Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(7):2914-2931. doi:10.7150/ijbs.71571 This issue Cite
Research Paper
GRP75-faciliated Mitochondria-associated ER Membrane (MAM) Integrity controls Cisplatin-resistance in Ovarian Cancer Patients
1. Department of Cell Biology, School of Medicine, Nankai University, Tianjin, 300071, P. R. China.
2. Department of Pathology, Third Central Hospital of Tianjin Medical University, 83 Jintang Road, Tianjin, 300170, P. R. China.
3. Tianjin's Clinical Research Center for Cancer, Tianjin Medical University Cancer Institute and Hospital, Tianjin, P. R. China.
4. Department of Pharmaceutical Sciences, College of Pharmacy and Health Sciences, St. John's University, Queens, New York, NY, 11439, USA.
*These authors contributed equally to this work.
Abstract
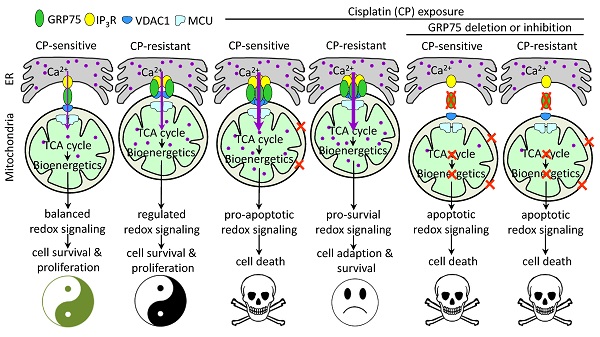
Background: Control of ER-mitochondrial Ca2+ fluxes is a critical checkpoint to determine cell fate under stress. The 75-kDa glucose-regulated protein (GRP75) is a key tether protein facilitating mitochondria-associated ER membrane (MAM) formation through the IP3R-GRP75-VDAC1 complex. Although GRP75 contributes to cisplatin (CP)-resistance of ovarian cancer (OC), the underlying mechanisms are not clear.
Methods: CP-resistant and -sensitive OC cell lines with GRP75 stable modulation were established. Confocal, PLA, co-IP, and TEM analysis were utilized to detect MAM integrity. Live cell Ca2+ imaging, intracellular ATP, ROS, and NAD+ assays were utilized to investigate ER-to-mitochondrial Ca2+ transfer and mitochondrial bioenergetics. Western blot, flow cytometry, CCK-8, Δψm, and mPTP assays were utilized to examine apoptotic cell death. Bioinformatics, patient's specimens, and immunohistochemistry were conducted to obtain the clinical relevance for GRP75-facilitated MAM formation.
Results: GRP75-faciliated MAM formation was enriched in CP-resistant OC cells. CP-exposure only increased MAM formation in CP-sensitive OC cells, and enrichment of GRP75 and VDAC1 at MAMs is indispensable to CP-resistance. Diminishing MAM integrity by GRP75-deficiency reduced ER-to-mitochondria Ca2+ transfer, accelerated CP-induced mitochondrial dysfunction, provoked catastrophic ROS, and enhanced CP-triggered apoptotic cell death in OC cells. Clinical investigations confirmed the enrichment of GRP75-faciliated MAM formation in relapsed OC patients, and such enrichment was associated with the CP-resistance phenotype.
Conclusion: GRP75-overexpression confers CP-resistance by distinctively managing MAM-facilitated Ca2+ fluxes and the pro-survival ROS signal, whereas GRP75-deficiency induces cell death via bioenergetic crisis and apoptotic ROS accumulation in OC cells. Our results show that GRP75-faciliated MAM formation is a potential target to overcome CP-resistance of OC.
Keywords: mitochondria-associated ER membrane, glucose-regulated protein, Ca2+ fluxes, cisplatin-resistance, ovarian cancer

 Global reach, higher impact
Global reach, higher impact