Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(4):1381-1397. doi:10.7150/ijbs.65476 This issue Cite
Research Paper
Berberine ameliorates DSS-induced intestinal mucosal barrier dysfunction through microbiota-dependence and Wnt/β-catenin pathway
1. Department of Integrated Traditional Chinese and Western Medicine, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China
2. Department of Physical Examination, The Central Hospital of Wuhan, Wuhan, China
3. Tianjin University of Traditional Chinese Medicine, Tianjin, China
4. Department of Gastroenterology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, China
5. School of Medical Biology, South Ural State University, 454080 Chelyabinsk, Russia
6. Institute of Hematology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China
7. Institute of Integrated Bioinfomedicine and Translational Science, School of Chinese Medicine, Hong Kong Baptist University, Hong Kong SAR 999077, China
† These authors contributed equally to this work
Abstract
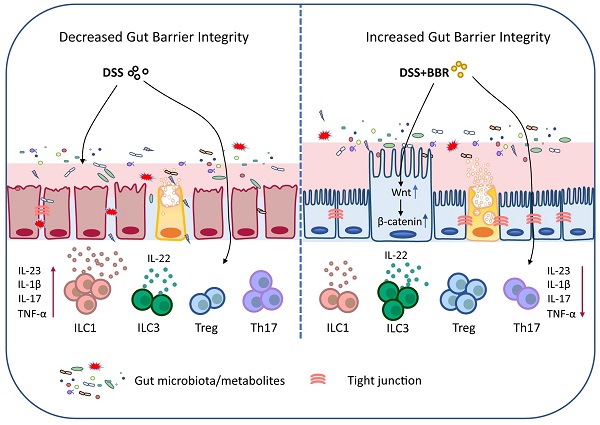
Ulcerative colitis (UC) is an idiopathic, chronic inflammatory disorder of the colon, and it has become one of the world-recognized medical problems as it is recurrent and refractory. Berberine (BBR) is an effective drug for UC treatment. However, the underlying mechanism and targets remain obscure. In this study, we systematically investigated the therapeutic effect and its mechanism of BBR in ameliorating DSS-induced mouse colitis. Expectedly, the colon inflammation was significantly relieved by BBR, and microbiota depletion by antibiotic cocktail significantly reversed the therapeutic effect. Further studies showed that BBR can regulate the abundance and component of bacteria, reestablish the broken chemical and epithelial barriers. Meanwhile, BBR administration dramatically decreased ILC1 and Th17 cells, and increased Tregs as well as ILC3 in colonic tissue of DSS-induced mice, and it was able to regulate the expression of various immune factors at the mRNA level. Moreover, a proteomic study revealed that Wnt/β-catenin pathway was remarkably enhanced in colonic tissue of BBR-treated mice, and the therapeutic effect of BBR was disappeared after the intervention of Wnt pathway inhibitor FH535. These results substantially revealed that BBR restores DSS-induced colon inflammation in a microbiota-dependent manner, and BBR performs its protective roles in colon by maintaining the structure and function of the intestinal mucosal barrier, regulating the intestinal mucosal immune homeostasis and it works through the Wnt/β-catenin pathway. Importantly, these findings also provided the proof that BBR serves as a potential gut microbiota modulator and mucosal barrier protector for UC prevention and therapy.
Keywords: Berberine, Ulcerative colitis, Microbiota, Intestinal mucosal barrier, Wnt/β-catenin pathway

 Global reach, higher impact
Global reach, higher impact