Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(7):3082-3101. doi:10.7150/ijbs.70524 This issue Cite
Research Paper
LAMC1-mediated preadipocytes differentiation promoted peritoneum pre-metastatic niche formation and gastric cancer metastasis
1. Department of Gastrointestinal Surgery & Department of Gastric and Colorectal Surgical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, Hubei, 430071, China.
2. Department of obstetrics and gynecology, Guangzhou Women and Children's Medical Center, Guangzhou, Guangdong, 510623, China.
3. Hubei Key Laboratory of Tumor Biological Behaviors, Wuhan, Hubei, 430071, China.
4. College of Life Sciences of Wuhan University, Wuhan, Hubei, 430072, China.
* Yan Fang, Rongzhang Dou, Sihao Huang and Lei Han contributed equally to this work.
Abstract
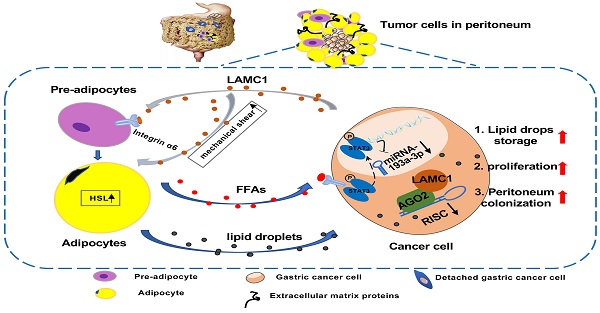
Gastric cancer is anatomically proximal to peritoneum. Gastric cancer peritoneal metastasis is a complex biological process which is corresponded with disharmony within dysfunctional adipose tissue and metabolism reprogramming. Laminin gamma 1 (LAMC1) is highly expressed in cancer cells of peritoneal metastatic sites, however, the mechanism of LAMC1-metiated gastric cancer metastases to adipose tissue-rich peritoneum remains unclear. In our study, immunohistochemical staining, single cell sequencing, a co-culture model, luciferase reporter, RNA immunoprecipitation (RIP), Chromatin immunoprecipitation (CHIP) and single-molecular magnetic tweezers assays were conducted, and our results showed that LAMC1 related to Perilipin-1 content was highly expressed in peritoneal metastatic sites and mainly secreted by tumor cells. Gastric cancer cells secreted LAMC1 in an autocrine manner to detached from the primary site and promoted preadipocytes mature, rupture and release of free fatty acids (FFAs) in the peritoneal microenvironment to form pre-metastatic niche by the paracrine pathway. Reversely, differentiated preadipocyte-derived conditioned medium inhibited glycolysis and enhanced fatty acid oxidation (FAO) rate to promote cell proliferation, mesenchymal-epithelial transformation which led to tumor peritoneal colonization. In terms of biological mechanisms, one of differentiated preadipocyte-derived FFAs, palmitic acid-activated STAT3 inhibited miR-193a-3p by binding to its promoter directly; Using single-molecular magnetic tweezers, this binding manner was proved to be stable, reversable and ATP-dependent. Moreover, miR-193a-3p regulated LAMC1 in a post-translational manner. Furthermore, high LAMC1 expression in serum predicted a higher risk of peritoneal metastasis. In conclusion, our results illustrated that palmitic acid/p-STAT3/miR-193a-3p/LAMC1 pathway promotes preadipocyte differentiation, pre-metastatic niche formation and gastric cancer cell colonization to peritoneum.
Keywords: LAMC1, Gastric cancer peritoneal metastasis, Preadipocyte, Free fatty acids (FFAs), Predictive value

 Global reach, higher impact
Global reach, higher impact