherapy could work effectively. Early detection is the cornerstone of breast cancer prevention. Mammography is an effective screening method to use low energy X-rays to obtain high-resolution images of the breast. The entire testing process only lasts for 20 minutes and it does not require any contrast-enhancing agent. Since the first recommendation for breast cancer screening by Professor Forrest, over 70% of women (aged 50-74 years) in America have been undergone breast cancer screening via mammography every 2 years [88]. A meta-analysis of 11 randomized trials showed that women aged 50-70 years had a significant reduction in breast cancer mortality after screening with mammography (RR=0.81) [89]. However, the reduction in mortality rate was not significant in women aged 40-49 years [90]. These results indicate the importance of mammography screening programs. Although the reported percentage of overdiagnosis due to mammography varies across trials, overdiagnosis is undoubtedly a serious problem that cannot be ignored during breast cancer screening.
MRI is another widely used screening tool for breast cancer. It is more sensitive than mammography in high-risk women, especially in detecting the invasive ductal carcinoma [91]. Compared to mammography, MRI is not affected by the breast density and has advantages in detecting occult primary breast cancer, axillary nodal metastasis, residual tumors after neoadjuvant chemotherapy or other small tumors [92]. Advanced MRI scanners can measure tissues as small as 0.5 mm3. However, there's no identified benefit of MRI in patient outcomes such as in the rate of detection of ipsilateral breast tumor recurrence and contralateral breast cancer incidence. The specificity of MRI is much poorer than that of mammography, with detection rates ranging from 37% to 100% [93]. Women with a family history of breast cancer have an approximately 20-25% or higher lifetime risk of breast cancer as demonstrated by MRI screening [94]. Each coin has two sides, and we should balance both the goodness and weakness. Considering its sensitivity, MRI may be a useful choice in high risk groups when the mammography results are normal.
Chemoprevention
The classical definition of chemotherapy by Sporn is “the use of pharmacologic or natural agents that inhibit the development of invasive breast cancer either by blocking the DNA damage that initiates carcinogenesis, or by arresting or reversing the progression of premalignant cells in which such damage has already occurred.” [95]. Estrogen receptor is a major target for chemotherapy because more than 70% of breast cancers are ER-positive breast cancers. Selective estrogen receptor modulators (SERMs) and the aromatase inhibitors (AIs) are two major classes of anti-estrogen drugs. SERMs are compounds that act as either agonists or antagonists of estrogen receptors. One of the most famous SERMs is tamoxifen (TAM), which has been used to treat breast cancer for more than 30 years [96]. Regardless of the number, the scale, the related areas or the time lasting in the follow-up visit of TAM's research, this drug has no doubt the most abundant clinical data in SERMs. Meanwhile, TAM is used to treat all stages of breast cancer [97]. Many large-scale trials including the Breast Cancer Prevention Trial (NSABP-1), the Royal Marsden Prevention Trial, the Italian Prevention Trial and the International Breast Cancer Intervention Study (IBIS-I Trial) have shown that TAM could reduce the risk of both invasive and non-invasive breast cancer. Despite the differences in data collection and study design, all these trials have demonstrated greater than 30% reduction in ER-positive breast cancer after 5 years treatment with TAM. However, no significant reduction has been observed in ER-negative tumors [98,99]. Nevertheless, there're some side-effects of TAM therapy. The risk of endometrial cancer, stroke, pulmonary embolism, and deep-vein thrombosis is increased in TAM-treated patients, and the risk is especially high among women older than 50 [100]. Therefore, TAM should be used individually by balancing between its toxicity and benefits.
Raloxifene, a second generation of SERMs with fewer side-effects than TAM, has been approved for the treatment of invasive breast cancer in postmenopausal women as well as osteoporosis and heart disease [101]. However, raloxifene shows no effect on ductal carcinoma in situ (DCIS) and lobular carcinoma in situ (LCIS). The Study of Tamoxifen and Raloxifene (STAR) trials showed that raloxifene was less effective than TAM [99]. Considering its less risk in endometrial cancers and thromboembolic complications, raloxifene is still a good therapeutic option for the invasive breast cancer. Several third generations of SERMs such as ospemifen, arzoxifene, lasofoxifene (LFX) and bazedoxifene (BZA) have also been discovered, but only BZA has reached the stage of clinical use. BZA has demonstrated potent effects in pre-clinical studies, but its efficacy was limited in pivotal clinical trials [102].
Recently, AIs are being used instead of TAM as the first line therapy in postmenopausal breast cancer patients. AIs reduce the plasma levels of estrogens by inhibiting aromatase, an enzyme that catalyzes the biosynthesis of estrogen from androgen [103]. There're two classes of AIs: steroidal inhibitors and non-steroidal inhibitors. Compared with exemestane, a steroidal inhibitor, non-steroidal inhibitors such as anastrozole and letrozole can bind to the enzyme's active site reversibly. However, these three third-generation AIs (exemestane, anastrozole and letrozole) have no significant differences in terms of efficacy in preventing breast cancer. Many trials such as the Italian Tamoxifen Anastrozole (ITA) trial, Intergroup Exemestane Study (IES), Austrian Breast and Colorectal Cancer Study Group (ABCSG-8), and the Breast International Group (BIG) 1-98 study have showed that in the adjuvant setting, AIs are more potent than TAM in reducing the incidence of breast cancer both as upfront monotherapy and after 2-3 years treatment with tamoxifen [104-110]. A 10-year ATAC trial study indicated fewer serious side-effects for anastrozole than for tamoxifen in postmenopausal women with early ER+ breast cancer [111]. However, there're some limitations of AIs. Because AIs inhibit the biosynthesis of estrogen, they are typically used only in postmenopausal women. The main side-effect of AIs is that they can increase the risk of osteoporosis, which is a significant health threat for older women. Other side-effects, such as joint pain and stiffness, incidence of carpal tunnel syndrome or dysregulated lipid metabolism, have also been reported for AIs, but these side-effects have a lower impact on the quality of life of patients than endometrial cancer and thromboembolic complications associated with TAM treatment. Acquired resistance to AIs has been observed after prolonged treatment, which also occurs in the case of SERMs. Crosstalks between estrogen receptor pathway and several signaling pathways such as PI3K/Akt/mTOR and Ras/Raf/MEK/MAPK could lead cancer cells to be resistant to AIs [112]. The combination of AIs and inhibitors of the related signaling pathways may be a promising strategy for AI-resistant patients.
Biological prevention
Recently, biological prevention, mainly known as the monoclonal antibodies for the breast cancer, has been developed to improve the quality of life in breast cancer patients. One of the major targets of these monoclonal antibodies is HER2. About 20-30% of all breast cancer cases exhibit HER2 protein overexpression or HER2 gene amplification [36]. Trastuzumab (Herceptin), a recombinant humanized monoclonal antibody, is the first HER2-targeted drug to be approved by the FDA. It can directly interact with the C-terminal portion of domain IV in the extracellular part of HER2 [113,114]. Up to now, the anti-tumor mechanism of trastuzumab has not been clearly elucidated. Some potential mechanisms may be that trastuzumab can suppress the growth and proliferation of cancer cells by recruiting ubiquitin to internalize and degrade HER2, by activating the immune system against cancer cells via a mechanism called antibody-dependent cell-mediated cytotoxicity (ADCC) or by inhibiting the MAPK and PI3K/Akt pathways [115-117]. Trastuzumab was initially used for treating metastatic breast cancer (MBC) and found to be efficacious as a single agent with an objective response rate (ORR) of 26%. In vitro experiments have shown that trastuzumab has a synergistic effect with other anti-tumor drugs such as nimotuzumab, carboplatin, 4-hydroxycyclophosphamide, docetaxel and vinorelbine [118,119]. The HERA and TRAIN trials demonstrated that chemotherapy combined with adjuvant trastuzumab for 1 year could improve the disease-free survival in patients with HER2+ breast cancer (HR=0.76) [120,121]. A randomized phase II trial executed by Marty also showed that trastuzumab plus docetaxel was more efficacious than docetaxel alone in treating HER2-positive MBC, with the ORR of 50% versus 32% [122]. However, side-effects such as congestive heart failure and left ventricular ejection fraction (LVEF) decline were found in trastuzumab-treated patients [123].
Similar to trastuzumab, pertuzumab (Perjeta), another humanized monoclonal antibody, can bind to the extracellular portion of HER2 like trastuzumab. However, the binding domain is different [124]. Pertuzumab combined with trastuzumab and docetaxel has been approved for treating HER2-positive breast cancer. The pathologic complete response (pCR) rate, as well as invasive-disease-free survival rate, significantly increased in HER+ tumors than those in HER- tumors (57.8% versus 22.0%) [125,126]. However, toxic side-effects like diarrhea and febrile neutropenia were common in pertuzumab-treated groups.
Recently, immunotherapy becomes a hot spot in cancer therapy, and it shows great potential in clinical use. Programmed cell death 1 (PD1) is a membrane protein expressed in various immune cells, including T cells, which can be engaged by its specific ligand to block the immune system. PD1 inhibitor drugs Nivolumab (Opdivo) and Pembrolizumab (Keytruda) were approved for the treatment of several solid tumors such as metastatic melanoma and non-small cell lung cancer. In the KEYNOTE-012 study, pembrolizumab was found to be effective in 27 TNBC patients with a clinical benefit rate of 20% [127]. Programmed cell death receptor ligand 1 (PDL1), a ligand of PD1, is detected in 20% of TNBC and in 50% of all breast cancers [128]. PDL1 inhibitor drug Atezolizumab (Tecentriq) exhibits a 19% of objective response rate in a phase Ⅰ study including 54 TNBC patients [129]. Though TNBC patients typically have poor clinical outcomes, anti-PD1/PDL1 drugs might be promising strategies for treating this subtype of breast cancer.
Conclusion and further directions
Breast cancer is the most frequently diagnosed cancer in women across 140 countries [1]. Approximately 1 in 8 women worldwide have a lifetime risk of developing breast cancer [130]. Breast cancer develops through a multistep process, and the pathogenesis of this disease has not yet been elucidated. In the last decade, the tumor microenvironment and breast CSCs have been identified as contributors to breast tumorigenesis. Breast cancer is also influenced by genetic and environmental factors. Targeted prevention strategies against these risk factors should be taken ahead of time.
Although the incidence rate of breast cancer is high in developed countries, the fact which we can't ignore is that almost half of the breast cancer cases and over half of deaths occur in developing countries. The 5-year relative survival rates of breast cancer varied widely in developed and developing countries. The rate is over 80% in North American and Japan, but below 40% in Africa countries like Algeria. Breast cancer is a preventable disease, and there are adequate medical resources available in developed countries, which can protect against this disease, such as annual mammography screening or the daily use of chemopreventative drugs. These may be attributable for the higher survival rate of breast cancer patients in developed countries than that in middle-income or low-income countries. Considering the financial burden of developing countries, the clinical breast examination is an effective way to diagnose breast cancer in the early stage. Moreover, if women are educated about breast cancer, breast self-examination may be a simple, economical and motivated method to prevent this disease. People know their own bodies more clearly than any doctors. However, most of the women in developing countries don't realize the importance of breast cancer prevention. Therefore, in these countries, more attention should be focused on breast health promotion ahead of clinical treatment.
Nowadays, with the reduction in the cost of DNA sequencing, individual genome sequencing may be affordable by middle-class populations, and this could be a new method in preventing breast cancer. If a woman have a family history of breast cancer, it is wise to do a screen especially on hereditary cancer susceptibility genes such as BRCA1 or BRCA2. The risk of breast cancer could then be evaluated based on the screening results and prevention advice could be offered personally. Individual genome sequencing may be a mainstream in the future for prevention of breast cancer as well as other hereditary disease. Additionally, risk factors should be taken more seriously either in normal or high-risk women. Environmental factors such as the exogenous estrogen intake, alcohol abuse and excess dietary fat consumption could be avoided to minimize breast cancer risk. Though some risk facts such as aging and reproductive factors are inevitable, measures should be taken ahead of time to reduce the risk. In the modern world, many people spend countless hours sitting at tables. People are more engaged in mental work rather than in physical work. However, physically active women have a 25% lower risk of breast cancer on average than women who are less active [131]. Regular physical exercise may be a convenient and inexpensive way to prevent breast cancer in women from both developed and developing countries.
Although traditional film mammography has limitations in detecting dense breasts, digital mammography can overcome this deficiency. It can capture images directly through an X-ray-sensitive detector and the digital data are analyzed in the computer. The Digital Mammographic Imaging Screening Trial (DMIST) showed that digital mammography had a better diagnostic performance than film mammography in pre- and perimenopausal women with dense breasts who were younger than 50 years of age. With advancements in digital technology, high-resolution digital mammography may replace film mammography in the future [132]. What's more, both mammography and MRI screening for a large population of women are expensive and only countries with good health insurance systems can offer these services. Direct breast ultrasonography, an adjuvant technique method to mammography and MRI, is less expensive and could be used widely in low- and middle-income countries. The ultrasonography is much more accurate if the operator is skilled and experienced.
Although great progress has been made in breast cancer prevention in the last decade, there is still a lack of effective therapies against TNBC. TNBC tends to have a higher relapse risk and is more aggressive than other subtypes, resulting in a poor 5-year survival rate [133]. Due to the absence of ER/PR expression and HER2 amplification, drugs targeted against these three receptors are useless in TNBC. In the last decade, several potential biomarkers in TNBC such as EGFR, androgen receptor (AR), PARP and mTOR, and microRNA-based biomarkers, such as miR-374b-5p and miR-629-3p have been identified and explored for targeted therapies [134-136]. The EGFR-inhibitor cetuximab combined with cisplatin increased the progression-free survival from 1.5 to 3.7 months, and the overall survival from 9.4 to 12.9 months in a phase Ⅱ clinical trial [137]. Expressions of AR is observed in 30% of TNBC patients, and the AR inhibitor bicalutaminde showed a clinical benefit rate of 19% in ER/PR negative breast cancer patients in a phase Ⅱ study [138,139]. PARP-inhibitor iniparib plus chemotherapy were also tested and showed promising results in phase Ⅰ and Ⅱ clinical trials. However, the phase III clinical trial failed with the lack of improvement in progression-free survival and overall survival [140]. Targeting the PI3K/AKT/mTOR pathway was thought to be an effective strategy to treat TNBC recently, and the mTOR inhibitor everolimus combined with doxorubicin and bevacizumab increased the objective response rate but not the clinical benefit rate [141]. Although many biomarker-based trials have been performed in TNBC, none has been successful finally. One of the main reasons for this failure could be the heterogeneity of TNBC. More work is needed to elucidate tumor heterogeneity, and the discovery of a robust biomarker regardless of tumor heterogeneity may be a breakthrough in TNBC treatment. Immunotherapy agents such as anti-PD1/PDL1 drugs will also shed light on treating TNBC.
In summary, breast cancer is preventable. Reducing risk factors and taking chemoprevention are two main measures to prevent breast cancer. However, there's a long way to go in creating public breast cancer awareness. Only 4.1% of high-risk women are willing to take chemoprevention drugs [142]. The fear of adverse effects and lack of understanding of breast cancer might be attributable for this unwillingness. Although, the Gail model or the IBIS model is widely used for determining the risk of breast cancer based on a woman's age, family history, race and reproductive factors, we still lack a reliable strategy to exactly evaluate the risk ratio of breast cancer. With improvements in sequencing technology, individual genome sequencing may be a powerful method to evaluate the risk of breast cancer. Better medicines with less adverse effects and a favorable risk-benefit ratio need to be developed in the future.
Acknowledgements
We would like to thank Dr. Yan-Ling Wu for her advice on this paper. This work is supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ15C040001 and LY18H190003, Social Development Project of Public Welfare Technology Research in Zhejiang Province (LGF18H100002), and Zhejiang Provincial Foundation for Scientific Research in Medicine and Health (2015RCB009).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Stewart BW, Wild CP. World Cancer Report 2014. Geneva, Switzerland: WHO Press. 2014
2. WHO: Geneva, Switzerland. Breast cancer. http://www.who.int/cancer/prevention/diagnosis-screening/breast-cancer/en/
3. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7-30
4. DeSantis CE, Fedewa SA, Goding Sauer A. et al. Breast cancer statistics, 2015: Convergence of incidence rates between black and white women. CA Cancer J Clin. 2016;66:31-42
5. Drukteinis JS, Mooney BP, Flowers CI. et al. Beyond mammography: new frontiers in breast cancer screening. Am J Med. 2013;126:472-479
6. Majeed W, Aslam B, Javed I. et al. Breast cancer: major risk factors and recent developments in treatment. APJCP. 2014;15:3353-3358
7. Maffini MV, Soto AM, Calabro JM. et al. The stroma as a crucial target in rat mammary gland carcinogenesis. J Cell Sci. 2004;117:1495-1502
8. Sonnenschein C, Soto AM. Carcinogenesis explained within the context of a theory of organisms. Progress in biophysics and molecular biology. 2016;122:70-76
9. Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010;141:39-51
10. Dumars C, Ngyuen JM, Gaultier A. et al. Dysregulation of macrophage polarization is associated with the metastatic process in osteosarcoma. Oncotarget. 2016;7:78343-78354
11. Polyak K. Breast cancer: origins and evolution. J Clin Invest. 2007;117:3155-3163
12. Basse C, Arock M. The increasing roles of epigenetics in breast cancer: Implications for pathogenicity, biomarkers, prevention and treatment. Int J Cancer. 2015;137:2785-2794
13. Baumann M, Krause M, Hill R. Exploring the role of cancer stem cells in radioresistance. Nat Rev Cancer. 2008;8:545-554
14. Smalley M, Piggott L, Clarkson R. Breast cancer stem cells: obstacles to therapy. Cancer Lett. 2013;338:57-62
15. Zhang M, Lee AV, Rosen JM. The Cellular Origin and Evolution of Breast Cancer. Cold Spring Harbor perspectives in medicine. 2017;7:a027128
16. Al-Hajj M, Wicha MS, Benito-Hernandez A. et al. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983-3988
17. Molyneux G, Geyer FC, Magnay FA. et al. BRCA1 basal-like breast cancers originate from luminal epithelial progenitors and not from basal stem cells. Cell Stem Cell. 2010;7:403-417
18. Valenti G, Quinn HM, Heynen G. et al. Cancer Stem Cells Regulate Cancer-Associated Fibroblasts via Activation of Hedgehog Signaling in Mammary Gland Tumors. Cancer Res. 2017;77:2134-2147
19. El Helou R, Pinna G, Cabaud O. et al. miR-600 Acts as a Bimodal Switch that Regulates Breast Cancer Stem Cell Fate through WNT Signaling. Cell reports. 2017;18:2256-2268
20. Shukla G, Khera HK, Srivastava AK. et al. Therapeutic Potential, Challenges and Future Perspective of Cancer Stem Cells in Translational Oncology: A Critical Review. Current stem cell research & therapy. 2017;12:207-224
21. Kasper M, Jaks V, Fiaschi M. et al. Hedgehog signalling in breast cancer. Carcinogenesis. 2009;30:903-911
22. Sgroi DC. Preinvasive breast cancer. Annu Rev Pathol. 2010;5:193-221
23. Deng CX. BRCA1: cell cycle checkpoint, genetic instability, DNA damage response and cancer evolution. Nucleic Acids Res. 2006;34:1416-1426
24. Dine J, Deng CX. Mouse models of BRCA1 and their application to breast cancer research. Cancer Metastasis Rev. 2013;32:25-37
25. Tan-Wong SM, French JD, Proudfoo NJ. et al. Dynamic interactions between the promoter and terminator regions of the mammalian BRCA1 gene. P Natl Acad Sci USA. 2008;105:5160-5165
26. Hegan DC, Lu Y, Stachelek GC. et al. Inhibition of poly(ADP-ribose) polymerase down-regulates BRCA1 and RAD51 in a pathway mediated by E2F4 and p130. Proc Natl Acad Sci U S A. 2010;107:2201-2206
27. Sanchez H, Paul MW, Grosbart M. et al. Architectural plasticity of human BRCA2-RAD51 complexes in DNA break repair. Nucleic Acids Res. 2017;45:4507-4518
28. Martinez JS, von Nicolai C, Kim T. et al. BRCA2 regulates DMC1-mediated recombination through the BRC repeats. Proc Natl Acad Sci U S A. 2016;113:3515-3520
29. Bane AL, Beck JC, Bleiweiss I. et al. BRCA2 mutation-associated breast cancers exhibit a distinguishing phenotype based on morphology and molecular profiles from tissue microarrays. Am J Surg Pathol. 2007;31:121-128
30. Balmana J, Diez O, Rubio IT. et al. BRCA in breast cancer: ESMO Clinical Practice Guidelines. Ann Oncol. 2011;22(Suppl 6):31-34
31. Paluch-Shimon S, Cardoso F, Sessa C. et al. Prevention and screening in BRCA mutation carriers and other breast/ovarian hereditary cancer syndromes: ESMO Clinical Practice Guidelines for cancer prevention and screening. Ann Oncol. 2016;27:103-110
32. Chen S, Parmigiani G. Meta-analysis of BRCA1 and BRCA2 penetrance. J Clin Oncol. 2007;25:1329-1333
33. Shih C, Padhy LC, Murray M. et al. Transforming genes of carcinomas and neuroblastomas introduced into mouse fibroblasts. Nature. 1981;290:261-264
34. Harbeck N, Gnant M. Breast cancer. The Lancet. 2017;389:1134-1150
35. Davis NM, Sokolosky M, Stadelman K. et al. Deregulation of the EGFR/PI3K/PTEN/Akt/mTORC1 pathway in breast cancer: possibilities for therapeutic intervention. Oncotarget. 2014;5:4603-4650
36. Elizalde PV, Cordo Russo RI, Chervo MF. et al. ErbB-2 nuclear function in breast cancer growth, metastasis and resistance to therapy. Endocr Relat Cancer. 2016;23:243-257
37. Appert-Collin A, Hubert P, Cremel G. et al. Role of ErbB Receptors in Cancer Cell Migration and Invasion. Frontiers in pharmacology. 2015;6:283
38. Ali R, Wendt MK. The paradoxical functions of EGFR during breast cancer progression. Signal transduction and targeted therapy. 2017;2:16042
39. Alanazi IO, Khan Z. Understanding EGFR Signaling in Breast Cancer and Breast Cancer Stem Cells: Overexpression and Therapeutic Implications. Asian Pacific journal of cancer prevention: APJCP. 2016;17:445-453
40. Zhang D, LaFortune TA, Krishnamurthy S. et al. Epidermal growth factor receptor tyrosine kinase inhibitor reverses mesenchymal to epithelial phenotype and inhibits metastasis in inflammatory breast cancer. Clin Cancer Res. 2009;15:6639-6648
41. Kim A, Jang MH, Lee SJ. et al. Mutations of the Epidermal Growth Factor Receptor Gene in Triple-Negative Breast Cancer. Journal of breast cancer. 2017;20:150-159
42. Green AR, Aleskandarany MA, Agarwal D. et al. MYC functions are specific in biological subtypes of breast cancer and confers resistance to endocrine therapy in luminal tumours. Br J Cancer. 2016;114:917-928
43. Poole CJ, van Riggelen J. MYC-Master Regulator of the Cancer Epigenome and Transcriptome. Genes. 2017;8:142
44. Chen Y, Olopade OI. MYC in breast tumor progression. Expert Rev Anticancer Ther. 2008;8:1689-1698
45. Jung M, Russell AJ, Liu B. et al. A Myc Activity Signature Predicts Poor Clinical Outcomes in Myc-Associated Cancers. Cancer Res. 2017;77:971-981
46. Pylayeva-Gupta Y, Grabocka E, Bar-Sagi D. RAS oncogenes: weaving a tumorigenic web. Nat Rev Cancer. 2011;11:761-774
47. Siewertsz van Reesema LL, Lee MP, Zheleva V. et al. RAS pathway biomarkers for breast cancer prognosis. Clinical laboratory international. 2016;40:18-23
48. Hoenerhoff MJ, Chu I, Barkan D. et al. BMI1 cooperates with H-RAS to induce an aggressive breast cancer phenotype with brain metastases. Oncogene. 2009;28:3022-3032
49. Fernandez-Medarde A, Santos E. Ras in cancer and developmental diseases. Genes & cancer. 2011;2:344-358
50. Ray A, Ray BK. Induction of Ras by SAF-1/MAZ through a feed-forward loop promotes angiogenesis in breast cancer. Cancer medicine. 2015;4:224-234
51. Varna M, Bousquet G, Plassa LF. et al. TP53 status and response to treatment in breast cancers. J Biomed Biotechnol. 2011;2011:284584
52. Hientz K, Mohr A, Bhakta-Guha D. et al. The role of p53 in cancer drug resistance and targeted chemotherapy. Oncotarget. 2017;8:8921-8946
53. Roberts MR, Sucheston-Campbell LE, Zirpoli GR. et al. Single nucleotide variants in metastasis-related genes are associated with breast cancer risk, by lymph node involvement and estrogen receptor status, in women with European and African ancestry. Molecular carcinogenesis. 2017;56:1000-1009
54. Qu S, Long J, Cai Q. et al. Genetic polymorphisms of metastasis suppressor gene NME1 and breast cancer survival. Clin Cancer Res. 2008;14:4787-4793
55. Cheng L, Zhou Z, Flesken-Nikitin A. et al. Rb inactivation accelerates neoplastic growth and substitutes for recurrent amplification of cIAP1, cIAP2 and Yap1 in sporadic mammary carcinoma associated with p53 deficiency. Oncogene. 2010;29:5700-5711
56. Lefebvre C, Bachelot T, Filleron T. et al. Mutational Profile of Metastatic Breast Cancers: A Retrospective Analysis. PLoS medicine. 2016;13:e1002201
57. Loibl S, Darb-Esfahani S, Huober J. et al. Integrated Analysis of PTEN and p4EBP1 Protein Expression as Predictors for pCR in HER2-Positive Breast Cancer. Clin Cancer Res. 2016;22:2675-2683
58. Hernandez-Aya LF, Gonzalez-Angulo AM. Targeting the phosphatidylinositol 3-kinase signaling pathway in breast cancer. The oncologist. 2011;16:404-414
59. Choi M, Kipps T, Kurzrock R. ATM Mutations in Cancer: Therapeutic Implications. Molecular cancer therapeutics. 2016;15:1781-1791
60. Desmedt C, Zoppoli G, Gundem G. et al. Genomic Characterization of Primary Invasive Lobular Breast Cancer. J Clin Oncol. 2016;34:1872-1881
61. Su Y, Wang X, Li J. et al. The clinicopathological significance and drug target potential of FHIT in breast cancer, a meta-analysis and literature review. Drug design, development and therapy. 2015;9:5439-5445
62. Berardi R, Morgese F, Onofri A. et al. Role of maspin in cancer. Clinical and translational medicine. 2013;2:8
63. Dabiri S, Moeini Aghtaei M, Shahryari J. et al. Maspin Gene Expression in Invasive Ductal Carcinoma of Breast. Iranian journal of pathology. 2016;11:104-111
64. Inoue K, Fry EA. Aberrant expression of cyclin D1 in cancer. Signal transduction insights. 2015;4:1-13
65. Brewer HR, Jones ME, Schoemaker MJ. et al. Family history and risk of breast cancer: an analysis accounting for family structure. Breast Cancer Res Treat. 2017;165:193-200
66. Washbrook E. Risk factors and epidemiology of breast cancer. Women's Health Medicine. 2006;3:8-14
67. Horn J, Vatten LJ. Reproductive and hormonal risk factors of breast cancer: a historical perspective. International journal of women's health. 2017;9:265-272
68. Dall GV, Britt KL. Estrogen Effects on the Mammary Gland in Early and Late Life and Breast Cancer Risk. Front Oncol. 2017;7:110
69. Horn J, Asvold BO, Opdahl S. et al. Reproductive factors and the risk of breast cancer in old age: a Norwegian cohort study. Breast Cancer Res Treat. 2013;139:237-243
70. Rosato V, Bosetti C, Negri E. et al. Reproductive and hormonal factors, family history, and breast cancer according to the hormonal receptor status. Eur J Cancer Prev. 2014;23:412-417
71. Endogenous H, Breast Cancer Collaborative G, Key TJ. et al. Sex hormones and risk of breast cancer in premenopausal women: a collaborative reanalysis of individual participant data from seven prospective studies. Lancet Oncol. 2013;14:1009-1019
72. Soroush A, Farshchian N, Komasi S. et al. The Role of Oral Contraceptive Pills on Increased Risk of Breast Cancer in Iranian Populations: A Meta-analysis. Journal of cancer prevention. 2016;21:294-301
73. Bethea TN, Rosenberg L, Hong CC. et al. A case-control analysis of oral contraceptive use and breast cancer subtypes in the African American Breast Cancer Epidemiology and Risk Consortium. Breast Cancer Res. 2015;17:22
74. Beral V. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003;362:419-427
75. Liu J-Y, Chen T-J, Hwang S-J. The Risk of Breast Cancer in Women Using Menopausal Hormone Replacement Therapy in Taiwan. International journal of environmental research and public health. 2016;13:482
76. Narod SA. Hormone replacement therapy and the risk of breast cancer. Nature reviews. Clinical oncology. 2011;8:669-676
77. Fahlen M, Fornander T, Johansson H. et al. Hormone replacement therapy after breast cancer: 10 year follow up of the Stockholm randomised trial. Eur J Cancer. 2013;49:52-59
78. Ravdin PM, Cronin KA, Howlader N. et al. The decrease in breast-cancer incidence in 2003 in the United States. N Engl J Med. 2007;356:1670-1674
79. Hamajima N, Hirose K, Tajima K. et al. Alcohol, tobacco and breast cancer-collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. Br J Cancer. 2002;87:1234-1245
80. Jung S, Wang M, Anderson K. et al. Alcohol consumption and breast cancer risk by estrogen receptor status: in a pooled analysis of 20 studies. International journal of epidemiology. 2016;45:916-928
81. Makarem N, Chandran U, Bandera EV. et al. Dietary fat in breast cancer survival. Annu Rev Nutr. 2013;33:319-348
82. Knight JA, Fan J, Malone KE. et al. Alcohol consumption and cigarette smoking in combination: A predictor of contralateral breast cancer risk in the WECARE study. Int J Cancer. 2017;141:916-924
83. Catsburg C, Miller AB, Rohan TE. Active cigarette smoking and risk of breast cancer. Int J Cancer. 2015;136:2204-2209
84. Gaudet MM, Carter BD, Brinton LA. et al. Pooled analysis of active cigarette smoking and invasive breast cancer risk in 14 cohort studies. International journal of epidemiology. 2017;46:881-893
85. McKenzie F, Ellison-Loschmann L, Jeffreys M. et al. Cigarette smoking and risk of breast cancer in a New Zealand multi-ethnic case-control study. PLoS One. 2013;8:e63132
86. Kispert S, McHowat J. Recent insights into cigarette smoking as a lifestyle risk factor for breast cancer. Breast Cancer: Targets and Therapy. 2017;9:127-132
87. Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147:275-292
88. Prevention CfDCa. Cancer screening - United States, 2010. Morb Mortal Wkly Rep. 2012;61:41-45
89. The benefits and harms of breast cancer screening. an independent review. Lancet. 2012;380:1778-1786
90. van den Ende C, Oordt-Speets AM, Vroling H. et al. Benefits and harms of breast cancer screening with mammography in women aged 40-49 years: A systematic review. Int J Cancer. 2017;141:1295-1306
91. Greenwood HI, Heller SL, Kim S. et al. Ductal Carcinoma in Situ of the Breasts: Review of MR Imaging Features. Radiographics. 2013;33:1569-1588
92. Morrow M, Waters J, Morris E. MRI for breast cancer screening, diagnosis, and treatment. Lancet. 2011;378:1804-1811
93. Enriquez L, Listinsky J. Role of MRI in breast cancer management. Cleve Clin J Med. 2009;76:525-532
94. Saslow D, Boetes C, Burke W. et al. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA Cancer J Clin. 2007;57:75-89
95. Sporn MB. Approaches to prevention of epithelial cancer during the preneoplastic period. Cancer Res. 1976;36:2699-2702
96. Bozovic-Spasojevic I, Azambuja E, McCaskill-Stevens W. et al. Chemoprevention for breast cancer. Cancer Treat Rev. 2012;38:329-339
97. Nagini S. Breast Cancer: Current Molecular Therapeutic Targets and New Players. Anti-cancer agents in medicinal chemistry. 2017;17:152-163
98. Cuzick J, Powles T, Veronesi U. et al. Overview of the main outcomes in breast-cancer prevention trials. Lancet. 2003;361:296-300
99. Cuzick J, Sestak I, Bonanni B. et al. Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data. Lancet. 2013;381:1827-1834
100. Yang Y, Pan W, Tang X. et al. A meta-analysis of randomized controlled trials comparing the efficacy and safety of anastrozole versus tamoxifen for breast cancer. Oncotarget. 2017;18:48362-48374
101. Barrett-Connor E, Mosca L, Collins P. et al. Effects of raloxifene on cardiovascular events and breast cancer in postmenopausal women. N Engl J Med. 2006;355:125-137
102. Coronado Martin PJ, Calaf Alsina J. Third generation selective estrogen receptor modulators benefits beyond bone: effects on breast. Med Clin (Barc). 2013;140:217-222
103. Hiscox S, Davies EL, Barrett-Lee P. Aromatase inhibitors in breast cancer. Maturitas. 2009;63:275-279
104. Dowsett M, Cuzick J, Ingle J. et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28:509-518
105. Goss PE, Ingle JN, Ales-Martinez JE. et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364:2381-2391
106. Boccardo F, Guglielmini P, Bordonaro R. et al. Switching to anastrozole versus continued tamoxifen treatment of early breast cancer: long term results of the Italian Tamoxifen Anastrozole trial. Eur J Cancer. 2013;49:1546-1554
107. Bliss JM, Kilburn LS, Coleman RE. et al. Disease-related outcomes with long-term follow-up: an updated analysis of the intergroup exemestane study. J Clin Oncol. 2012;30:709-717
108. Dubsky PC, Jakesz R, Mlineritsch B. et al. Tamoxifen and anastrozole as a sequencing strategy: a randomized controlled trial in postmenopausal patients with endocrine-responsive early breast cancer from the Austrian Breast and Colorectal Cancer Study Group. J Clin Oncol. 2012;30:722-728
109. Regan MM, Neven P, Giobbie-Hurder A. et al. Assessment of letrozole and tamoxifen alone and in sequence for postmenopausal women with steroid hormone receptor-positive breast cancer: the BIG 1-98 randomised clinical trial at 8.1 years median follow-up. Lancet Oncol. 2011;12:1101-1108
110. Forbes JF, Cuzick J, Buzdar A. et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the ATAC trial. Lancet Oncol. 2008;9:45-53
111. Cuzick J, Sestak I, Baum M. et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol. 2010;11:1135-1141
112. Lonning PE, Eikesdal HP. Aromatase inhibition 2013: clinical state of the art and questions that remain to be solved. Endocr Relat Cancer. 2013;20:R183-201
113. Cho HS, Mason K, Ramyar KX. et al. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature. 2003;421:756-760
114. Nielsen DL, Andersson M, Kamby C. HER2-targeted therapy in breast cancer. Monoclonal antibodies and tyrosine kinase inhibitors. Cancer Treat Rev. 2009;35:121-136
115. Junttila TT, Akita RW, Parsons K. et al. Ligand-independent HER2/HER3/PI3K complex is disrupted by trastuzumab and is effectively inhibited by the PI3K inhibitor GDC-0941. Cancer Cell. 2009;15:429-440
116. Vu T, Claret FX. Trastuzumab: updated mechanisms of action and resistance in breast cancer. Front Oncol. 2012;2:62
117. Arnould L, Gelly M, Penault-Llorca F. et al. Trastuzumab-based treatment of HER2-positive breast cancer: an antibody-dependent cellular cytotoxicity mechanism? Br J Cancer. 2006;94:259-267
118. Vogel CL, Cobleigh MA, Tripathy D. et al. Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. Journal of Clinical Oncology. 2002;20:719-726
119. Yang Y, Guo R, Tian X. et al. Synergistic anti-tumor activity of Nimotuzumab in combination with Trastuzumab in HER2-positive breast cancer. Biochemical and biophysical research communications. 2017;489:523-527
120. Cameron D, Piccart-Gebhart MJ, Gelber RD. et al. 11 years' follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive early breast cancer: final analysis of the HERceptin Adjuvant (HERA) trial. Lancet. 2017;389:1195-1205
121. van Ramshorst MS, van Werkhoven E, Mandjes IAM. et al. Trastuzumab in combination with weekly paclitaxel and carboplatin as neo-adjuvant treatment for HER2-positive breast cancer: The TRAIN-study. Eur J Cancer. 2017;74:47-54
122. Marty M, Cognetti F, Maraninchi D. et al. Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. Journal of Clinical Oncology. 2005;23:4265-4274
123. Balduzzi S, Mantarro S, Guarneri V. et al. Trastuzumab-containing regimens for metastatic breast cancer. Cochrane Database Syst Rev. 2014;12:CD006242
124. Franklin MC, Carey KD, Vajdos FF. et al. Insights into ErbB signaling from the structure of the ErbB2-pertuzumab complex. Cancer Cell. 2004;5:317-328
125. Loibl S, Jackisch C, Schneeweiss A. et al. Dual HER2-blockade with pertuzumab and trastuzumab in HER2-positive early breast cancer: a subanalysis of data from the randomized phase III GeparSepto trial. Ann Oncol. 2017;28:497-504
126. von Minckwitz G, Procter M, de Azambuja E. et al. Adjuvant Pertuzumab and Trastuzumab in Early HER2-Positive Breast Cancer. N Engl J Med. 2017;377:122-131
127. Hu X, Huang W, Fan M. Emerging therapies for breast cancer. Journal of hematology & oncology. 2017;10:98
128. Sabatier R, Finetti P, Mamessier E. et al. Prognostic and predictive value of PDL1 expression in breast cancer. Oncotarget. 2015;6:5449-5464
129. Gibson J. Anti-PD-L1 for metastatic triple-negative breast cancer. Lancet Oncol. 2015;16:e264
130. Rojas K, Stuckey A. Breast Cancer Epidemiology and Risk Factors. Clinical obstetrics and gynecology. 2016;59:651-672
131. Lynch BM, Neilson HK, Friedenreich CM. Physical activity and breast cancer prevention. Recent Results Cancer Res. 2011;186:13-42
132. Pisano ED, Hendrick RE, Yaffe MJ. et al. Diagnostic accuracy of digital versus film mammography: exploratory analysis of selected population subgroups in DMIST. Radiology. 2008;246:376-383
133. Aysola K, Desai A, Welch C. et al. Triple Negative Breast Cancer - An Overview. Hereditary genetics: current research. 2013;2013(Suppl 2):001
134. Wang J, Song C, Tang H. et al. miR-629-3p may serve as a novel biomarker and potential therapeutic target for lung metastases of triple-negative breast cancer. Breast Cancer Res. 2017;19:72
135. Mathe A, Scott RJ, Avery-Kiejda KA. MiRNAs and Other Epigenetic Changes as Biomarkers in Triple Negative Breast Cancer. International journal of molecular sciences. 2015;16:28347-28376
136. Massihnia D, Galvano A, Fanale D. et al. Triple negative breast cancer: shedding light onto the role of pi3k/akt/mtor pathway. Oncotarget. 2016;7:60712-60722
137. Baselga J, Gomez P, Greil R. et al. Randomized phase II study of the anti-epidermal growth factor receptor monoclonal antibody cetuximab with cisplatin versus cisplatin alone in patients with metastatic triple-negative breast cancer. J Clin Oncol. 2013;31:2586-2592
138. Anestis A, Karamouzis MV, Dalagiorgou G. et al. Is androgen receptor targeting an emerging treatment strategy for triple negative breast cancer? Cancer Treatment Reviews. 2015;41:547-553
139. Gucalp A, Tolaney S, Isakoff SJ. et al. Phase II trial of bicalutamide in patients with androgen receptor-positive, estrogen receptor-negative metastatic Breast Cancer. Clin Cancer Res. 2013;19:5505-5512
140. O'Shaughnessy J, Schwartzberg L, Danso MA. et al. Phase III study of iniparib plus gemcitabine and carboplatin versus gemcitabine and carboplatin in patients with metastatic triple-negative breast cancer. J Clin Oncol. 2014;32:3840-3847
141. Basho RK, Gilcrease M, Murthy RK. et al. Targeting the PI3K/AKT/mTOR Pathway for the Treatment of Mesenchymal Triple-Negative Breast Cancer: Evidence From a Phase 1 Trial of mTOR Inhibition in Combination With Liposomal Doxorubicin and Bevacizumab. JAMA oncology. 2017;3:509-515
142. Ropka ME, Keim J, Philbrick JT. Patient decisions about breast cancer chemoprevention: a systematic review and meta-analysis. J Clin Oncol. 2010;28:3090-3095
Author contact
Corresponding authors: Ping-ping Yao, Key Lab of Vaccine against Hemorrhagic Fever with Renal Syndrome, Zhejiang Provincial Center for Disease Control and Prevention, Hangzhou, China. Phone: 86-571-87115312; Fax: 86-571-87115314; E-mail: ppyaozj.cn. Han-Ping Zhu, Key Lab of Vaccine against Hemorrhagic Fever with Renal Syndrome, Zhejiang Provincial Center for Disease Control and Prevention, Hangzhou, China. Phone: 86-571-87115313; Fax: 86-571-87115316; E-mail: hpzhuzj.cn.

 Global reach, higher impact
Global reach, higher impact