Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(2):527-538. doi:10.7150/ijbs.55873 This issue Cite
Research Paper
The histone demethylase KDM2B regulates human primordial germ cell-like cells specification
1. State Key Laboratory of Organ Failure Research, Department of Developmental Biology, School of Basic Medical Sciences, Southern Medical University, Guangzhou, Guangdong, China
2. Shunde Hospital of Southern Medical University, Shunde, Guangdong, China
3. Shenzhen Hospital of Southern Medical University, Shenzhen, Guangdong, China
4. Bioland Laboratory (Guangzhou Regenerative Medicine and Health Guangdong Laboratory), Guangzhou, China
5. Sino-America Joint Research Center for Translational Medicine in Developmental Disabilities
6. Department of Gynecology, Zhujiang Hospital, Southern Medical University, Guangzhou, Guangdong, China
7. National Clinical Research Center for Kidney Disease, Guangzhou, China
# These authors contributed equally to this study
Abstract
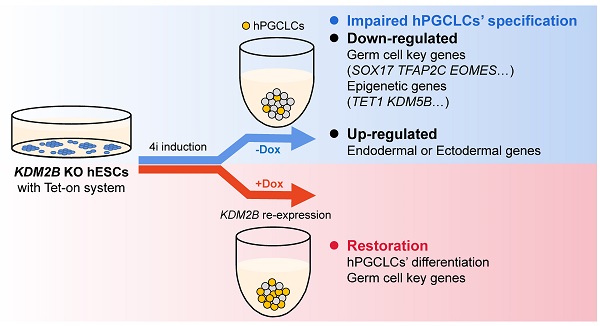
Germline specification is a fundamental step for human reproduction and this biological phenomenon possesses technical challenges to study in vivo as it occurs immediately after blastocyst implantation. The establishment of in vitro human primordial germ cell-like cells (hPGCLCs) induction system allows sophisticated characterization of human primordial germ cells (hPGCs) development. However, the underlying molecular mechanisms of hPGCLC specification are not fully elucidated. Here, we observed particularly high expression of the histone demethylase KDM2B in male fetal germ cells (FGCs) but not in male somatic cells. Besides, KDM2B shared similar expression pattern with hPGC marker genes in hPGCLCs, suggesting an important role of KDM2B in germ cell development. Although deletion of KDM2B had no significant effects on human embryonic stem cell (hESC)'s pluripotency, loss of KDM2B dramatically impaired hPGCLCs differentiation whereas ectopically expressed KDM2B could efficiently rescue such defect, indicating this defect was due to KDM2B's loss in hPGCLC specification. Mechanistically, as revealed by the transcriptional profiling, KDM2B suppressed the expression of somatic genes thus inhibited somatic differentiation during hPGCLC specification. These data collectively indicate that KDM2B is an indispensable epigenetic regulator for hPGCLC specification, shedding lights on how epigenetic regulations orchestrate transcriptional events in hPGC development for future investigation.
Keywords: KDM2B, human primordial germ cell-like cells, epigenetic regulator, fertility

 Global reach, higher impact
Global reach, higher impact