10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(5):2202-2219. doi:10.7150/ijbs.65973 This issue Cite
Research Paper
Melatonin reverses tumor necrosis factor-alpha-induced metabolic disturbance of human nucleus pulposus cells via MTNR1B/Gαi2/YAP signaling
1. Department of Orthopedic Surgery, Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China.
2. Musculoskeletal Research Laboratory, Department of Orthopaedics & Traumatology, Faculty of Medicine, The Chinese University of Hong Kong, Hong Kong, China.
3. Department of Orthopedic Surgery, The Third Affiliated Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China.
4. Department of Orthopedic Surgery, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China.
5. Research Centre for Translational Medicine, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, Guangdong, China.
*Xianjian Qiu, Tongzhou Liang and Zizhao Wu contributed equally to this work.
Abstract
Background: Intervertebral disc degeneration (IDD), the main cause of low back pain, is closely related to the inflammatory microenvironment in the nucleus pulposus (NP). Tumor necrosis factor-α (TNF-α) plays an important role in inflammation-related metabolic disturbance of NP cells. Melatonin has been proven to regulate the metabolism of NP cells, but whether it can protect NP cells from TNF-α-induced damage is still unclear. Therefore, this study aims to investigate the role and specific mechanism of melatonin on regulating the metabolism of NP cells in the inflammatory microenvironment.
Methods: Western blotting, RT-qPCR and immunohistochemistry were used to detect the expression of melatonin membrane receptors (MTNR1A/B) and TNF-α in human NP tissues. In vitro, human primary NP cells were treated with or without vehicle, TNF-α and melatonin. And the metabolic markers were also detected by western blotting and RT-qPCR. The activity of NF-κB signaling and Hippo/YAP signaling were assessed by western blotting and immunofluorescence. Membrane receptors inhibitors, pathway inhibitors, lentiviral infection, plasmids transfection and immunoprecipitation were used to explore the specific mechanism of melatonin. In vivo, the rat IDD model was constructed and melatonin was injected intraperitoneally to evaluate its therapeutical effect on IDD.
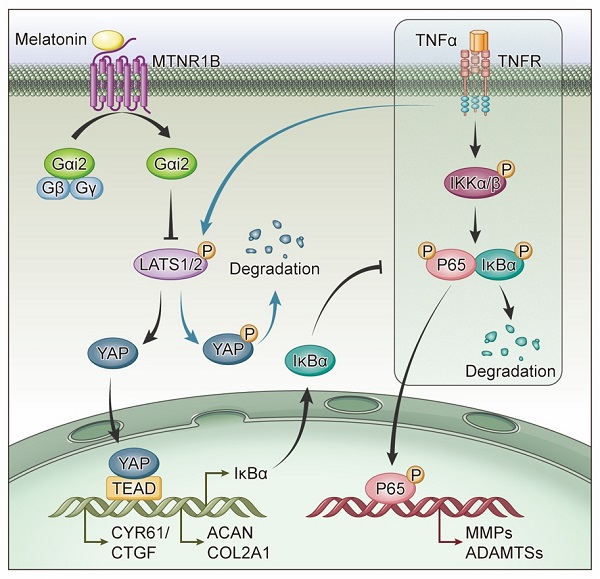
Results: The upregulation of TNF-α and downregulation of melatonin membrane receptors (MTNR1A/B) were observed in degenerative NP tissues. Then we demonstrated that melatonin could alleviate the development of IDD in a rat model and reverse TNF-α-impaired metabolism of NP cells in vitro. Further investigation revealed that the protective effects of melatonin on NP cells mainly rely on MTNR1B, which subsequently activates Gαi2 protein. The activation of Gαi2 could upregulate the yes-associated protein (YAP) level, resulting in anabolic enhancement of NP cells. In addition, melatonin-mediated YAP upregulation increased the expression of IκBα and suppressed the TNF-α-induced activation of the NF-κB pathway, thereby inhibiting the catabolism of NP cells.
Conclusions: Our results revealed that melatonin can reverse TNF-α-impaired metabolism of NP cells via the MTNR1B/Gαi2/YAP axis and suggested that melatonin can be used as a potential therapeutic drug in the treatment of IDD.
Keywords: intervertebral disc degeneration, nucleus pulposus cells, melatonin, TNF-α, MTNR1B, yes-associated protein.

 Global reach, higher impact
Global reach, higher impact