ISSN: 1449-2288
Int J Biol Sci 2022; 18(16):6129-6144. doi:10.7150/ijbs.74951 This issue Cite
Research Paper
Down-regulation of BMAL1 by MiR-494-3p Promotes Hepatocellular Carcinoma Growth and Metastasis by Increasing GPAM-mediated Lipid Biosynthesis
1. Department of Pain Treatment, Tangdu Hospital, Fourth Military Medical University, Xi'an, Shaanxi, 710038, China.
2. State Key Laboratory of Cancer Biology and Department of Physiology and Pathophysiology, Fourth Military Medical University, Xi'an, Shaanxi, 710032, China.
3. Third Department of Medical Oncology, Shaanxi Provincial Cancer Hospital, Xi'an, Shaanxi, 710032, China.
4. Department of Hepatobiliary Surgery, Xijing Hospital, Fourth Military Medical University, Xi'an, Shaanxi, 710032, China.
5. Experimental Teaching Center of Basic Medicine, Fourth Military Medical University, Xi'an, Shaanxi, 710032, China.
#These authors contributed equally to this work.
Abstract
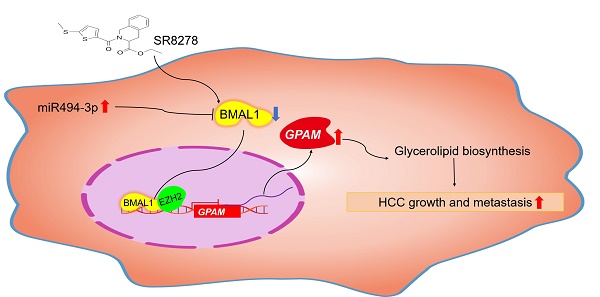
The circadian clock confers daily rhythmicity to many crucial biological processes and behaviors and its disruption is closely associated with carcinogenesis in several types of cancer. Brain and muscle arnt-like protein 1 (BMAL1) is a core circadian rhythm component in mammals and its dysregulation has been shown to contribute to aberrant metabolism in human diseases. However, the biological functions of BMAL1, especially its involvement in aberrant lipid metabolism in hepatocellular carcinoma (HCC), remain elusive. In the present study, we found that BMAL1 was frequently down-regulated in HCC cells mainly due to the up-regulation of miR-494-3p. Down-regulation of BMAL1 was significantly associated with poor survival in HCC patients. BMAL1 down-regulation promoted HCC cell growth and metastasis both in vitro and in vivo. Mechanistically, through cooperating with EZH2, BMAL1 transcriptionally suppressed the expression of glycerol-3-phosphate acyltransferase mitochondrial (GPAM), a key enzyme involved in the regulation of lipid biosynthesis, leading to reduced levels lysophosphatidic acid (LPA), which have long been known as mediator of oncogenesis. Particularly, treatment with SR8278, an activator of BMAL1, exhibited a therapeutic effect on HCC in vitro and in vivo. In conclusion, BMAL1 plays a critical anti-oncogenic role in HCC, providing strong research evidence for BMAL1 as a prospective target for HCC therapy.
Keywords: BMAL1, GPAM, EZH2, lipid biosynthesis, growth, metastasis, hepatocellular carcinoma

 Global reach, higher impact
Global reach, higher impact