10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(4):915-925. doi:10.7150/ijbs.56733 This issue Cite
Research Paper
FOXM1-induced miR-552 expression contributes to pancreatic cancer progression by targeting multiple tumor suppressor genes
1. Department of Oncology, Shanghai East Hospital, Tongji University School of Medicine, Shanghai 200120, China.
2. Department of Oncology, Ren Ji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200127, China.
#These authors contributed equally to this work.
Abstract
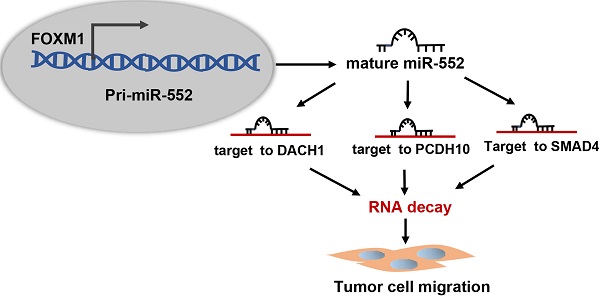
Dysregulation of microRNAs (miRNAs) plays important roles during carcinogenesis. Forkhead box M1 (FOXM1), a well-known oncogenic transcription factor, has been implicated in the progression of multiple cancer types. To find out FOXM1-induced abnormal miRNAs in pancreatic cancer, we analyzed TCGA database and figured out miR-552 as the most relevant miRNA with FOXM1. Molecular experimental results demonstrated that FOXM1 transcriptionally activated miR-552 expression by directly binding to the promoter region of miR-552. In a pancreatic cancer tissue microarray, miR-552 expression was positively correlated with FOXM1 and high expression of miR-552 could predict poor patient outcome. Functionally, overexpression of miR-552 promoted pancreatic cancer cell migration and inhibition of miR-552 attenuated this phenotype. The inhibitory effect on cell migration caused by FOXM1 knockdown could be restored by exogenous expression of miR-552. By informatics analysis, we identified three tumor suppressor genes: DACH1, PCDH10 and SMAD4, all of which were negatively associated with FOXM1 and validated as functionally relevant targets of miR-552. Taken together, our findings provide a new FOXM1-miR-552-DACH1/PCDH10/SMAD4 axis to regulate pancreatic cancer cell progression and new opportunities for therapeutic intervention against this disease.
Keywords: FOXM1, miR-552, pancreatic cancer, migration

 Global reach, higher impact
Global reach, higher impact