Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(11):4388-4399. doi:10.7150/ijbs.69890 This issue Cite
Research Paper
Fibroblast-like cells Promote Wound Healing via PD-L1-mediated Inflammation Resolution
1. Department of Stem Cell & Regenerative Medicine, State Key Laboratory of Trauma, Burn and Combined Injury, Daping Hospital, Army Medical University, Chongqing 400042, P.R. China.
2. Department of Dermatology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou 325035, P.R. China.
3. College of Sericulture, Textile and Biomass Sciences, Southwest University, Chongqing 400715, P.R. China.
* These authors contributed equally to this work
Abstract
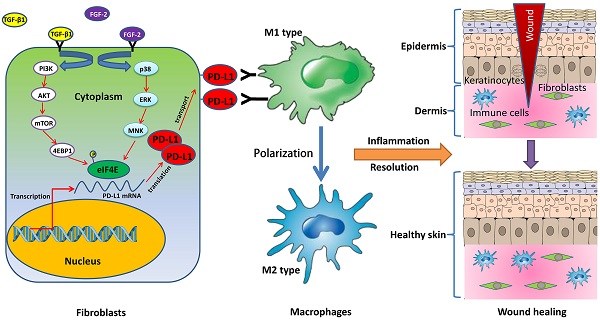
Chronic non-healing wounds fail to progress beyond the inflammatory phase, characterized by a disorder of inflammation resolution. PD-1/PD-L1, a major co-inhibitory checkpoint signaling, plays critical roles in tumor immune surveillance and the occurrence of inflammatory or autoimmune diseases, but its roles in wound healing remains unclear. Here, we described a novel function of PD-L1 in fibroblast-like cells as a positive regulator of wound healing. PD-L1 dynamically expressed on the fibroblast-like cells in the granulation tissue during wound healing to form a wound immunosuppressive microenvironment, modulate macrophages polarization from M1-type to M2-type, and initiates resolution of inflammation, finally accelerate wound healing. Loss of PD-L1 delayed wound healing, especially in mice with LPS-induced severe inflammation. Furthermore, the mainly regulatory mechanism is that combination of FGF-2 and TGF-β1 promotes PD-L1 translation in fibroblasts through enhancing the eIF4E availability regulated by both PI3K-AKT-mTOR-4EBP1 and p38-ERK-MNK signaling pathways. Our results reveal the positive role of PD-L1 in wound healing, and provide a new strategy for the treatment of chronic wounds.
Keywords: PD-L1, FGF-2, TGF-β1, Fibroblast, Wound healing, Inflammation resolution

 Global reach, higher impact
Global reach, higher impact