Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2020; 16(6):981-993. doi:10.7150/ijbs.39919 This issue Cite
Research Paper
Crystal Structures of Human C4.4A Reveal the Unique Association of Ly6/uPAR/α-neurotoxin Domain
1. State Key Laboratory of Structure Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, Fujian, 350002, China.
2. University of Chinese Academy of Sciences, Beijing 100049, China.
3. Institute of Oceanography, Minjiang University, Fuzhou, 350108, China.
4. College of Chemistry, Fuzhou University, Fuzhou, Fujian, China.
5. Finsen Laboratory, Rigshospitalet, DK-2200 Copenhagen N, Denmark.
6. Biotech Research and Innovation Centre (BRIC), University of Copenhagen, DK-2220 Copenhagen N, Denmark.
7. College of Biological Science and Engineering, Fuzhou University, Fuzhou, Fujian, 350116, China.
Abstract
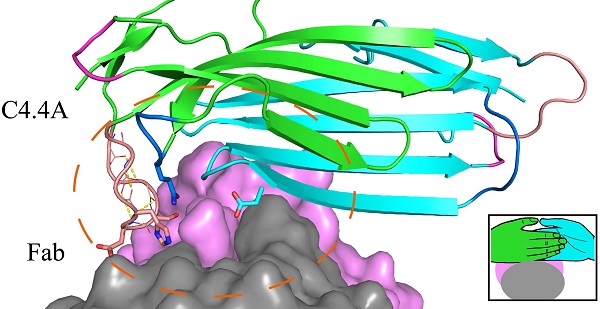
Ly6/uPAR/α-neurotoxin domain (LU-domain) is characterized by the presence of 4-5 disulfide bonds and three flexible loops that extend from a core stacked by several conversed disulfide bonds (thus also named three-fingered protein domain). This highly structurally stable protein domain is typically a protein-binder at extracellular space. Most LU proteins contain only single LU-domain as represented by Ly6 proteins in immunology and α-neurotoxins in snake venom. For Ly6 proteins, many are expressed in specific cell lineages and in differentiation stages, and are used as markers. In this study, we report the crystal structures of the two LU-domains of human C4.4A alone and its complex with a Fab fragment of a monoclonal anti-C4.4A antibody. Interestingly, both structures showed that C4.4A forms a very compact globule with two LU-domain packed face to face. This is in contrast to the flexible nature of most LU-domain-containing proteins in mammals. The Fab combining site of C4.4A involves both LU-domains, and appears to be the binding site for AGR2, a reported ligand of C4.4A. This work reports the first structure that contain two LU-domains and provides insights on how LU-domains fold into a compact protein and interacts with ligands.
Keywords: C4.4A, uPAR, three-fingered fold, LU-domain.

 Global reach, higher impact
Global reach, higher impact