Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(6):2568-2582. doi:10.7150/ijbs.71327 This issue Cite
Research Paper
ERK-mediated Cytoplasmic Retention of USP11 Contributes to Breast Cancer Cell Proliferation by Stabilizing Cytoplasmic p21
1. Molecular Science and Biomedicine Laboratory (MBL), State Key Laboratory of Chemo/Biosensing and Chemometrics, College of Biology, College of Chemistry and Chemical Engineering, Aptamer Engineering Center of Hunan Province, Hunan University, Changsha, Hunan 410082, China.
2. Molecular Biology Research Center and Center for Medical Genetics, School of Life Sciences, Central South University, Changsha, Hunan 410078, China.
3. Department of Gynecology, Xiangya Hospital, Central South University, Changsha, Hunan 410078, China.
4. Department of Critical Care Medicine, the Second Xiangya Hospital, Central South University, Changsha, Hunan 410011, China.
#These authors contributed equally to this work.
Abstract
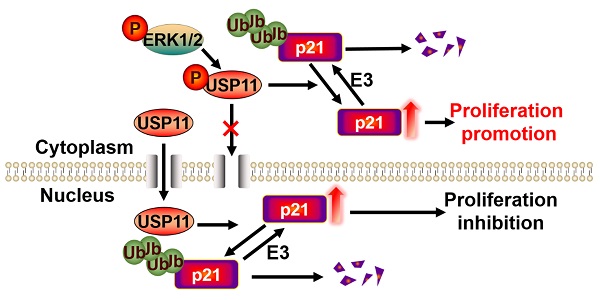
Breast cancer ranks as the most frequently diagnosed cancer among women worldwide. Elevated cytoplasmic p21 levels are often found in breast cancer tissues and related to a poor prognosis. However, the underlying mechanisms that lead to the stabilization of cytoplasmic p21 protein, which normally has a very short half-life, remain obscure. In this study, we found that there was a strong correlation between p21 and USP11 in the cytoplasm of breast cancer tissues and cells. Furthermore, we revealed that ERK1/2 phosphorylated USP11 at the Ser905 site, which promoted the cytoplasmic localization of USP11. In the cytoplasm, USP11 colocalized and interacted with p21. As a result, USP11 catalyzed the removal of polyubiquitin chains bound to cytoplasmic p21 and resulted in its stabilization. Functionally, USP11-mediated stabilization of cytoplasmic p21 induced breast cancer cell proliferation in vitro and in vivo. Our findings provide the first evidence that ubiquitinated p21 in the cytoplasm can be recycled through USP11-mediated deubiquitination, and we identified the USP11-p21 axis in the cytoplasm as a potential therapeutic target for breast cancer control.
Keywords: Cytoplasmic p21, ERK1/2, Phosphorylation, Stabilization, USP11

 Global reach, higher impact
Global reach, higher impact