Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(9):1563-1574. doi:10.7150/ijbs.41653 This issue Cite
Research Paper
Downregulation of Nitric Oxide Collaborated with Radiotherapy to Promote Anti-Tumor Immune Response via Inducing CD8+ T Cell Infiltration
1. Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
2. Department of Biological Repositories, Zhongnan Hospital of Wuhan University, Wuhan, China
3. Hubei Key Laboratory of Tumour Biological Behaviors, Zhongnan Hospital of Wuhan University, Wuhan, China
4. Hubei Cancer Clinical Study Center, Zhongnan Hospital of Wuhan University, Wuhan, China
Abstract
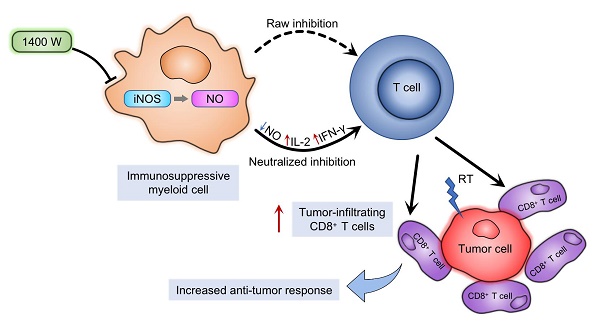
The production of nitric oxide (NO) is a key feature of immunosuppressive myeloid cells, which impair T cell activation and proliferation via reversibly blocking interleukin-2 receptor signaling. NO is mainly produced from L-arginine by inducible NO synthase (iNOS). Moreover, L-arginine is an essential element for T cell proliferation and behaviors. Impaired T cell function further inhibits anti-tumor immunity and promotes tumor progression. Previous studies indicated that radiotherapy activated anti-tumor immune responses in multiple tumors. However, myeloid-derived cells in the tumor microenvironment may neutralize these responses. We hypothesized that iNOS, as an important regulator of the immunosuppressive effects in myeloid-derived cells, mediated radiation resistance of cancer cells. In this study, we used 1400W dihydrochloride, a potent small-molecule inhibitor of iNOS, to explore the regulatory roles of NO in anti-tumor immunity. Radiotherapy and iNOS inhibition by 1400W collaboratively suppressed tumor growth and increased survival time, as well as increased tumor-infiltrating CD8+ T cells and specific inflammatory cytokine levels, in both lung and breast cancer cells in vivo. Our results also suggested that myeloid cell-mediated inhibition of T cell proliferation was effectively counteracted by radiation and 1400W-mediated NO blockade in vitro. Thus, these results demonstrated that iNOS was an important regulator of radiotherapy-induced antitumor immune responses. The combination of radiotherapy with iNOS blockade might be an effective therapy to improve the response of tumors to clinical radiation.
Keywords: iNOS, myeloid cells, radiotherapy, immunotherapy, tumor microenvironment

 Global reach, higher impact
Global reach, higher impact