Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(11):4329-4340. doi:10.7150/ijbs.71581 This issue Cite
Research Paper
PRMT5 acts as a tumor suppressor by inhibiting Wnt/β-catenin signaling in murine gastric tumorigenesis
1. State Key Laboratory of Proteomics, Beijing Proteome Research Centre, National Centre for Protein Sciences, Beijing Institute of Lifeomics, Beijing 102206, China.
2. Laboratory Animal Center, the Academy of Military Medical Sciences, Beijing 100071, China.
3. Department of Urology, the Third Medical Center of Chinese PLA General Hospital, Beijing 100039, China.
*These authors contributed equally to this work.
Abstract
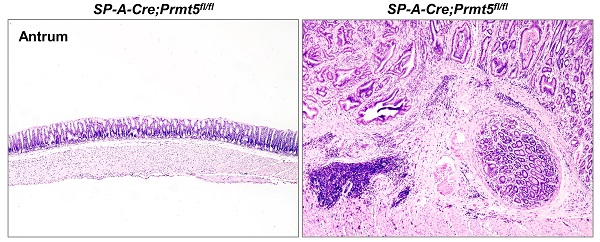
Previous studies have demonstrated the in vitro oncogenic role of protein arginine methyltransferase 5 (PRMT5) in gastric cancer cell lines. The in vivo function of PRMT5 in gastric tumorigenesis, however, is still unexplored. Here, we showed that Prmt5 deletion in mouse gastric epithelium resulted in spontaneous tumorigenesis in gastric antrum. All Prmt5-deficient mice displayed intestinal-type gastric cancer within 4 months of age. Of note, 20% (2/10) of Prmt5 mutants finally developed into invasive gastric cancer by 8 months of age. Gastric cancer caused by PRMT5 loss exhibited the increase in Lgr5+ stem cells, which are proposed to contribute to both the gastric tumorigenesis and progression in mouse models. Consistent with the notion that Lgr5 is the target of Wnt/β-catenin signaling, whose activation is the most predominant driver for gastric tumorigenesis, Prmt5 mutant gastric cancer showed the activation of Wnt/β-Catenin signaling. Furthermore, in human gastric cancer samples, PRMT5 deletion and downregulation were frequently observed and associated with the poor prognosis. We propose that as opposed to the tumor-promoting role of PRMT5 well-established in the progression of various cancer types, PRMT5 functions as a tumor suppressor in vivo, at least during gastric tumor formation.
Keywords: PRMT5, gastric epithelium, tumorigenesis, Wnt/β-Catenin signaling

 Global reach, higher impact
Global reach, higher impact