Impact Factor ISSN: 1449-2288
Int J Biol Sci 2019; 15(12):2607-2614. doi:10.7150/ijbs.32889 This issue Cite
Research Paper
Osimertinib and pterostilbene in EGFR-mutation-positive non-small cell lung cancer (NSCLC)
1. Pangaea Oncology, Laboratory of Molecular Biology, Quirón-Dexeus University Institute, Barcelona, Spain
2. Instituto Oncológico Dr Rosell (IOR), University Hospital Sagrat Cor, QuironSalud Group, Barcelona, Spain
3. Institut d'Investigació en Ciències Germans Trias i Pujol, Badalona, Spain
4. Institut Català d'Oncologia, Hospital Germans Trias i Pujol, Badalona, Spain
Abstract
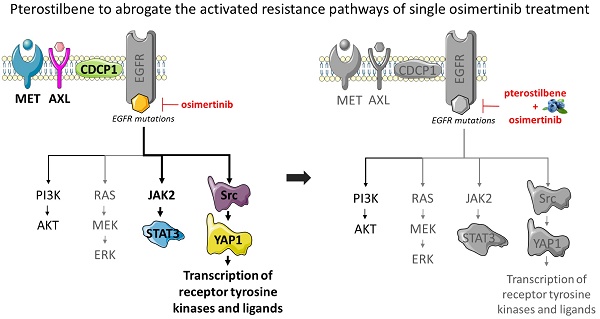
Monotherapy with epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) still leads to incomplete responses in most EGFR-mutation positive non-small cell lung cancer (NSCLC) patients, often due to acquired resistance through activation of parallel compensatory pathways. We have previously shown that co-targeting EGFR, signal transducer and activator of transcription 3 (STAT3), and Src-yes-associated protein 1 (YAP1) was highly synergistic in vitro and in vivo. In the present study, we treated EGFR-mutation positive cell lines with the combination of osimertinib plus a natural compound, pterostilbene, which has been reported to abrogate Src and STAT3 activation.
Methods: Cell viability assays and immunoblotting were performed to reveal the mechanisms of action of pterostilbene, osimertinib and pterostilbene plus osimertinib in five EGFR-mutation positive NSCLC and one triple negative breast cancer (TNBC) cell lines.
Results: Osimertinib plus pterostilbene yielded synergistic effects in all EGFR-mutation positive NSCLC cell lines investigated. Surprisingly, pterostilbene alone did not inhibit, nor downregulate Src phosphorylation in the EGFR-mutation positive NSCLC cell lines or the TNBC cell line, MDA-MB-231. However, the double combination of osimertinib plus pterostilbene reversed the osimertinib-induced STAT3, YAP1, and CUB domain-containing protein-1 (CDCP1) phosphorylation and slightly suppressed Src phosphorylation in PC9 and H1975 cells.
Conclusion: The results of this study indicate that pterostilbene may be used to abrogate the activated resistance pathways of single osimertinib treatment in EGFR-mutation positive NSCLC. Future studies should focus on in vivo translation and confirmation of these results.
Keywords: Pterostilbene, NSCLC, osimertinib, therapy resistance

 Global reach, higher impact
Global reach, higher impact