10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(8):1712-1722. doi:10.7150/ijbs.33986 This issue Cite
Research Paper
Serum microRNA Signature Is Capable of Early Diagnosis for Non-Small Cell Lung Cancer
1. Department of Respiratory and Critical Care Medicine, The Second Affiliated Hospital of Xian Jiaotong University, Xi'an, Shaanxi, China;
2. Affiliated Hospital of Guangdong Medical University, Zhanjiang, China;
3. Section of Thoracic Surgery, Department of Surgery, University of Michigan, Ann Arbor, MI, USA;
4. School of Medicine, Southern University of Science and Technology, Shenzhen, China.
Abstract
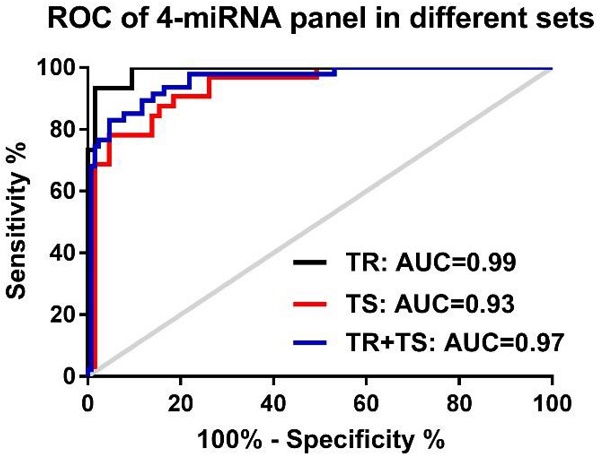
Despite decades of efforts, non-small-cell lung cancer (NSCLC) remains the leading cause of cancer mortality globally primarily due to the challenge in early detection of the cancer. Being an important player in cancer development, the dysregulated miRNAs have been shown promising values as non-invasive diagnostic and prognostic biomarkers for NSCLC. The aim of our study is to access the efficacy and reliability of a potential circulating miRNA panel in early diagnosis of NSCLC. We first selected eight candidate miRNAs, miR-146b, miR-205, miR-29c, miR-31, miR-30b, miR-337, miR-411, and miR-708, which have been shown frequently aberrant in primary NSCLC patients based on our previous studies and other reports. The serum level of each of these miRNAs was evaluated by quantitative real-time PCR (qRT-PCR) in training and testing sets. We found that 5 out of 8 miRNAs (miR-146b, miR-205, miR-29c, miR-30b, and miR-337) were significantly up-regulated in NSCLCs patients compared to healthy or cancer-free controls in both training and testing sets. Based on the logistic regression model, a 4-miRNAs set (miR-146b, miR-205, miR-29c and miR-30b) was picked out of the 5 miRNAs owing to its excellent diagnostic power for NSCLC patients in the training set (AUC=0.99, accuracy=95.00%), the testing set (AUC=0.93, accuracy=89.69%), and the training-testing combined set ( AUC=0.96, accuracy=92.00%). When pathological subtypes of NSCLC are compared, this 4-miRNA panel carried a relatively higher prediction power and higher sensitivity for adenocarcinoma (AC) (AUC=0.98, sensitivity=99.10%) than for squamous cell carcinoma (SCC) (AUC=0.93, sensitivity=90.32%). Additionally, this panel demonstrated a comparable diagnostic capacity for stage I (AUC=0.96) and stage II-III (AUC=0.95) of NSCLC, suggesting its role in reflecting the tumor load. Importantly, the high levels of miR-146b and miR-29c in serum were significantly associated with poor 5-year overall survival (OS) (both p=0.04). Further survival analysis showed that high level of miR-146b in serum is specifically correlated with poor survival rate in SCC patients (p=0.0035) but not in AC patients (p=0.83), consistent with our previous finding that the high tissue expression of miR-146b in lung cancer specimen is indicative of a poor prognosis for SCC patients. Altogether, our study demonstrated that the 4-miRNA panel is a novel, sensitive and non-invasive serum marker for the early diagnosis of NSCLC.
Keywords: non-small-cell lung cancer (NSCLC), miRNA, serum biomarker, early diagnosis, prognosis

 Global reach, higher impact
Global reach, higher impact