Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(10):4118-4134. doi:10.7150/ijbs.72528 This issue Cite
Research Paper
Targeting Ferroptosis as a Novel Approach to Alleviate Aortic Dissection
1. Division of Cardiothoracic and Vascular Surgery, Sino-Swiss Heart-Lung Transplantation Institute, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, China.
2. Department of Cardiology, Renmin Hospital of Wuhan University, Wuhan, Hubei, China.
3. Key Laboratory of Organ Transplantation, Ministry of Education; NHC Key Laboratory of Organ Transplantation; Key Laboratory of Organ Transplantation, Chinese Academy of Medical Sciences, Wuhan, Hubei, China.
*These authors contributed equally to this work.
Abstract
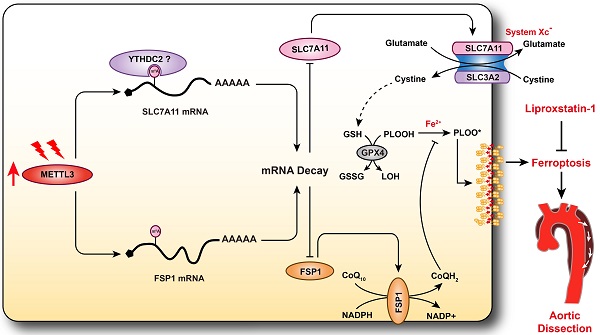
A variety of programmed cell death types have been shown to participate in the loss of smooth muscle cells (SMCs) during the development of aortic dissection (AD), but it is still largely unclear whether ferroptosis is involved in the development of AD. In the present study, we found that the expression of key ferroptosis regulatory proteins, solute carrier family 7 member 11 (SLC7A11), ferroptosis suppressor protein 1 (FSP1) and glutathione peroxidase 4 (GPX4) were downregulated in aortas of Stanford type A AD (TAAD) patients, and liproxstatin-1, a specific inhibitor of ferroptosis, obviously abolished the β-aminopropionitrile (BAPN)-induced development and rupture of AD in mice. Furthermore, the expression of methyltransferase-like 3 (METTL3), a major methyltransferase of RNA m6A, was remarkably upregulated in the aortas of TAAD patients, and the protein levels of METTL3 were negatively correlated with SLC7A11 and FSP1 levels in human aortas. Overexpression of METTL3 in human aortic SMCs (HASMCs) inhibited, while METTL3 knockdown promoted SLC7A11 and FSP1 expression. More importantly, overexpression of METTL3 facilitated imidazole ketone erastin- and cystine deprivation-induced ferroptosis, while knockdown of METTL3 repressed ferroptosis of HASMCs. Overexpression of either SLC7A11 or FSP1 largely abrogated the effect of METTL3 on HASMC ferroptosis. Therefore, we have revealed that ferroptosis is a critical cause of AD in both humans and mice and that METTL3 promotes ferroptosis of HASMCs by inhibiting the expression of SLC7A11 and FSP1. Thus, targeting ferroptosis or m6A RNA methylation is a potential novel strategy for the treatment of AD.
Keywords: Aortic dissection, METTL3, Ferroptosis, SLC7A11, FSP1/AIFM2, Liproxstatin-1

 Global reach, higher impact
Global reach, higher impact