10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2022; 18(14):5539-5553. doi:10.7150/ijbs.71809 This issue Cite
Research Paper
Thioredoxin 1 supports colorectal cancer cell survival and promotes migration and invasion under glucose deprivation through interaction with G6PD
1. Central Laboratory, the First Affiliated Hospital of Wenzhou Medical University, Wenzhou 325000, China.
2. Changzhou maternal and Child Health Care Hospital, Changzhou Medical Center, Nanjing Medical University, Changzhou, 213000, China.
#These authors contributed equally to this paper.
Abstract
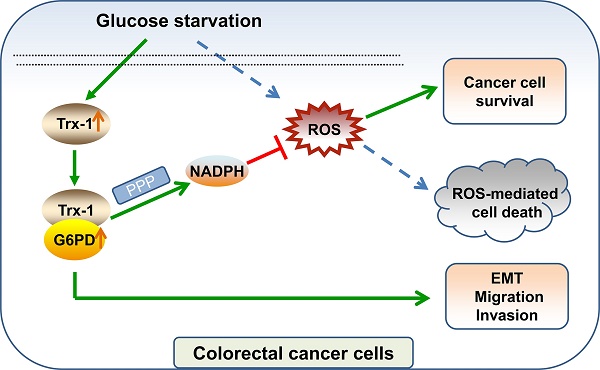
Overcoming energy stress is a critical step for cells in solid tumors. Under this stress microenvironment, cancer cells significantly alter their energy metabolism to maintain cell survival and even metastasis. Our previous studies have shown that thioredoxin-1 (Trx-1) expression is increased in colorectal cancer (CRC) and promotes cell proliferation. However, the exact role and mechanism of how Trx-1 is involved in energy stress are still unknown. Here, we observed that glucose deprivation of CRC cells led to cell death and promoted the migration and invasion, accompanied by upregulation of Trx-1. Increased Trx-1 supported CRC cell survival under glucose deprivation. Whereas knockdown of Trx-1 sensitized CRC cells to glucose deprivation-induced cell death and reversed glucose deprivation-induced migration, invasion, and epithelial-mesenchymal transition (EMT). Furthermore, we identified glucose-6-phosphate dehydrogenase (G6PD) interacting with Trx-1 by HuPortTM human protein chip, co-IP and co-localization. Trx-1 promoted G6PD protein expression and activity under glucose deprivation, thereby increasing nicotinamide adenine dinucleotide phosphate (NADPH) generation. Moreover, G6PD knockdown sensitized CRC cells to glucose deprivation-induced cell death and suppressed glucose deprivation-induced migration, invasion, and EMT. Inhibition of Trx-1 and G6PD, together with inhibition of glycolysis using 2-deoxy-D-glucose (2DG), resulted in significant anti-tumor effects in CRC xenografts in vivo. These findings demonstrate a novel mechanism and may represent a new effective therapeutic regimen for CRC.
Keywords: Thioredoxin 1, G6PD, Glucose deprivation, Colorectal cancer

 Global reach, higher impact
Global reach, higher impact