Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(15):2951-2963. doi:10.7150/ijbs.47999 This issue Cite
Research Paper
IU1 suppresses proliferation of cervical cancer cells through MDM2 degradation
1. Department of Pathology and Pathophysiology, Hubei Provincial Key Laboratory of Developmentally Originated Disease, School of Basic Medical Sciences, Wuhan University, Wuhan, Hubei 430071, P.R. China.
2. Department of Pathology, Wuhan No. 1 Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430022, P.R. China.
3. Department of Breast and Thyroid Surgery, Zhongnan Hospital of Wuhan University, Hubei Key Laboratory of Tumor Biological Behaviors, Hubei Cancer Clinical Study Center, Wuhan, Hubei 430071, P.R. China.
4. Department of Human Anatomy, School of Basic Medical Sciences, Wuhan University, Wuhan, Hubei 430071, P.R. China.
#These authors contributed equally to this work.
Abstract
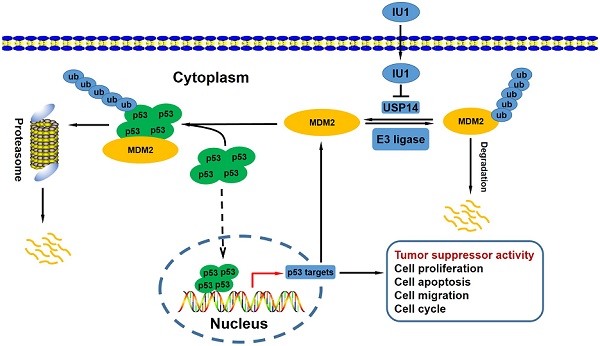
Previous studies have demonstrated that the antitumor potential of IU1 (a pharmacological compound), which was mediated by selective inhibition of proteasome-associated deubiquitinase ubiquitin-specific protease 14 (USP14). However, the underlying molecular mechanisms remain elusive. It has been well established that mdm2 (Murine double minute 2) gene was amplified and/or overexpressed in a variety of human neoplasms, including cervical cancer. Furthermore, MDM2 is critical to cervical cancer development and progression. Relatively studies have reported that USP15 and USP7 stabilized MDM2 protein levels by removing its ubiquitin chain. In the current study, we studied the cell proliferation status after IU1 treatment and the USP14-MDM2 protein interaction in cervical cancer cells. This study experimentally revealed that IU1 treatment reduced MDM2 protein expression in HeLa cervical cancer cells, along with the activation of autophagy-lysosomal protein degradation and promotion of ubiquitin-proteasome system (UPS) function, thereby blocked G0/G1 to S phase transition, decreased cell growth and triggered cell apoptosis. Thus, these results indicate that IU1 treatment simultaneously targets two major intracellular protein degradation systems, ubiquitin-proteasome and autophagy-lysosome systems, which leads to MDM2 degradation and contributes to the antitumor effect of IU1.
Keywords: cervical cancer, USP14, MDM2, IU1, UPS

 Global reach, higher impact
Global reach, higher impact