Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(4):1050-1060. doi:10.7150/ijbs.57160 This issue Cite
Research Paper
Theaflavin protects against oxalate calcium-induced kidney oxidative stress injury via upregulation of SIRT1
1. Department of Urology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
2. Department of Urology, The Second Affiliated Hospital of Kunming Medical University, Kunming, China.
3. Department of Urology, The Third Affiliated Hospital of Chongqing Medical University, Chongqing, China.
4. Department of Urology, Guizhou Provincial People's Hospital, Guiyang, China.
*These authors contributed equally to this work.
Abstract
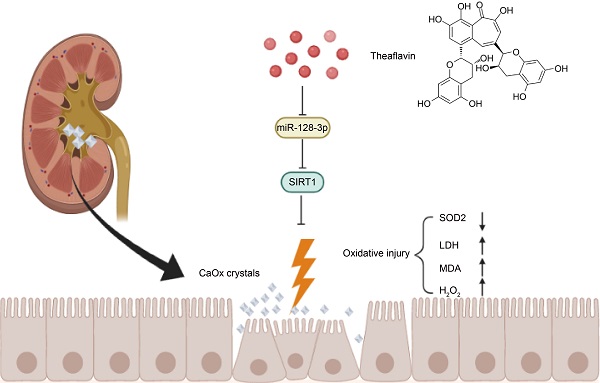
Renal tubular cell injury induced by calcium oxalate (CaOx) is a critical initial stage of kidney stone formation. Theaflavin (TF) has been known for its strong antioxidative capacity; however, the effect and molecular mechanism of TF against oxidative stress and injury caused by CaOx crystal exposure in kidneys remains unknown. To explore the potential function of TF on renal crystal deposition and its underlying mechanisms, experiments were conducted using a CaOx nephrocalcinosis mouse model established by glyoxylate intraperitoneal injection, and HK-2 cells were subjected to calcium oxalate monohydrate (COM) crystals, with or without the treatment of TF. We discovered that TF treatment remarkably protected against CaOx-induced kidney oxidative stress injury and reduced crystal deposition. Additionally, miR-128-3p expression was decreased and negatively correlated with SIRT1 level in mouse CaOx nephrocalcinosis model following TF treatment. Moreover, TF suppressed miR-128-3p expression and further abolished its inhibition on SIRT1 to attenuate oxidative stress in vitro. Mechanistically, TF interacted with miR-128-3p and suppressed its expression. In addition, miR-128-3p inhibited SIRT1 expression by directly binding its 3'-untranslated region (UTR). Furthermore, miR-128-3p activation partially reversed the acceerative effect of TF on SIRT1 expression. Taken together, TF exhibits a strong nephroprotective ability to suppress CaOx-induced kidney damage through the recovery of the antioxidant defense system regulated by miR-128-3p/SIRT1 axis. These findings provide novel insights for the prevention and treatment of renal calculus.
Keywords: Theaflavin, miR-128-3p, SIRT1, oxidative stress, nephrocalcinosis

 Global reach, higher impact
Global reach, higher impact