Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(8):3223-3236. doi:10.7150/ijbs.70335 This issue Cite
Research Paper
METTL3 promotes cell cycle progression via m6A/YTHDF1-dependent regulation of CDC25B translation
1. Department of Pathology, Shanghai First Maternity and Infant Hospital, Tongji University, Shanghai 200120, China.
2. Department of Pathology, School of Medicine, Jinan University, Guangzhou 510632, China.
3. Department of Gynecology, Shanghai East Hospital, School of Medicine, Tongji University, Shanghai 200120, China.
4. The Third Affiliated Hospital, Sun Yat-Sen University, Guangzhou 510630, China.
*These authors contribute equally.
Abstract
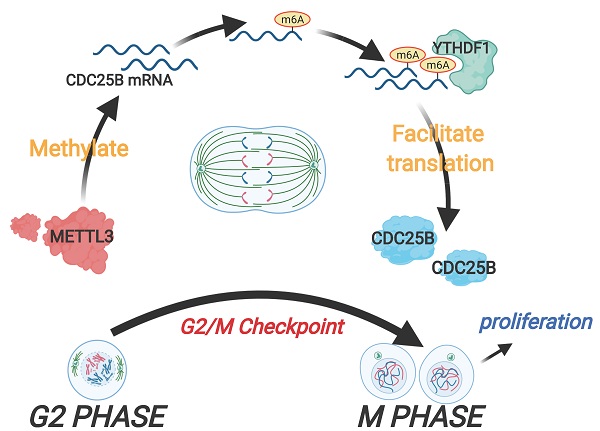
The cell cycle machinery controls cell proliferation and the dysregulation of the cell cycle lies at the heart of carcinogenesis. Thus, exploring the unknown regulators involved in the cell cycle not only contribute to better understanding of cell proliferation but also provide substantial improvement to cancer therapy. In this study, we identified that the expression of methyltransferase METTL3 was upregulated in the M phase. Overexpression of METTL3 facilitated cell cycle progression, induced cell proliferation in vitro and enhanced tumorigenicity in vivo, while knockdown of METTL3 reversed these processes. METTL3 induced CDC25B mRNA m6A modification in the M phase, which accelerated the translation of CDC25B mRNA through YTHDF1-dependent m6A modification. Clinical data analysis showed that METTL3 and CDC25B were highly expressed in cervical cancer. Our work reveals that a new mechanism regulates cell cycle progression through the METTL3/m6A/CDC25B pathway, which provides insight into the critical roles of m6A methylation in the cell cycle.
Keywords: METTL3, cell cycle, m6A

 Global reach, higher impact
Global reach, higher impact