Impact Factor ISSN: 1449-2288
Int J Biol Sci 2019; 15(12):2719-2732. doi:10.7150/ijbs.35930 This issue Cite
Research Paper
Optimization Strategy for Generating Gene-edited Tibet Minipigs by Synchronized Oestrus and Cytoplasmic Microinjection
1. School of Basic Medical Sciences, Southern Medical University, Guangzhou 510515, China
2. Institute of Comparative Medicine & Laboratory Animal Center, Southern Medical University, Guangzhou 510515, China
3. Songshan Lake Pearl Laboratory Animal Sci. & Tech. Co., Ltd., Dongguan 523808, China
4. Guangdong Provincial Key Laboratory of Cancer Immunotherapy Research and Guangzhou Key Laboratory of Tumor Immunology Research, Cancer Research Institute, Southern Medical University, Guangzhou 510515, China
5. School of Biotechnology and Health Sciences, Wuyi University, Jiangmen 529020, China
* These authors contributed equally to this work.
Abstract
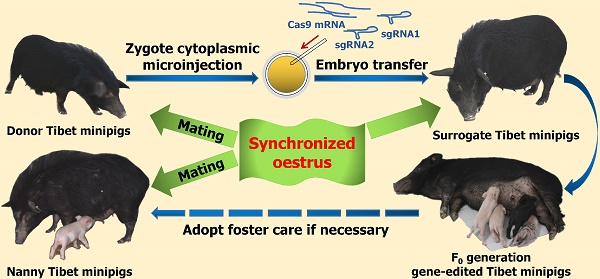
The Tibet minipig is a rare highland pig breed worldwide and has many applications in biomedical and agricultural research. However, Tibet minipigs are not like domesticated pigs in that their ovulation number is low, which is unfavourable for the collection of zygotes. Partly for this reason, few studies have reported the successful generation of genetically modified Tibet minipigs by zygote injection. To address this issue, we described an efficient way to generate gene-edited Tibet minipigs, the major elements of which include the utilization of synchronized oestrus instead of superovulation to obtain zygotes, optimization of the preparation strategy, and co-injection of clustered regularly interspaced short palindromic repeat sequences associated protein 9 (Cas9) mRNA and single-guide RNAs (sgRNAs) into the cytoplasm of zygotes. We successfully obtained allelic TYR gene knockout (TYR-/-) Tibet minipigs with a typical albino phenotype (i.e., red-coloured eyes with light pink-tinted irises and no pigmentation in the skin and hair) as well as TYR-/-IL2RG-/- and TYR-/-RAG1-/- Tibet minipigs with typical phenotypes of albinism and immunodeficiency, which was characterized by thymic atrophy and abnormal immunocyte proportions. The overall gene editing efficiency was 75% for the TYR single gene knockout, while for TYR-IL2RG and TYR-RAG1 dual gene editing, the values were 25% and 75%, respectively. No detectable off-target mutations were observed. By intercrossing F0 generation minipigs, targeted genetic mutations can also be transmitted to gene-edited minipigs' offspring through germ line transmission. This study is a valuable exploration for the efficient generation of gene-edited Tibet minipigs with medical research value in the future.
Keywords: Tibet minipig, cytoplasmic injection, gene editing, oestrus synchronization, CRISPR/Cas9

 Global reach, higher impact
Global reach, higher impact