Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(8):1685-1695. doi:10.7150/ijbs.35419 This issue Cite
Research Paper
CXXC5 Mediates P. gingivalis-suppressed Cementoblast Functions Partially via MAPK Signaling Network
1. The State Key Laboratory Breeding Base of Basic Science of Stomatology (Hubei-MOST KLOS) & Key Laboratory of Oral Biomedicine Ministry of Education (KLOBME), School & Hospital of Stomatology, Wuhan University, Wuhan, China
2. Department of Periodontology, School & Hospital of Stomatology, Wuhan University, Wuhan, China
Abstract
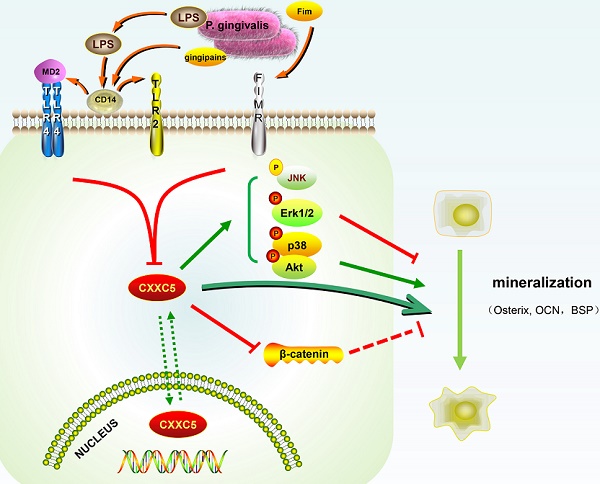
Porphyromonas (P.) gingivalis associates tightly with periodontal diseases and it is also a dominant pathogen of periapical periodontitis. However, the influence of P. gingivalis on cementoblasts, root surface cells pivotal in the apical areas, and the possible involvement of other molecules remain largely elusive. CXXC5 is a nuclear protein that regulates gene expression as well as cell growth, differentiation, and apoptosis. In this study, P. gingivalis repressed the mineralization capacity of cementoblasts by inducing inflammatory reactions and inhibiting cell differentiation. Intriguingly, the expression of CXXC5 decreased in P. gingivalis-treated OCCM-30 cells and apical periodontitis models but gradually increased during mineralization. Furthermore, RNA interference of CXXC5 significantly inhibited cementoblast differentiation, represented by decline of bone-associated markers Osterix, osteocalcin (OCN), and alkaline phosphatase (ALP). CXXC5 overexpression facilitated differentiation, and therefore attenuated the P. gingivalis-repressed effects on OCCM-30 cells. In addition, Erk1/2, p38, and PI3K-Akt were inactivated by silencing CXXC5 and activated upon its overexpression, whereas Wnt/β-catenin exhibited an opposite trend. The employment of specific inhibitors revealed that the CXXC5-dependent promotions of cementoblast differentiation were partially abrogated by p38 and PI3K-Akt inhibitors but were exacerbated by inhibiting Erk1/2. Overall, our experiment demonstrated a novel function of CXXC5 in the regeneration of impaired cementum caused by P. gingivalis invasion and suggested that MAPK signaling network balances the facilitation effects of CXXC5 in cementoblast differentiation.
Keywords: Porphyromonas gingivalis, cementum, CXXC5, periodontitis, apical periodontitis, signaling regulation

 Global reach, higher impact
Global reach, higher impact