Impact Factor ISSN: 1449-2288
Int J Biol Sci 2023; 19(2):552-570. doi:10.7150/ijbs.76187 This issue Cite
Review
Targeting Breast Cancer Stem Cells
1. Fudan University Shanghai Cancer Center & Institutes of Biomedical Sciences; State Key Laboratory of Genetic Engineering; Cancer Institutes; Key Laboratory of Breast Cancer in Shanghai; The Shanghai paracrine Key Laboratory of Medical Epigenetics; Shanghai Key Laboratory of Radiation Oncology; The International Co-laboratory of Medical Epigenetics and Metabolism, Ministry of Science and Technology; Shanghai Medical College; Fudan University, Shanghai 200032, China.
2. Key Laboratory of Animal Models and Human Disease Mechanisms of Chinese Academy of Sciences and Yunnan Province, Kunming Institute of Zoology, Kunming 650201, China.
3. Kunming College of Life Sciences, the University of the Chinese Academy of Sciences, Kunming 650201, China.
4. Jiangsu Key Lab of Cancer Biomarkers, Prevention and Treatment, Collaborative Innovation Center for Cancer Medicine, Nanjing Medical University, Nanjing 211166, China.
5. Academy of Biomedical Engineering, Kunming Medical University, Kunming 650500, China.
6. The Third Affiliated Hospital, Kunming Medical University, Kunming 650118, China.
#Equal contributions to this work.
Received 2022-6-15; Accepted 2022-12-9; Published 2023-1-1
Abstract
The potential roles of breast cancer stem cells (BCSCs) in tumor initiation and recurrence have been recognized for many decades. Due to their strong capacity for self-renewal and differentiation, BCSCs are the major reasons for poor clinical outcomes and low therapeutic response. Several hypotheses on the origin of cancer stem cells have been proposed, including critical gene mutations in stem cells, dedifferentiation of somatic cells, and cell plasticity remodeling by epithelial-mesenchymal transition (EMT) and the tumor microenvironment. Moreover, the tumor microenvironment, including cellular components and cytokines, modulates the self-renewal and therapeutic resistance of BCSCs. Small molecules, antibodies, and chimeric antigen receptor (CAR)-T cells targeting BCSCs have been developed, and their applications in combination with conventional therapies are undergoing clinical trials. In this review, we focus on the features of BCSCs, emphasize the major factors and tumor environment that regulate the stemness of BCSCs, and discuss potential BCSC-targeting therapies.
Keywords: Breast cancer stem cell, Epithelial-Mesenchymal Transition, Tumor microenvironment, Therapeutic strategies
Introduction
The mammary epithelium is composed of two main cellular lineages: luminal epithelial cells and myoepithelial cells. Accumulated evidence from both mice and humans showed that these two epithelial cells are derived from common ancestors, namely the mammary epithelial stem cells [1, 2]. The mammary stem cells (MaSCs) were observed to reside in the outer basal compartment and are responsible for the production of basal myoepithelial progeny cells (unipotent stem cells) or basal inner luminal progeny cells (multi/bi-potent stem cell) [3, 4]. The MaSCs are heterogeneous and comprise multiple diverse subsets to meet the developmental needs including protein C receptor (PROCR)-expressing cells, transcription factor B-cell lymphoma/leukemia 11b (Bcl11b)-expressing cells, etc. [5-7].
Interestingly, a correlation between breast cancer subtypes and distinct stem cell populations is observed. Based on the expressions of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor 2 (HER2/ERBB2), breast cancer is simply classified into three major subtypes: luminal type with hormone receptor positive/ERBB2 negative (70% of patients), ERBB2 positive (15-20%), and triple-negative (TNBC, tumors lacking all three standard molecular markers; 15%) [8, 9]. Generally, the luminal subtype is ER-positive and the TNBC subtype largely overlaps with the basal-like subtype. It has been found that luminal cancers have similar profiles to mature ER+PR+ luminal cells [10]. In addition, ER-PR- luminal progenitors share similar expression patterns with basal-like breast cancers [11]. Molyneux et al. found breast cancer 1 (BRCA1) deletion in mouse mammary epithelial luminal progenitors grows tumors that resemble human BRCA1 breast cancers, supporting a derivation of the majority of human BRCA1-associated tumors from luminal progenitors [12].
Surgery is usually the first choice for breast cancer treatment. Most women may receive additional treatment, including chemotherapy, hormone therapy, radiation or targeted therapy. More than 90% breast cancer don't occur metastasis at the time of diagnosis [13]. Nevertheless, it is estimated that nearly 30% of women with early-stage breast cancer will develop metastatic disease [14], and the median survival for these women ranges from 8 to 36 months [15].
Conventional therapies for breast cancer are insufficient to eliminate all cancer cells in the tumors, especially cancer stem cells (CSCs), which can lead to recurrence and drug resistance [16]. BCSCs are characterized by their ability to initiate tumors from tiny numbers and are insensitive to chemo- or radio-therapies [17, 18]. Analogous to stem cells, BCSCs exhibit a delicate equilibrium between self-renewal and differentiation to maintain tumor homeostasis [19]. On the one hand, they differentiate into various and heterogeneous cancer cells, forming the tumor bulk [20, 21]. On the other hand, they perpetuate the stem cell pool by self-renewal throughout cancer progression [22, 23]. In response to environmental stimuli, when non-CSCs are eliminated by traditional chemo- or radio-therapies, BCSCs survive oxidative stress or DNA damage due to their cell dormancy and enhanced ability for DNA damage repair and drug efflux [24]. During tumor metastasis, BCSCs first infiltrate the surrounding mesenchyme or enter the circulation by EMT. Then, BCSCs undergo a reciprocal program called mesenchymal-epithelial transition (MET) to form sizable metastatic colonies at distal organ sites [25]. Considering that BCSCs are closely related to cancer prognosis and progression, numerous efforts have been made to characterize and eradicate BCSCs.
In this review, we comprehensively introduced the origin of BCSCs and summarized the latest research progresses on BCSCs, including features, functions, and targeting strategies. Finally, we discuss future research directions in this field.
The origin and heterogeneity of BCSCs
The observation that BCSCs share many traits with MaSCs indicated that these two cell populations might have a common origin. For example, CD44 and PROCR label both MaSCs and BCSCs [5, 26-28]. Several signaling networks essential for stemness maintenance are common in both MaSCs and BCSCs, including the NOTCH, Hedgehog, and Wnt pathways.
CD133 is initially discovered in hematopoietic stem cells and bone marrow-derived circulating endothelial progenitors [29-31]. Then, CD133+ cancer cells were reported to be featured by strong self-renewal potential and contributed to vasculogenic mimicry (VM) in TNBC [32]. In BRCA1-associated breast cancer, CD133+ BCSCs are distinct from CD24-CD44+ BCSCs, suggesting BCSC populations are heterogenous [33].
Integrin α6, also known as CD49f, may be the only biomarker present in more than 30 different populations of stem cells including embryonic stem cells, embryonic neuronal stem cells, hematopoietic stem cells, and cancer stem cells [34]. Therefore, a hypothesis that mutagenesis in normal stem cells results in CSC formation has been proposed [35]. For instance, Van Keymeulen et al. discovered that mutation of phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA) in mammary luminal stem cells leads to luminal or basal-like breast tumors. In contrast, BRCA1-basal-like breast cancers might originate from basal stem cells [36]. Liu et al. found that BRCA1 participated in the differentiation of stem cells. When BRCA1 is lost, uncontrollable accumulation of stem cells occurs, and finally, a small number of stem cells missing BRCA1 develop into cancer [37].
Another speculation on the origin of cancer stem cells is EMT, an essential program for embryonic development. The first work on the relationship between EMT and stemness was conducted in the mammary gland. microRNA (miRNA)-200 family comprises five members (miRNA-200a, -200b, 200c, -141 and -429) and all members were markedly downregulated in cells with EMT [38]. EMT induced mitochondrial fusion during asymmetric division of MaSCs via miR-200c-peroxisome proliferator-activated receptor-γ coactivator 1 alpha (PGC1α)-mitofusin (MFN1) pathway. The fused mitochondrial was asymmetrically separated to stem cell-like progeny with enhanced reactive oxygen species (ROS) scavenging and glutathione synthesis capacities. Downregulation of the EMT/miR-200 axis promotes the expansion of mammary stem cells by transforming growth factor beta (TGF-β)-induced asymmetric division [39]. Evidence has shown that during tumor progression, whether in the very beginning stage or the final metastatic phase, cancer cells activate EMT transcription factors (EMT-TFs) to favor their proliferation and survival. For example, Wellner et al. showed that zinc finger E-box binding homeobox 1 (ZEB1), a vital activator of cancer metastasis, promoted tumor-initiation capacity by repressing stemness-inhibiting miRNAs [40]. Poornima et al. overexpressed EMT-TF SLUG in CD24+/CD44- MCF-10A cells and MCF-7 breast cancer cells and found a small population of CD24-/CD44+ stem-like cells emerged [41]. Their work proved that the association of EMT and stem cells was adopted by normal epithelial cells and the corresponding derived neoplastic cells. However, whether cells gain pluripotency by EMT remains controversial. Ocana et al. discovered that the EMT activator paired related homeobox 1 (PRRX1) cooperates with EMT-TF Twist1 in all EMT-related characteristics, but it suppressed stemness in breast cancer [25, 42].
Acquisition of a partial EMT phenotype or a hybrid E/M state seems more critical for stemness and tumorigenicity of breast cancer cells. Liu's study found the first evidence of an association between BCSC heterogeneity and EMT. They discovered that a CD24-/CD44+/ALDH+ BCSC population with intermediate EMT characteristics had the most vital capacity for stemness and tumorigenesis [43]. Another study on integrin subunit beta 4 (ITGB4)-positive BCSCs showed that this BCSC subpopulation was in an intermediate E/M state and induced a worse five-year probability of relapse-free survival [44]. Indeed, more and more evidence showed that the hybrid E/M state led to strong stemness and poor prognosis and was independent of cellular origin [45-48]. Cornelia et al. isolated highly malignant CD104+/CD44hi breast cancer cells expressing both epithelial and mesenchymal markers. Complete transition to a mesenchymal state decreases their capacity for stemness and tumorigenesis [49]. Satiwik et al. employed a mechanism-based mathematical modeling framework to demonstrate that intermediate E/M phenotype enrichment is coupled with enhanced stemness and stemness is more likely to develop in intermediate E/M phenotypes than in “pure” epithelial/mesenchymal phenotypes [50]. Meredith et al. analyzed three single-cell clones with intermediate E/M state and demonstrated core-binding factor subunit beta (CBFβ) was responsible for stabilizing and maintaining metastatic ability. Their results showed that EMT score alone was not associated with survival while CBFβ showed predictive value for survival outcomes [51]. Recently, nuclear factor erythroid 2-related factor 2 (NRF2) functioned as a stability factor for intermediate E/M cells. NRF2-EMT-NOTCH network signaling is spatially coordinated near the leading edge during collective cancer migration [52].
Another model currently predominates in explaining the origin of CSC is the stochastic model because CSC and non-CSC interconvert. Thus, CSC cannot be isolated by cell-sorting methods based on intrinsic features [53]. It is believed that cancer cells commonly sustain equilibria in the proportion of cells, and every cell owns an equal probability of initiating tumor growth [43, 54]. For instance, Wang et al. showed the existence of distinctive CSC populations and bidirectional inter-conversion between non-CSC and CSCs occurred stochastically [55]. Besides breast cancer, stochastic model can also apply to other solid and non-solid tumors, including lung adenocarcinomas and lymphoblastic leukemias [56-58]. Like the hierarchical model, the stochastic model is composed of retro-differentiating cancer cells into stem-like cancer cells [59]. However, the stochastic model primarily addresses genetic heterogeneity without consideration of potential phenotypic variations within the genetically homogenous tumor cell population.
Genetic and epigenetic alterations and abnormal activation of signaling pathways can promote the malignant transformation of normal stem cells. Human stem cells with the surface marker phenotype Lin-CD10-CD24-PROCR+CD44+ were identified in normal mammary epithelium and breast carcinomas. Notably, both PROCR and CD44 are target genes of the Wnt pathway [5, 60]. The altered Hippo signaling pathway also confers self-renewal and metastatic ability to BCSCs. The signaling effectors transcriptional co-activator with PDZ-binding motif (TAZ) and yes-associated protein (YAP) have been shown to bind to the promoters of mammary stem cell signature genes to induce BCSCs [61, 62], and high expression of TAZ was detected in CD44+CD24- BCSCs [63]. YAP/TAZ amplification correlates with the poor prognostic outcome and increased therapeutic resistance [64]. In addition, gene fusions were found in TAZ, NF2, and LATS1/2 in lung cancer [65], and TAZ-CAMTA fusion or YAP-TFE3 fusion is proved to be the initiating mutation in a vascular cancer [66]. These gene fusions induce the Hippo pathway hyperactive and drive tumor initiation and proliferation in vivo [67].
In addition, several studies have reported that some mammary epithelial cells in a dormant state might give rise to BCSCs under specific circumstances. For example, Guo et al. found that co-expression of SRY-box transcription factor 9 (SOX9) and SLUG in differentiated luminal cells was sufficient to induce stem cell-like properties, such as EMT activation and metastasis-seeding ability [68]. Similarly, Dravis et al. found that the binding of SOX10 to genes related to EMT or to genes that regulate neural crest cell identity promoted the stem-like features of mammary tumor cells [69]. Recent studies employing single-cell sequencing with chromatin accessibility indicated that, due to epigenetic regulation, some epithelial cells expressed both luminal and basal signature genes within the mammary epithelium, due to epigenetic regulation, which might explain the multipotent capacity of basal cells observed upon transplantation [70].
CSCs in tumors exhibit very heterogeneous metabolic states, and each CSC has an adaptable metabolism. Somatic stem cells [71], embryonic stem cells [72, 73], and induced pluripotent stem cells [74] are all reported to increase their glycolysis activity to maintain their stem cell features. Angela et al. compared the CSCs to the parental and benign precursor cells and found that CSCs with increasingly glycolytic phenotypes are more adaptable to specific microenvironmental conditions [75]. Luo et al. illustrated how metabolic or oxidative stress modulated the BCSC dynamic state. The transition between mesenchymal-like state and epithelial-like state relied on redox metabolism change. For epithelial-like BCSCs, they exhibit strong antioxidant capacity due to NRF2 hyperactivity. Oxidative stress transited epithelial-like BCSCs to mesenchymal-like state [76]. Patricia et al. also observed similar phenomenon in pancreatic cancer, supporting that some CSC populations are dependent on oxidative metabolism [77].
BCSCs are involved in multiple biological behaviors of breast cancer
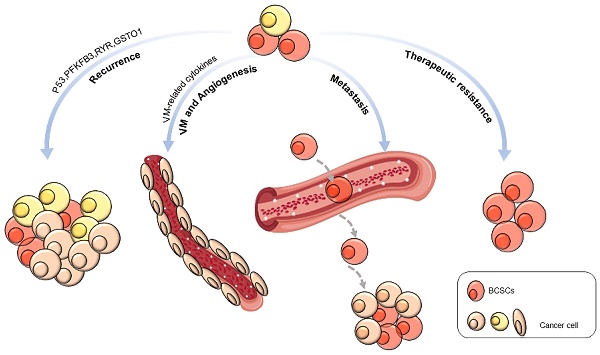
BCSCs are engaged in the physical behaviors of cancer, including recurrence, metastasis, vasculogenic mimicry, angiogenesis, and therapeutic resistance (Figure 1).
Biological behaviors of BCSCs.
Tumor recurrence
Emerging evidence suggests that BCSCs promote tumor recurrence, leading to poor prognosis [78]. The silencing of p53 promotes the division of BCSCs, increasing their renewal and contributing to tumor recurrence [79]. Inhibition of ryanodine receptor (RyR1) and glutathione S-transferase omega 1 (GSTO1) expression cleared off chemotherapy derivational BCSC enrichment and postponed cancer recurrence [80]. Abnormal expression of 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 (PFKFB3), a gene associated with tumor recurrence, can be suppressed by autophagy activation, keeping BCSCs in a resting state [81].
Vasculogenic mimicry and angiogenesis
Recent studies indicated that BCSCs might contribute to tumor-associated angiogenesis by VM or trans-differentiation [82]. For example, Bussolati et al. isolated CD24-/CD44+ BCSCs and cultured them to differentiate into an endothelial lineage with endothelial markers and properties in the presence of vascular endothelial growth factor (VEGF). They also observed intratumor vessels of human origin in transplanted tumors [83]. Using a 3D reconstructed image, Sun et al. provided direct evidence of the relationship between BCSCs and VM formation. The CD133+ BCSCs were observed to line VM channels, and breast cancer cells encircled VM channels. They also observed a close correlation between BCSC proportion and VM in invasive breast cancer [84]. In addition to differentiation into endothelial cells, BCSCs might provide VM-related cytokines, such as Nodal protein, to support angiogenesis [85]. Tumor endothelial marker 8 (TEM8) is highly correlated with VM, and TEM8 coincidentally induces VM and promotes stemness through the Ras homolog family member C (RhoC)/rho associated coiled-coil containing protein kinase 1 (ROCK1)/smad family member 5 (SMAD5) axis [86].
Tumor metastasis
CSCs are closely related to tumor metastasis [87]. BCSCs are more metastatic than other types of breast cancer cells, which was realized by increasing the expression of cell metastasis and migration-related proteins while significantly reducing the level of adhesion proteins [88]. In BCSCs, a subset of the cell population expressing CD44v and epithelial splicing regulatory protein 1 (ESRP1) has a stronger lung metastasis ability [89]. Six homeobox 2 (Six2) promotes the expression of SRY-box transcription factor 2 (SOX2) and NANOG, which induces stem cell characteristics and increases TNBC metastasis [90]. The inflammatory cytokine interleukin-8 (IL8) promotes BCSC metastasis by activating C-X-C motif chemokine receptor (CXCR1/2) [91]. Transforming growth factor beta 1 (TGF-β1) significantly increases the number of BCSCs in MDA-MB-231 cells and promotes breast cancer metastasis to the liver [92]. C-C motif chemokine ligand 2 (CCL2) enhances CSC self-renewal and expansion through the signal transducer and activator of transcription 3 (STAT3) and neurogenic locus notch homolog protein 1 (NOTCH-1) signaling pathways and promotes breast tumor growth and metastasis [93, 94]. Stromal cell-derived factor-1 (SDF-1/CXCL12) activates the nuclear factor-κB (NF-κB) pathway to increase the proportion of BCSCs and promote the metastasis of MCF-7 cells [95]. Consistently, Shan et al. showed that CXCL12 overexpression induced MCF-7 cells to form a BCSC phenotype through the Wnt/β-catenin pathway, thereby enhancing metastasis [96].
Inactivation of tumor suppressor genes and activation of oncogenes also regulate the metastatic potential of breast cancer stem cells. Knockout of p21 in a breast tumor model inhibited the self-renewal and lung metastasis of BCSCs [97]. Silencing AKT serine/threonine kinase 2 (AKT2) reduces the invasion and colony formation abilities of BCSCs, which are mediated by the Twist/mTOR signaling axis [98]. Inhibition of RhoC dramatically reduces the probability of lung metastasis of SUM149 [99]. Extracellular signal-regulated kinase 2 (ERK2) promotes BCSC self-renewal and lung metastasis, and knockout of ERK2 significantly inhibits colony formation and mammosphere formation [100]. Deletion of sirtuin 1 (SIRT1) accelerates the degradation of PRRX1 and sequentially activates the transcription of krüppel-like factor 4 (KLF4) and aldehyde dehydrogenase 1 (ALDH1), thereby inducing BCSCs and lung metastasis [101]. GD3 synthase (ST8SIA1) is highly expressed in BCSCs, and inhibiting this synthase reduces tumor growth and metastasis by eliminating BCSCs [102]. OTU deubiquitinase 7B (OTUD7B) deubiquitinates lysine-specific demethylase 1 (LSD1) to decrease histone H3 lysine 4 (H3K4)/H3K9 methylation, thereby sustaining breast cancer metastasis potential [103].
Overexpression of miR-7 in BCSCs decreases endothelial cell adhesion molecule (ESAM) expression by targeting RelA and inhibits tumorigenesis and distant lung metastasis [104]. miRNA-628 directly targets SOS Ras/Rac guanine nucleotide exchange factor 1 (SOS1) to inhibit the migration and invasion of BCSCs [105]. Specifically, hypoxia-mediated upregulation of miRNA-210 induces BCSC migration by blocking E-cadherin expression [106].
LncRNA MALAT-1 is upregulated in BCSCs, and it promotes self-renewal, migration, and invasion of BCSCs by regulating SOX2 [107]. Linc-ROR promotes the proliferation and invasion of BCSCs by inhibiting the abnormal overexpression of critical factors such as SMAD family member 2 (SMAD2) and alpha-smooth muscle actin (α-SMA) in the TGF-β signaling pathway [108]. LncRNA NR2F1-NAS1 binds to the 5'-UTR of nuclear receptor subfamily 2 group F member 1 (NR2F1) and recruits polypyrimidine tract binding protein 1 (PTBP1) to enhance the translation of NR2F1. The latter inhibits the transcription of ΔNp63, thereby inhibiting the MET of BCSCs and ultimately inducing the metastatic dormancy of cancer cells [109].
Therapeutic resistance
Current studies have shown that BCSCs are one of the main reasons for radiation resistance and chemotherapy resistance. Yang et al. reported that aurora kinase A (AURKA) promotes forkhead box M1 (FOXM1) transcription, self-renewal, and drug resistance in BCSCs [110]. The deubiquitinase ubiquitin specific peptidase 28 (USP28) can stabilize LSD1 to promote stemness and drug resistance in breast cancer cells [111]. Metformin induces miR-708 expression in BCSCs and increases the chemotherapy sensitivity of BCSCs [112]. Signal-induced proliferation-related protein 1 (SIPA1) promotes the expression of stem cell-related transcription factors, such as octamer-binding transcription factor-4 (OCT4), NANOG, SOX2, and B lymphoma Mo-MLV insertion region one homolog (BMI-1), by increasing the expression of SMAD2/3, resulting in chemotherapy resistance [113].
The drug resistance of BCSCs is mainly caused by the expression of transporters that cause drug efflux and the expression of high-level ALDH1 as a detoxification enzyme to metabolize anticancer substances. In addition, BCSCs have a strong DNA damage repair ability and show radiation resistance through high expression of related stemness genes and activation of antiapoptotic and antioxidant signaling pathways [114, 115].
ALDH is closely related to BCSC therapeutic resistance. One of the reasons is that it can inactivate the metabolism of chemotherapy drugs such as cyclophosphamide [78]. ALDH can produce nicotinamide to achieve antioxidant function [116]. Overexpression of NANOG enhances ALDH activity by activating the NOTCH-1 and AKT pathways, thereby inducing radiation resistance [117]. The drug resistance of BCSCs is also driven by a high mitochondrial quality caused by ALDH activity [116].
In addition, Hippo signaling is emphasized to play a key role in mediating therapeutic resistance. For example, the hyperactivation of YAP and TAZ is indicative of resistance to paclitaxel, lapatinib, doxorubicin, or CDK4/6 inhibitors [118-120]. Cysteine-rich protein 61 (Cyr61) and connective tissue growth factor (CTGF) are the transcriptional targets of TAZ/ transcriptional enhanced associate domain (TEAD). Evidence showed that they also confer paclitaxel resistance to breast cells and inhibit Cyr61 and CTGF by shRNA re-sensitized breast cancer cells to paclitaxel. Macrophage stimulating (MST) as an important component of Hippo signaling is responsible for YAP and TAZ phosphorylation and degradation. Low expression of MST protein is associated with poor prognosis in breast cancer. Pauliina et al. revealed that phosphorylation of MST1 by fibroblast growth factor receptor 4 (FGFR4) helped cell evade from mitochondrial apoptosis and become resistant to HER2/epidermal growth factor receptor (EGFR), AKT, or mTOR inhibitors [121].
The resistance of BCSCs to radiotherapy may be caused by apoptosis reduction and DNA damage checkpoint activation. Double-strand breaks in DNA damage are mainly repaired by homologous recombination (HR) or nonhomologous end-joining (NHEJ) [78, 122]. Jiao et al. showed that C-C motif chemokine receptor 5 (CCR5) enhanced DNA repair in breast cancer after chemotherapy [123]. The activities of checkpoint kinase 1 (CHK1) and CHK2 are also improved in BCSCs to avoid mitotic catastrophe and to repair damaged DNA [124]. By activating the phosphatidylinositol 3-kinase (PI3K) and NRF2 signaling pathways, BCSCs are more resistant to drug- or radiation-induced apoptosis [125]. Activated PI3K/AKT downregulates forkhead box O3 (FOXO3a) expression levels and enhances breast cancer stemness and therapeutic resistance [126]. The transcription factor NRF2 has a high expression level in BCSCs to maintain a relatively low level of ROS [127]. Sequestosome 1 (SQSTM1/p62) activates NRF2 expression to overcome the drug resistance of BCSCs [128, 129]. Up-regulation of the expression level of nuclear-Dbf2-related 1 (NRD1), a component of the Hippo pathway will increase the proportion of BCSCs, thereby resisting the lethal effect of drugs [130].
The tumor microenvironment (TME) regulates BCSCs
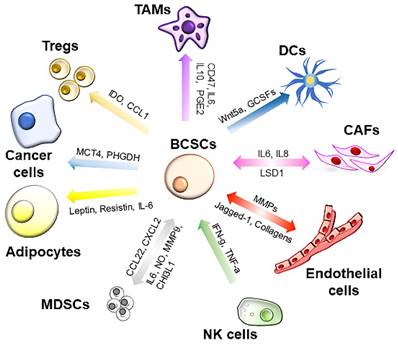
The TME consists of noncellular components, such as extracellular matrix (ECM) nutrients, metabolites and cytokines, and cellular components, including fibroblasts, adipocytes, immune cells and endothelial cells. All these components exhibit dynamic changes during cancer progression and are associated with cancer stemness (Figure 2).
Interplays between tumor microenvironment (TME) and BCSCs. The TME is a complex network consisting of cellular and noncellular components. BCSCs interact with the TME through cytokines in a paracrine manner or through direct interactions. The cellular components in the TME secrete various cytokines/chemokines to support the self-renewal of BCSCs and help BCSCs escape immune attack. DCs: dendritic cells; TAMs: tumor-associated macrophages; MDSCs: myeloid-derived suppressor cells; CAFs: cancer-associated fibroblasts; MMPs: matrix metalloproteinases; GCSFs: granulocyte colony-stimulating factor; SCF: stem cell factor; PHGDH: phosphoglycerate dehydrogenase; MCT4: monocarboxylate transporter-4.
Hypoxia
Hypoxia is a well-known niche for CSCs. Several proteases, including serine proteases, thrombin, and matrix metalloproteinases, are activated by the acidic microenvironment around hypoxic cells and promote cancer metastasis [131-133]. For example, cells with high expressions of platelet-derived growth factor (PDGF)-D and hypoxia inducible factor 1 subunit alpha (HIF-1α) exhibited more aggressive phenotypes by increasing matriptase activation [134]. Thrombin was reported to be closely related to VM formation and spontaneous metastases in tumors [135]. Jewer et al. discovered various transcript isoforms of NANOG, SNAIL, and Nodal. When cancer cells are under hypoxic conditions, they prefer to translate these variants to facilitate protein expression and acquire a stem cell phenotype [136]. Consistently, remarkably elevated SOX2, OCT4, KLF4, and NANOG expression levels were observed after hypoxia [137, 138]. The sphere formation rate under hypoxia was much higher than normal culture conditions [138]. The enrichment of BCSCs was also observed in hypoxic tumors in vivo [139]. Hypoxia-inducible factors (HIFs) play a master role in regulating malignant phenotypes. The impact of HIFs on BCSCs is mainly due to HIF-dependent hyperactivation of pluripotent factors or EMT-TFs [137]. For example, Zhang et al. reported that m6A-demethylation of NANOG mRNA by HIFs, including HIF-1α and HIF-2α, significantly increased NANOG mRNA and protein expression, which further enhanced the BCSC phenotype under hypoxia [140]. HIF-1α also causes abnormal nuclear translocation of FOXO3 and transcriptional activation of NANOG, which increases chemotherapy-enriched BCSCs [141]. Hypoxia induces a metabolic energy change in stem cells and thus enhances stemness. It is proposed that stem cells have a unique metabolism to protect themselves from oxidant exposure. For example, upregulation of monocarboxylate transporter 4 (MCT-4) induced by hypoxia facilitates a stem-like feature of cancer cells by changing the acidic pH with increased lactic acid efflux [142]. Zhu et al. identified a hypoxia-induced lncRNA KB-1980E6.3 closely related to poor prognosis, and upregulated expression of lncRNA promoted the stemness of breast cancer cells [143].
Cytokines
There are two types of cytokines: pro-inflammatory and anti-inflammatory. Pro-inflammatory cytokines, such as IL1, IL6 and IL8 are involved in forming an inflammatory milieu and shaping pre-metastatic niches. While anti-inflammatory cytokines, including IL4, IL6, IL10, and IL13, etc., are released to prevent sustained or excessive inflammatory reactions [144, 145]. Both pro-inflammatory cytokines and anti-inflammatory cytokines are closely related to BCSCs, and they show synergistic effects in drug resistance [146]. In breast cancer, the induction of IL1α secretion triggers a proinflammatory environment to maintain CSCs [147]. As previously mentioned, IL8 and IL6 maintain the characteristics of BCSCs by regulating CXCR1/2 and STAT3, respectively [91, 148]. IL6 secreted by TAMs activates the JAK/STAT3 pathway to induce CSC enrichment and promote tumor growth [149]. Blocking the NF-κB/IL8 pathway attenuates BCSC activity [150]. Similarly, IL10 plays an important role in maintaining BCSCs, and the proliferation and self-renewal of BCSCs are blocked by inhibiting IL10 [151]. Shi and colleagues reported that the proportion of BCSCs is reduced by partial inhibition of KLF5/fibroblast growth factor-binding protein 1 (FGF-BP1) [152]. CCL20 not only facilitates the expansion of BCSCs but also enhances drug resistance in TNBC [153]. C-C Motif Chemokine Ligand 3 (CCL3) from cancer cells and macrophages enhances the phagocytic ability of docetaxel-induced M1 macrophages to BCSCs [154]. Blocking the C-X-C motif chemokine ligand-1 (CXCL1) produced by BCSCs impedes BCSC proliferation and mammosphere formation [155]. Type I interferon (IFN-I) is related to cancer cell stemness, and the expression of ALDH1A1 is increased when IFN-I signal transduction is destroyed [156].
Cells in the tumor microenvironment
Carcinoma-associated fibroblasts (CAFs)
The interaction between fibroblasts and cancer cells was first observed by pathologists who noticed that the expansion of fibroblasts in the tumor context increased the levels of collagen and the abnormal expression of α-smooth muscle actin (αSMA), a phenomenon called desmoplasia [157]. All fibroblasts within the TME, including normal fibroblasts and actively proliferating fibroblasts, are CAFs. Because there are functionally heterogeneous CAF groups, studies suggest a bimodal influence of CAFs on tumor progression, with both cancer-advancing and cancer-constraining effects [158, 159]. Notably, tumor fibrosis occurs at the beginning of carcinogenesis, although most cancer cells arise from the epithelium. Such fibrosis in the early stage induces a chronic proinflammatory environment and directly impacts epithelial cell transformation.
Proinflammatory cytokines produced by CAFs, such as interleukin (IL) 1β and leukemia inhibitory factor (LIF), promote the inflammatory reaction in TME [160, 161]. By secreting proinflammatory cytokines and proinflammatory paracrine factors, CAFs actively cross-talk with CSCs, foster the dedifferentiation of cancer cells, and support the self-renewal of CSCs [162]. CAFs are also the major source for immune suppression in the TME based on the observations that CAFs hamper the recruitment of T cells to the TME and secrete immunosuppressive chemokines to promote M2 macrophages [163-165].
Activation of the IL6/STAT3/ NF-κB signaling pathway in HER2-positive PTEN-depleted breast cancer cells induces an increase in the CSC population. Inhibition of IL6- or IL8-secreting CAFs slows the growth of BCSCs [166]. Recently, exosomes secreted by CAFs were reported to promote cancer cell transformation to a metastatic and tumor-initiating phenotype [167, 168]. miRNAs, such as miR-21-5p, miR-143-3p and miR-378e, contained in exosomes of CAFs facilitated the EMT phenotype and dedifferentiation of breast cancer cells with increased expression of SOX2 and NANOG [169]. In addition to secretory proteins and exosomes, CAFs also regulate the stemness of cancer cells via cell-cell interactions. For example, CD44 expressed on CAFs can promote the secretion of SDF-1, further motivating the stemness of CSCs [170]. CD10+GPR77+ CAFs have been identified as a protumorigenic subpopulation that maintains the stem niche of CSCs [171].
Adipocytes
As the primary component in breast tissue, an increasing amount of evidence has shown that adipocytes enhance tumor malignancy by releasing inflammatory factors, metabolites, and exosomes. Indeed, the Body Mass Index (BMI) of women with breast cancer is higher than that of the general population at the time of diagnosis, and patients might benefit from modest weight loss after diagnosis [172]. Several studies have shown that adipocytes in the microenvironment of invasive breast cancer are distinguished by their phenotypes, including smaller size and fibroblast-like shape, and molecular markers, such as collagen VI overexpression and low adiponectin expression. Thus, adipocytes that are adjacent to or communicate with cancer cells are defined as cancer-associated adipocytes (CAAs) [173, 174]. CAAs have been proven to secrete more chemokines, including CCL2, CCL5 and IL6, to enhance the metastasis, stemness, angiogenesis, and proliferation of breast cancer cells [175]. For example, CCL2 secreted by CAAs increased the recruitment and activation of macrophages in breast tissues to accelerate oncogenesis and angiogenesis [176]. Adipocyte-secreted IL6 promoted the self-renewal of BCSCs and stimulated the invasion of cancer cells in an adipocyte/breast cancer cell coculture system [174, 177].
Multiple hormones, such as leptin, resistin and adiponectin, which are abnormally secreted by CAAs, are also responsible for the enhanced stemness of breast cancer. Bowers et al. reduced the expression of the leptin receptor (LEPR) in breast cancer cell lines and observed dramatically decreased expression of stem cell markers [178]. Thiagarajan et al. further proved that leptin not only promoted BCSC survival by phosphorylating STAT3 but also transformed non-BCSCs into stem-like cells by binding to its receptor LEPR and inducing the expression of NANOG, SOX2 and OCT4 in TNBC [179]. In addition, CAAs were reported to promote metastasis of TNBC via leptin signaling in vitro and in a PDX model [180]. In the obese state, TAZ-dependent resistin expression was able to promote breast tumorigenesis [181]. Furthermore, the high estrone (E1): estradiol (E2) ratio was proposed to facilitate tumor stemness properties in obese patients by activating NF-κB signaling [182]. In contrast, circulating adiponectin, as a starvation hormone, was indicated to induce apoptosis and inhibit tumor cell proliferation, invasion and migration [183, 184].
The mechanisms by which obesity-induced dysfunctional adipocytes contribute to breast cancer stemness have not been clearly elucidated. Gao et al. reported that the transcription factor TAZ in adipocytes played an important role in upregulating cytokine secretion. TAZ knockdown or deficiency also impaired the tumor-supporting function of CAAs [181]. Recently, Liu et al. demonstrated that cellular adaptation instead of expansion of preexisting clones is the primary driver responsible for obesity-related tumor formation. They discovered that palmitic acid, a metabolite of CAAs, enhanced the tumor initiation of breast cancer cells in a transcription factor CCAAT/enhancer-binding protein beta (C/EBPB)-dependent manner [185]. In addition to cytokines and metabolites, CAAs have been reported to promote BCSCs by metabolic reprogramming. Dai et al. proved that elevated CAA-derived fatty acids fueled the stemness of breast cancer via the fatty acid oxidation (FAO)-AMP-activated protein kinase (AMPK)-YAP signaling axis. As a ROS sensor, YAP is induced by conditioned CAAs in cancer cells and sustains mitochondrial redox homeostasis [186].
Endothelial cells
CSCs prefer to be located near the vasculature due to the conveniences of migration and nutrition acquisition. The recent discovery that BCSCs tend to gather with the arteriolar niche in ER-positive breast cancer demonstrated a bidirectional interaction driven by lysophosphatidic acid (LPA)/protein kinase D (PKD-1) signaling between BCSCs and endothelial cells [22]. The crosstalk between tumor cells and endothelial cells is essential for tumor angiogenesis. For example, cancer cells communicate with endothelial cells via cell adhesion. By gap junctions or adhesion receptors, they exchange ions and small metabolites to meet the demands for tumor proliferation [187]. By analyzing single-cell RNA sequencing and protein expression profiles of primary tumor cells and lung metastases of TNBC, Rokana et al. found that the expression level of intercellular adhesion molecule 1 (ICAM1) was increased 200-fold in lung metastases. Further examination revealed that tumor cells employed ICAM1 to connect with endothelial or tumor cells to form cell clusters. ICAM1 promotes tumor cell stemness and transendothelial migration [188]. Myc target protein 1 (Myct1), which is almost explicitly expressed in endothelial cells, interacts with the tight junction protein zona occludens 1 (ZO1) of cancer cells to promote a unique tumor niche for cancer angiogenesis and anti-immunity [189].
Meanwhile, the endothelial cells of the vasculature secrete soluble factors to support the growth and self-renewal of CSCs and promote drug resistance. Soluble factors such as VEGFs tend to bind with tyrosine kinase receptors and thus induce tumor cell proliferation or migration [190, 191]. During progression, cancer cells in hypoxic regions recruit endothelial cells to build new vessels for oxygen and nutrition. These endothelial cells employ collagens to reconstruct the extracellular matrix and the interconnection of cancer cells. Cancer cells and stromal cells also secrete VEGF into the microenvironment to drive angiogenesis [192]. Liu et al. observed that CD133 expression was closely related to VM in different breast cancer subtypes, especially TNBC. Further study confirmed that CD133-positive MDA-MB-231 cells could form tubular structures and express VE-cadherin, MMP-2, and MMP-9.
In addition, cancer cells might mimic the embryonic vasculogenesis process during metastasis. The vessel-like structures are favorable for periodic acid-Schiff (PAS) staining and negative for CD31. Notably, the VM showed perfusion capacity, and erythrocytes were found inside the VM, suggesting that they might support tumor cells in hypoxia by transporting nutrients and oxygen. In breast cancer, ALDH1 expression was associated with the formation of VM, especially in TNBC [193]. For example, ALDH1A3+ HCC1937 cells with inducible p53 transfection showed a high capacity to form tubular structures when cultured in Matrigel, while ALDH1A3- cells failed to create such structures. ALDH1A3+ cells also exhibit characteristics of CSCs and are resistant to p53-induced apoptosis [194]. Xu et al. reported that the endothelial marker TEM8 was highly expressed in BCSCs and that TEM8+ breast cancer cells represented a kind of special BCSC to initiate VM [86]. Tiara et al. developed a TEM8-specific CAR-T immunotherapeutic strategy to target TEM8+ endothelial cells and TNBC cells. They found that TEM8+ endothelial cells were killed and neovascularization was blocked and CD24-/CD44+ BCSC numbers were reduced, offering preclinical proof for immunotherapeutic targeting of tumor vascularization [195]. However, we cannot determine whether there is a single subtype of BCSCs in tumors or whether BCSC subpopulations are involved because the marker of CSCs involved in VM formation varies in different studies.
Endothelial cells also support BCSCs independent of their vascular functions. Pegah et al. found that the NOTCH ligand Jagged1 was secreted by endothelial cells and activated the NOTCH pathway in BCSCs; thus, endothelial cells conferred a survival advantage and metastatic potential for BCSCs. Esak et al. cocultured MDA-MB-231 cells with lymphatic endothelial cells (LECs), microvascular endothelial cells (MECs), or human umbilical vein endothelial cells (HUVECs) and found that only LECs supported tumor cell growth. They discovered that tumor-educated LECs secreted amounts of epidermal growth factor (EGF) and PDGF-BB to promote tumor growth [196].
Immune cells
The interaction between the host immune system and cancer progression consists of three stages: clearance, balance, and escape. During the balance phase, CSCs might be the first batch of tumor cells that escape immune surveillance. Benefiting from a long quiescent stage, CSCs are slippery and rarely targeted by the immune system even when they enter the bloodstream and are surrounded by immune cells. In addition, BCSCs express low levels of MHC class I molecules and defects in antigen processing to escape immune cell killing [197, 198]. BCSCs also secrete cytokines to suppress the activation of immune cells, including TGF-β, IL4, IL10 and IL33 [199, 200]. Jiang et al. employed single-cell analysis to uncover the dynamic change in immunity for heterogeneous BCSCs during tumor progression. They found that C-X-C motif chemokine ligand-16 (CXCL16) and CXCL1 were highly expressed in one of five BCSC clusters. Their corresponding receptors, CXCR2 and C-X-C motif chemokine receptor 6 (CXCR6), were also abundant in macrophages and T cells, respectively, suggesting communication between BCSCs and immune cells [201].
A large number of studies has reported the association between TAMs and poor clinical outcome. CSCs in breast cancer escape innate immune surveillance by upregulating the expression of CD47 [202]. Cancer stem cells promote macrophage M2 differentiation, which has protumoral and immune-suppressive functions, while the interaction between CSCs and macrophages elevates ALDH activity and chemoresistance [203, 204]. Targeting colony stimulating factor 1 receptor (CSF1R) or C-C motif chemokine receptor 2 (CCR2) relieved immunosuppression, decreases the proportion of CSCs, and sensitizes tumors to chemotherapy and immunotherapy [205-207].
MDSCs are a heterogeneous population of immunosuppressive cells. The high number of MDSCs in circulation is related to tumor progression, including metastasis. By depleting required amino acids, MDSCs restrict T-cell proliferation and suppress T-cell function [208]. Wei et al. observed a linear correlation between MDSC expansion and large tumor size [209]. MDSCs also confer cancer cells with stem-like characteristics and EMT through IL6- and NO-mediated STAT3 and NOTCH pathway activation [210]. Kumar et al. observed that TNBC cells recruited MDSCs via ΔNp63-dependent secretion of CXCL2 and CCL22. Meanwhile, recruited MDSCs secreted MMP-9 and chitinase 3-like 1 to promote TNBC stemness [211].
Although the infiltration of T cells is a favorable index of chemotherapy response and patient survival, several studies revealed that Tregs promote BCSCs. Roland et al. observed that CD8+ T cells upregulated the expression of stemness genes and immune checkpoint genes [212]. Cells that preferentially exclude DNA binding dye Hoechst 33342 are more capable of initiating tumors and were called side population according to its unique pattern on fluorescence-activated cell sorting (FACS) analysis [213]. It was reported that Foxp3+ Tregs increased the proportions of side populations and ALDH+ BCSCs in three mouse breast tumor cell lines [214]. They also discovered that SOX2 promoted the expression of p65 and CCL1 in BCSCs to recruit Tregs [214]. Indoleamine 2,3-dioxygenase (IDO) is an enzyme responsible for the degradation of the essential amino acid tryptophan and is highly expressed in BCSCs [215]. IDO secreted by BCSCs suppresses cytotoxic T-cell expansion and promotes Treg activation [216]. Nevertheless, different T-cell subsets recruited by BCSCs facilitate them maintenance of an immunosuppressive microenvironment.
Strategies targeting BCSCs
Natural compounds and their products
Nature is a treasure trove of natural compounds produced quantitatively through synthetic biology. It is a candidate drug for treating BCSCs (Table 1). Baicalein (5,6,7-trihydroxy-2-phenyl-4H-1-benzopyran-4-one) is an active ingredient of the roots of Scutellaria baicalensis Georgi. Baicalein reverses the drug resistance of MDA-MB-231/IR cells by upregulating interferon induced protein with tetratricopeptide repeats 2 (IFIT2) to induce the apoptosis of BCSCs [217]. Phenethyl isothiocyanate (PEITC) can eliminate CSCs in MDA-MB-231/IR cells by reducing glutathione levels and promoting ROS accumulation and significantly inhibited the formation of mammospheres [218]. Psoralidin, an active compound extracted from Psoralea corylifolia, targets NOTCH-1 in BCSCs and inhibits EMT and breast tumor growth [219].
Natural pharmaceutical products targeting BCSCs
| Names | Target | Effects of a model | Ref. |
|---|---|---|---|
| Triterpene acid | C-MYC | Inhibits MDA-MB-231 stemness and the formation of mammospheres. | [220] |
| Gomisin M2 | Wnt/β-catenin | Decreases the proliferation of TNBCs and mammosphere formation. | [221] |
| Honokiol | STAT3 | Reduces of secondary transplanted tumors and the prolongation of metastasis time of MDA-MB-231 xenografts. | [222] |
| Apigenin | YAP/TAZ-TEAD | Inhibits TNBC stemness, migration, and tumor growth. | [223] |
| Andrographolide | Survivin | Inhibits the activity of human BCSCs in a dose-dependent manner and reduces the formation of mammospheres. | [224] |
| Ellagic acid | Actinin alpha 4 and β-catenin | Inhibits MDA-MB-231 and BT549 cell proliferation and prolongs the survival time of MMTV-PyMT mice. | [225] |
| 8-Hydroxydaidzein | JAK2 and STAT3 | Decreases BCSC characteristics and triggers apoptosis. | [226] |
| Sulforaphane | Wnt/β-catenin | Eliminates BCSCs and abrogates tumor growth. | [227] |
| Withaferin A | BMI-1 | Attenuates BCSCs and inhibits breast tumor burden. | [228] |
| Benzyl isothiocyanate | Ron receptor tyrosine kinase | Inhibits the self-renewal ability of BCSCs. | [229] |
| Glabriden | TGF-β/SMAD2 | Attenuates the breast CSC-like properties. | [230] |
| Isoliquiritigenin | DNA methyltransferase 1 (DNMT1) | Reduces the BCSC-like population and suppresses breast cancer initiation and progression. | [231] |
| Caffeic Acid Phenethyl Ester | CD44 | Interfering with the growth of TNBC CSCs. | [232] |
Antibody-based biopharmaceuticals
Antibodies have therapeutic effects on cancer by highly specifically targeting tumor cell surface antigens and immune cells, but their killing in tumor cells is limited [233]. Because of its wide, “off-the-shelf” availability to become promising biopharmaceuticals [234]. mAb4C5 is a monoclonal antibody that inhibits the colony formation of MDA-MB-231-derived BCSCs and reduces primary tumor growth by binding to extracellular heat shock protein 90 (HSP90) [235]. The monoclonal antibody 602.101 specifically recognizes NOTCH-1, inhibits the expression of downstream target genes, reduces the MDA-MB-231 cancer stem-like cell subpopulation, inhibits the efficiency of mammosphere formation, and induces apoptosis [236]. Monoclonal antibody J1-65D against NOTCH ligand jagged 1 (JAG1) reduces MDA-MB-231 BCSC numbers and breast tumor growth in a xenograft model by blocking NOTCH signaling [237]. In MDA-MB-231 and T47D cells, the CD47 antibody B6H12 significantly downregulates KLF4 levels and negatively regulates EGFR phosphorylation to prevent BCSC proliferation, thereby inhibiting tumor growth [238]. Dinutuximab, an anti-GD2 antibody, inhibits BCSC function and induces TNBC apoptosis via antibody-dependent cell-mediated cytotoxicity [239]. The rhoptry protein 1 (ROP1)-specific antibody cirmtuzumab effectively attenuates chemotherapy-resistant BCSCs by reverting stemness [240]. An anti-Cadherin 11 (CDH11) monoclonal antibody attenuates CSC-like properties and breast cell metastasis by upregulating miRNA-335 [241]. A single-domain antibody can block the interaction of PROCR with its ligands, effectively reducing BCSCs without tumor recurrence [242]. John et al. synthesized antihuman CD133 scFv-PE38KDEL to kill CSCs and inhibit tumor growth in breast cancer [243]. Not unique but has a double, CD133-targeted polymeric nanoparticles effectively impair the mammosphere formation ability of CSCs via conjugating anti-CD133 monoclonal antibody [244].
Cytotherapy
The efficacy of cytotherapy in removing malignant tumor cells is encouraging and can prolong the survival of patients. Its biggest advantage is that enhances or alters intrinsic immune capacity, but there are fatal adverse reactions [245, 246]. Third-generation cytotherapy is a combination of dendritic cells and cytokine-induced killer cells (DC-CIKs). Chen et al. reported that total RNA-loaded DC-CIK immunotherapy is extremely effective in counteracting BCSCs [247]. TEM8 CAR-T can kill BCSCs, inhibit mammosphere formation, eliminate tumor angiogenesis, and induce PDX regression [195]. GD2 has also been identified as a BCSC-related marker [248]. GD2-CAR-T immunotherapy has site-specific activation, which can eliminate BCSCs and prevent the formation of metastasis [249].
Synthetic small molecule compounds
Small molecule compounds have significant advantages such as the possibility of oral administration, stability, membrane permeability and non-immunogenicity in spite of off-target toxicity. Its convenient production has higher expected benefits [250-252]. Currently, several synthetic compounds inhibit BCSCs (Table 2). Palbociclib is an oral biologically effective second-generation CDK4/6 inhibitor approved by the FDA that induces cell cycle arrest in breast cancer. Palbociclib inhibits MCF-7 BCSCs and effectively slows the formation of 3D spheroids [253]. Protein arginine methyltransferase 5 (PRMT5) was reported to stabilize KLF4/5 proteins in breast cancer [254, 255]. WX2-43 specifically blocked PRMT5-mediated KLF4 methylation and inhibited MDA-MB-231 BCSC activity [256]. Consistently, the PRMT5 inhibitor PJ-68 efficiently promotes KLF5 degradation and inhibits stemness in BLBC [255]. Ladademstat, an LSD1 inhibitor, blocks SOX2 activation, thus significantly reducing the number of mammospheres [257].
Synthetic compounds targeting BCSCs
| Name | Targets | Effect of the model | Ref. |
|---|---|---|---|
| WX2-43 | Blocks PRMT5-mediated KLF4 methylation | Inhibits MDA-MB-231 BCSC activity. | [256] |
| PJ-68 | Promotes KLF5 degradation | Inhibits stemness in BLBC. | [255] |
| B591 | mTOR | Reduces the self-renewal ability of MCF-7, SUM159PT and MBA-MB-231 and tumorigenesis. | [258] |
| Pyrvinium pamoate | Cholesterol and fatty acid synthesis | Inhibits SUM159PT tumor growth and metastasis. | [259] |
| AZD1775 | Inhibits mucin 1 (MUC1) expression and cell cycle arrest | Reduces the BT474 CSC percentage and inhibits tumor growth. | [260] |
| Tannic acid | p65-IL6 | Decreases the formation of mammospheres in MCF-7, T47D and MDA-MB-231 cells. | [261] |
| Propofol | PD-L1 and NANOG | Decreases MCF-7 and MDA-MB-231 CSC mammosphere forming ability. | [262] |
| Dodecyl-TPP | mitochondria | Reduces cell viability and mammosphere formation. | [263] |
| Ferutinin analog | ERα | Induces apoptosis and the anti-proliferative activity of BCSCs. | [264] |
| Lx2-32c | FoxM1 and CD44 | Decreases MDA-MB-231 derived cancer stem cell-like characteristics. | [265] |
| 108600 | CK2/DYRK/TNIK | Has a curative effect on BCSCs. | [266] |
| Doxycycline | CD44 and ALDH1 | Likely extinguishes BCSCs. | [267] |
New uses of old drugs
With the increasing difficulty of drug development, new uses of conventional drugs have broad application prospects. It is highly safe and may reduce overall development costs and shorten development time, but its use in cancer is still affected by multi-drug resistance mechanisms [268]. The antilipemic agent lovastatin restores the sensitivity of triple-negative BCSCs to tyrosine kinase-targeted drugs by inducing HER2 expression and the stress response pathway [269]. Salinomycin decreases NANOG, OCT4, SOX2, and glioma-associated homolog-1 (GLI-1) expression levels to attenuate the BCSC ratio [270, 271]. Interestingly, the salinomycin derivative ionomycin also shows the ability to kill BCSCs [272]. Disulfiram, a drug for alcoholism, inhibits breast cancer cell stemness by upregulating miR-30a to target SOX4, thereby blocking the TGF-β/SMAD pathway [273]. As a first-line drug for diabetes, metformin partially reduces the percentage of TNBC stem cells through the PKA-GSK3β-KLF5 signaling pathway [152]. In addition, metformin kills MCF-7 stem cells, and its medicinal properties are improved by enhancing AMPK activation in combination with hyperthermia [274]. Mifepristone, an abortifacient, inhibits KLF5 expression by inducing the expression of miR-153, thereby reducing the TNBC stem cell population [275]. Furthermore, the mifepristone derivatives FZU-00,003 and FZU-00,004 exert anticancer activities [276, 277]. Flubendazole is a clinically anthelmintic that exhibits potential anti-tumor activity by reducing the expression of CD49f and ALDH1 in vitro and in vivo and killing BCSCs cells [278].
Nucleic acid medicines
Nucleic acid medicines have achieved a short reaction time, long-lasting therapeutic effect, minor side effects, and high specificity. Still, their shortcomings are lacking available genetic information and limited transmission to target organs and cells. Once these urgent problems are solved, chemical synthesis can significantly improve its application ability [279, 280]. Cripto-1 is the downstream target of NANOG and OCT4. The Cripto-1 encoding DNA vaccine can target BCSCs, reduce breast cancer metastasis and improve the survival rate [281]. Cystine-glutamic acid reverse transport protein xCT (SLC7A11) is overexpressed in BCSCs. The antibody produced by the anti-xCT virus vaccine directly impaired BCSCs and weakened tumor growth and metastasis [282]. The complex composed of an RNA aptamer targeting epithelial cell adhesion molecules and survivin siRNA was delivered to BCSCs to induce apoptosis, inhibiting tumor growth [283].
Currently, there are several strategies to target BCSCs, each with its own advantages and disadvantages (Table 3). So far, there are a variety of drugs have been introduced into the clinical to bring great cure hope to patients. However, the problem is that breast cancer will develop resistance to certain drugs, and one of the reasons is the presence of CSCs. There are also a variety of therapies targeting CSCs. CAR-T therapy in immunotherapy has attracted much attention due to its high efficiency in killing cancer cells, but its fatal disadvantage is that it cannot distinguish the expression level of the same target protein in different cells, which will lead to strong side effects. At present, there are several difficulties in the treatment of CSCs. Firstly, it is difficult to distinguish the molecular characteristics of normal stem cells and CSCs. They share cell surface markers and signaling pathways, and the specificity of CSCs remains to be explored. Secondly, it is necessary to use a variety of combined treatments to extinguish the dormant and proliferating CSCs. Finally, if the CSCs can be eliminated early in the tumor, then the treatment effect will be more effective.
Advantages and disadvantages of therapeutic strategies
| Strategy | Advantages | Disadvantages | Ref. |
|---|---|---|---|
| Natural compounds | Have biological activity and high bioavailability. | Separation, synthesis and the target protein analysis are difficult. | [284] |
| Antibody-based biopharmaceuticals | Have highly specific targeting tumor cell surface antigens and immune cells. | Its killing in tumor cells is limited. | [233, 234] |
| Cytotherapy | Enhance or alter intrinsic immune capacity. | There are fatal adverse reactions. | [245, 246] |
| Synthetic small molecule compounds | Have the possibility of oral administration, stability, membrane permeability. | Has off-target toxicity. | [250-252] |
| New uses of old drugs | Highly safe and may reduce overall development costs and shorten development time. | Is still affected by multi-drug resistance. | [268] |
| Nucleic acid medicines | Achieved a short reaction time, minor side effects, high specificity and long-lasting therapeutic effect. | Are lacking available genetic information and limited transmission. | [279, 280] |
Conclusions and perspectives
Current breast cancer therapies have significantly improved in eliminating primary tumors and prolonging survival. However, effectively eliminating BCSCs is difficult. First, the dynamic intrinsic property of BCSCs and the unique CSC niche make it challenging to eradicate BCSCs. Second, BCSCs can switch their state between dormancy and rapid reproduction. Killing cells in a quiescent state is still a challenge. Third, the unique microenvironment of BCSCs is not well elucidated, and most current in vivo studies on BCSCs are performed in an immune-deficient environment, making it challenging to recapitulate biological complications in the clinic.
Although the single-cell sequencing studies did not detect a single distinct stem cell population with unique transcriptional features in the adult murine gland, these sequencing data provided a detailed transcriptional map of mammary epithelial differentiation, suggesting that scRNA-sequencing coupled with flow cytometry might be a powerful tool for CSC discovery and isolation. In addition, targeting unique regulatory factors that contribute to maintaining CSC niches in combination with traditional treatment against rapidly proliferating cancer cells will be a promising approach and has shown synergistic effects. However, the safety and efficacy of the combined strategies need to be evaluated in preclinical and clinical studies. Modulating the tumor microenvironment to target BCSCs also exhibited encouraging results. At present, there are various therapies for BCSCs, each with its own advantages and disadvantages. In addition to the drugs mentioned above, EpCAM-CAR-T (NCT02915445) has entered the clinical state for BCSCs immunotherapy, and good results are expected. Currently, only cervical cancer vaccine has entered clinical application, specific antigens for BCSCs to develop antibodies against breast cancer need further investigation. With increasing progress in BCSC research, patients will benefit from BCSC-based personal cancer treatment.
Funding
This work was supported by National Key R&D Program of China (2020YFA0112300 and 2020YFA0803200), National Natural Science Foundation of China (U2102203 and 81830087), and Yunnan Fundamental Research Projects (202101AS070050). “Ten Thousand Plan” - National High-Level Talents Special Support Plan (WR-YK5202101); Program for Outstanding Leading Talents in Shanghai; Program of Shanghai Academic/Technology Research Leader (20XD1400700); Program for Outstanding Medical Academic Leader in Shanghai (2019LJ04); The innovative research team of high-level local university in Shanghai.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Gudjonsson T, Villadsen R, Nielsen HL, Ronnov-Jessen L, Bissell MJ, Petersen OW. Isolation, immortalization, and characterization of a human breast epithelial cell line with stem cell properties. Genes Dev. 2002;16:693-706
2. Pechoux C, Gudjonsson T, Ronnov-Jessen L, Bissell MJ, Petersen OW. Human mammary luminal epithelial cells contain progenitors to myoepithelial cells. Dev Biol. 1999;206:88-99
3. van Amerongen R, Bowman AN, Nusse R. Developmental stage and time dictate the fate of Wnt/beta-catenin-responsive stem cells in the mammary gland. Cell Stem Cell. 2012;11:387-400
4. Van Keymeulen A, Rocha AS, Ousset M, Beck B, Bouvencourt G, Rock J. et al. Distinct stem cells contribute to mammary gland development and maintenance. Nature. 2011;479:189-93
5. Wang D, Cai C, Dong X, Yu QC, Zhang XO, Yang L. et al. Identification of multipotent mammary stem cells by protein C receptor expression. Nature. 2015;517:81-4
6. Cai S, Kalisky T, Sahoo D, Dalerba P, Feng W, Lin Y. et al. A Quiescent Bcl11b High Stem Cell Population Is Required for Maintenance of the Mammary Gland. Cell Stem Cell. 2017;20:247-60 e5
7. Watson CJ. How should we define mammary stem cells? Trends Cell Biol. 2021;31:621-7
8. Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001;98:10869-74
9. Perou CM, Sorlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA. et al. Molecular portraits of human breast tumours. Nature. 2000;406:747-52
10. Cheang MC, Chia SK, Voduc D, Gao D, Leung S, Snider J. et al. Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst. 2009;101:736-50
11. Lim E, Vaillant F, Wu D, Forrest NC, Pal B, Hart AH. et al. Aberrant luminal progenitors as the candidate target population for basal tumor development in BRCA1 mutation carriers. Nat Med. 2009;15:907-13
12. Molyneux G, Geyer FC, Magnay FA, McCarthy A, Kendrick H, Natrajan R. et al. BRCA1 basal-like breast cancers originate from luminal epithelial progenitors and not from basal stem cells. Cell Stem Cell. 2010;7:403-17
13. Waks AG, Winer EP. Breast Cancer Treatment: A Review. JAMA. 2019;321:288-300
14. Riggio AI, Varley KE, Welm AL. The lingering mysteries of metastatic recurrence in breast cancer. Br J Cancer. 2021;124:13-26
15. Wang R, Zhu Y, Liu X, Liao X, He J, Niu L. The Clinicopathological features and survival outcomes of patients with different metastatic sites in stage IV breast cancer. BMC Cancer. 2019;19:1091
16. Lee KL, Kuo YC, Ho YS, Huang YH. Triple-Negative Breast Cancer: Current Understanding and Future Therapeutic Breakthrough Targeting Cancer Stemness. Cancers (Basel). 2019 11
17. Ginestier C, Hur MH, Charafe-Jauffret E, Monville F, Dutcher J, Brown M. et al. ALDH1 is a marker of normal and malignant human mammary stem cells and a predictor of poor clinical outcome. Cell Stem Cell. 2007;1:555-67
18. Phillips TM, McBride WH, Pajonk F. The response of CD24(-/low)/CD44+ breast cancer-initiating cells to radiation. J Natl Cancer Inst. 2006;98:1777-85
19. Beck B, Blanpain C. Unravelling cancer stem cell potential. Nat Rev Cancer. 2013;13:727-38
20. Ricardo S, Vieira AF, Gerhard R, Leitao D, Pinto R, Cameselle-Teijeiro JF. et al. Breast cancer stem cell markers CD44, CD24 and ALDH1: expression distribution within intrinsic molecular subtype. J Clin Pathol. 2011;64:937-46
21. Li W, Ma H, Zhang J, Zhu L, Wang C, Yang Y. Unraveling the roles of CD44/CD24 and ALDH1 as cancer stem cell markers in tumorigenesis and metastasis. Sci Rep. 2017;7:13856
22. Jiang Y, Guo Y, Hao J, Guenter R, Lathia J, Beck AW. et al. Development of an arteriolar niche and self-renewal of breast cancer stem cells by lysophosphatidic acid/protein kinase D signaling. Commun Biol. 2021;4:780
23. Luo H, Chen CY, Li X, Zhang X, Su CW, Liu Y. et al. Increased lipogenesis is critical for self-renewal and growth of breast cancer stem cells: Impact of omega-3 fatty acids. Stem Cells. 2021;39:1660-70
24. Lytle NK, Barber AG, Reya T. Stem cell fate in cancer growth, progression and therapy resistance. Nat Rev Cancer. 2018;18:669-80
25. Ocana OH, Corcoles R, Fabra A, Moreno-Bueno G, Acloque H, Vega S. et al. Metastatic colonization requires the repression of the epithelial-mesenchymal transition inducer Prrx1. Cancer Cell. 2012;22:709-24
26. Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci U S A. 2003;100:3983-8
27. Wang D, Hu X, Liu C, Jia Y, Bai Y, Cai C. et al. Protein C receptor is a therapeutic stem cell target in a distinct group of breast cancers. Cell Res. 2019;29:832-45
28. Maruyama R, Choudhury S, Kowalczyk A, Bessarabova M, Beresford-Smith B, Conway T. et al. Epigenetic regulation of cell type-specific expression patterns in the human mammary epithelium. PLoS Genet. 2011;7:e1001369
29. Yin AH, Miraglia S, Zanjani ED, Almeida-Porada G, Ogawa M, Leary AG. et al. AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood. 1997;90:5002-12
30. Miraglia S, Godfrey W, Yin AH, Atkins K, Warnke R, Holden JT. et al. A novel five-transmembrane hematopoietic stem cell antigen: isolation, characterization, and molecular cloning. Blood. 1997;90:5013-21
31. Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T. et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964-7
32. Liu TJ, Sun BC, Zhao XL, Zhao XM, Sun T, Gu Q. et al. CD133+ cells with cancer stem cell characteristics associates with vasculogenic mimicry in triple-negative breast cancer. Oncogene. 2013;32:544-53
33. Wright MH, Calcagno AM, Salcido CD, Carlson MD, Ambudkar SV, Varticovski L. Brca1 breast tumors contain distinct CD44+/CD24- and CD133+ cells with cancer stem cell characteristics. Breast Cancer Res. 2008;10:R10
34. Krebsbach PH, Villa-Diaz LG. The Role of Integrin alpha6 (CD49f) in Stem Cells: More than a Conserved Biomarker. Stem Cells Dev. 2017;26:1090-9
35. Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105-11
36. Van Keymeulen A, Lee MY, Ousset M, Brohee S, Rorive S, Giraddi RR. et al. Reactivation of multipotency by oncogenic PIK3CA induces breast tumour heterogeneity. Nature. 2015;525:119-23
37. Liu S, Ginestier C, Charafe-Jauffret E, Foco H, Kleer CG, Merajver SD. et al. BRCA1 regulates human mammary stem/progenitor cell fate. Proc Natl Acad Sci U S A. 2008;105:1680-5
38. Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G. et al. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008;10:593-601
39. Wu MJ, Chen YS, Kim MR, Chang CC, Gampala S, Zhang Y. et al. Epithelial-Mesenchymal Transition Directs Stem Cell Polarity via Regulation of Mitofusin. Cell Metab. 2019;29:993-1002 e6
40. Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A. et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol. 2009;11:1487-95
41. Bhat-Nakshatri P, Appaiah H, Ballas C, Pick-Franke P, Goulet R Jr, Badve S. et al. SLUG/SNAI2 and tumor necrosis factor generate breast cells with CD44+/CD24- phenotype. BMC Cancer. 2010;10:411
42. Elegbe IA. An evaluation of the New Roche Diagnostics Kit for the rapid identification of clinically important non-dextrose, non-fastidious gram-negative rods. Afr J Med Med Sci. 1980;9:159-62
43. Liu S, Cong Y, Wang D, Sun Y, Deng L, Liu Y. et al. Breast cancer stem cells transition between epithelial and mesenchymal states reflective of their normal counterparts. Stem Cell Reports. 2014;2:78-91
44. Bierie B, Pierce SE, Kroeger C, Stover DG, Pattabiraman DR, Thiru P. et al. Integrin-beta4 identifies cancer stem cell-enriched populations of partially mesenchymal carcinoma cells. Proc Natl Acad Sci U S A. 2017;114:E2337-E46
45. Lourenco AR, Ban Y, Crowley MJ, Lee SB, Ramchandani D, Du W. et al. Differential Contributions of Pre- and Post-EMT Tumor Cells in Breast Cancer Metastasis. Cancer Res. 2020;80:163-9
46. Jolly MK, Huang B, Lu M, Mani SA, Levine H, Ben-Jacob E. Towards elucidating the connection between epithelial-mesenchymal transitions and stemness. J R Soc Interface. 2014;11:20140962
47. Deshmukh AP, Vasaikar SV, Tomczak K, Tripathi S, den Hollander P, Arslan E. et al. Identification of EMT signaling cross-talk and gene regulatory networks by single-cell RNA sequencing. Proc Natl Acad Sci U S A. 2021 118
48. Colacino JA, Azizi E, Brooks MD, Harouaka R, Fouladdel S, McDermott SP. et al. Heterogeneity of Human Breast Stem and Progenitor Cells as Revealed by Transcriptional Profiling. Stem Cell Reports. 2018;10:1596-609
49. Kroger C, Afeyan A, Mraz J, Eaton EN, Reinhardt F, Khodor YL. et al. Acquisition of a hybrid E/M state is essential for tumorigenicity of basal breast cancer cells. Proc Natl Acad Sci U S A. 2019;116:7353-62
50. Pasani S, Sahoo S, Jolly MK. Hybrid E/M Phenotype(s) and Stemness: A Mechanistic Connection Embedded in Network Topology. J Clin Med. 2020 10
51. Brown MS, Abdollahi B, Wilkins OM, Lu H, Chakraborty P, Ognjenovic NB. et al. Phenotypic heterogeneity driven by plasticity of the intermediate EMT state governs disease progression and metastasis in breast cancer. Sci Adv. 2022;8:eabj8002
52. Vilchez Mercedes SA, Bocci F, Ahmed M, Eder I, Zhu N, Levine H. et al. Nrf2 Modulates the Hybrid Epithelial/Mesenchymal Phenotype and Notch Signaling During Collective Cancer Migration. Front Mol Biosci. 2022;9:807324
53. Dick JE. Looking ahead in cancer stem cell research. Nat Biotechnol. 2009;27:44-6
54. Gupta PB, Fillmore CM, Jiang G, Shapira SD, Tao K, Kuperwasser C. et al. Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell. 2011;146:633-44
55. Wang W, Quan Y, Fu Q, Liu Y, Liang Y, Wu J. et al. Dynamics between cancer cell subpopulations reveals a model coordinating with both hierarchical and stochastic concepts. PLoS One. 2014;9:e84654
56. Lee S, Zelen M. A stochastic model for predicting the mortality of breast cancer. J Natl Cancer Inst Monogr. 2006:79-86
57. Wiesweg M, Eberhardt WEE, Reis H, Ting S, Savvidou N, Skiba C. et al. High Prevalence of Concomitant Oncogene Mutations in Prospectively Identified Patients with ROS1-Positive Metastatic Lung Cancer. J Thorac Oncol. 2017;12:54-64
58. Williams RT, den Besten W, Sherr CJ. Cytokine-dependent imatinib resistance in mouse BCR-ABL+, Arf-null lymphoblastic leukemia. Genes Dev. 2007;21:2283-7
59. Melzer C, von der Ohe J, Lehnert H, Ungefroren H, Hass R. Cancer stem cell niche models and contribution by mesenchymal stroma/stem cells. Mol Cancer. 2017;16:28
60. Schmitt M, Metzger M, Gradl D, Davidson G, Orian-Rousseau V. CD44 functions in Wnt signaling by regulating LRP6 localization and activation. Cell Death Differ. 2015;22:677-89
61. Kim T, Yang SJ, Hwang D, Song J, Kim M, Kyum Kim S. et al. A basal-like breast cancer-specific role for SRF-IL6 in YAP-induced cancer stemness. Nat Commun. 2015;6:10186
62. Maugeri-Sacca M, De Maria R. Hippo pathway and breast cancer stem cells. Crit Rev Oncol Hematol. 2016;99:115-22
63. Cordenonsi M, Zanconato F, Azzolin L, Forcato M, Rosato A, Frasson C. et al. The Hippo transducer TAZ confers cancer stem cell-related traits on breast cancer cells. Cell. 2011;147:759-72
64. Calses PC, Crawford JJ, Lill JR, Dey A. Hippo Pathway in Cancer: Aberrant Regulation and Therapeutic Opportunities. Trends Cancer. 2019;5:297-307
65. Dhanasekaran SM, Balbin OA, Chen G, Nadal E, Kalyana-Sundaram S, Pan J. et al. Transcriptome meta-analysis of lung cancer reveals recurrent aberrations in NRG1 and Hippo pathway genes. Nat Commun. 2014;5:5893
66. Tanas MR, Ma S, Jadaan FO, Ng CK, Weigelt B, Reis-Filho JS. et al. Mechanism of action of a WWTR1(TAZ)-CAMTA1 fusion oncoprotein. Oncogene. 2016;35:929-38
67. Merritt N, Garcia K, Rajendran D, Lin ZY, Zhang X, Mitchell KA. et al. TAZ-CAMTA1 and YAP-TFE3 alter the TAZ/YAP transcriptome by recruiting the ATAC histone acetyltransferase complex. Elife. 2021 10
68. Guo W, Keckesova Z, Donaher JL, Shibue T, Tischler V, Reinhardt F. et al. Slug and Sox9 cooperatively determine the mammary stem cell state. Cell. 2012;148:1015-28
69. Dravis C, Chung CY, Lytle NK, Herrera-Valdez J, Luna G, Trejo CL. et al. Epigenetic and Transcriptomic Profiling of Mammary Gland Development and Tumor Models Disclose Regulators of Cell State Plasticity. Cancer Cell. 2018;34:466-82 e6
70. Anstine LJ, Keri R. A new view of the mammary epithelial hierarchy and its implications for breast cancer initiation and metastasis. J Cancer Metastasis Treat. 2019 5
71. Folmes CD, Nelson TJ, Martinez-Fernandez A, Arrell DK, Lindor JZ, Dzeja PP. et al. Somatic oxidative bioenergetics transitions into pluripotency-dependent glycolysis to facilitate nuclear reprogramming. Cell Metab. 2011;14:264-71
72. Ito K, Suda T. Metabolic requirements for the maintenance of self-renewing stem cells. Nat Rev Mol Cell Biol. 2014;15:243-56
73. Varum S, Rodrigues AS, Moura MB, Momcilovic O, Easley CAt, Ramalho-Santos J. et al. Energy metabolism in human pluripotent stem cells and their differentiated counterparts. PLoS One. 2011;6:e20914
74. Wu J, Ocampo A, Belmonte JCI. Cellular Metabolism and Induced Pluripotency. Cell. 2016;166:1371-85
75. Anderson AS, Roberts PC, Frisard MI, Hulver MW, Schmelz EM. Ovarian tumor-initiating cells display a flexible metabolism. Exp Cell Res. 2014;328:44-57
76. Luo M, Shang L, Brooks MD, Jiagge E, Zhu Y, Buschhaus JM. et al. Targeting Breast Cancer Stem Cell State Equilibrium through Modulation of Redox Signaling. Cell Metab. 2018;28:69-86 e6
77. Sancho P, Burgos-Ramos E, Tavera A, Bou Kheir T, Jagust P, Schoenhals M. et al. MYC/PGC-1alpha Balance Determines the Metabolic Phenotype and Plasticity of Pancreatic Cancer Stem Cells. Cell Metab. 2015;22:590-605
78. Palomeras S, Ruiz-Martinez S, Puig T. Targeting Breast Cancer Stem Cells to Overcome Treatment Resistance. Molecules. 2018 23
79. Payne KK, Manjili MH. Adaptive immune responses associated with breast cancer relapse. Arch Immunol Ther Exp (Warsz). 2012;60:345-50
80. Xiang L, Semenza GL. Hypoxia-inducible factors promote breast cancer stem cell specification and maintenance in response to hypoxia or cytotoxic chemotherapy. Adv Cancer Res. 2019;141:175-212
81. La Belle Flynn A, Calhoun BC, Sharma A, Chang JC, Almasan A, Schiemann WP. Autophagy inhibition elicits emergence from metastatic dormancy by inducing and stabilizing Pfkfb3 expression. Nat Commun. 2019;10:3668
82. Li F, Xu J, Liu S. Cancer Stem Cells and Neovascularization. Cells. 2021 10
83. Bussolati B, Grange C, Sapino A, Camussi G. Endothelial cell differentiation of human breast tumour stem/progenitor cells. J Cell Mol Med. 2009;13:309-19
84. Sun H, Yao N, Cheng S, Li L, Liu S, Yang Z. et al. Cancer stem-like cells directly participate in vasculogenic mimicry channels in triple-negative breast cancer. Cancer Biol Med. 2019;16:299-311
85. Gong W, Sun B, Zhao X, Zhang D, Sun J, Liu T. et al. Nodal signaling promotes vasculogenic mimicry formation in breast cancer via the Smad2/3 pathway. Oncotarget. 2016;7:70152-67
86. Xu J, Yang X, Deng Q, Yang C, Wang D, Jiang G. et al. TEM8 marks neovasculogenic tumor-initiating cells in triple-negative breast cancer. Nat Commun. 2021;12:4413
87. Cong M, Wang Y, Yang Y, Lian C, Zhuang X, Li X. et al. MTSS1 suppresses mammary tumor-initiating cells by enhancing RBCK1-mediated p65 ubiquitination. Nat Cancer. 2020;1:222-34
88. Gallardo-Perez JC, Adan-Ladron de Guevara A, Marin-Hernandez A, Moreno-Sanchez R, Rodriguez-Enriquez S. HPI/AMF inhibition halts the development of the aggressive phenotype of breast cancer stem cells. Biochim Biophys Acta Mol Cell Res. 2017;1864:1679-90
89. Hu J, Li G, Zhang P, Zhuang X, Hu G. A CD44v(+) subpopulation of breast cancer stem-like cells with enhanced lung metastasis capacity. Cell Death Dis. 2017;8:e2679
90. Oliphant MUJ, Vincent MY, Galbraith MD, Pandey A, Zaberezhnyy V, Rudra P. et al. SIX2 Mediates Late-Stage Metastasis via Direct Regulation of SOX2 and Induction of a Cancer Stem Cell Program. Cancer Res. 2019;79:720-34
91. Singh JK, Simoes BM, Howell SJ, Farnie G, Clarke RB. Recent advances reveal IL-8 signaling as a potential key to targeting breast cancer stem cells. Breast Cancer Res. 2013;15:210
92. Zhang C, Gao H, Li C, Tu J, Chen Z, Su W. et al. TGFbeta1 Promotes Breast Cancer Local Invasion and Liver Metastasis by Increasing the CD44(high)/CD24(-) Subpopulation. Technol Cancer Res Treat. 2018;17:1533033818764497
93. Lim SY, Yuzhalin AE, Gordon-Weeks AN, Muschel RJ. Targeting the CCL2-CCR2 signaling axis in cancer metastasis. Oncotarget. 2016;7:28697-710
94. Fang WB, Yao M, Brummer G, Acevedo D, Alhakamy N, Berkland C. et al. Targeted gene silencing of CCL2 inhibits triple negative breast cancer progression by blocking cancer stem cell renewal and M2 macrophage recruitment. Oncotarget. 2016;7:49349-67
95. Kong L, Guo S, Liu C, Zhao Y, Feng C, Liu Y. et al. Overexpression of SDF-1 activates the NF-kappaB pathway to induce epithelial to mesenchymal transition and cancer stem cell-like phenotypes of breast cancer cells. Int J Oncol. 2016;48:1085-94
96. Shan S, Lv Q, Zhao Y, Liu C, Sun Y, Xi K. et al. Wnt/beta-catenin pathway is required for epithelial to mesenchymal transition in CXCL12 over expressed breast cancer cells. Int J Clin Exp Pathol. 2015;8:12357-67
97. Benard O, Qian X, Liang H, Ren Z, Suyama K, Norton L. et al. p21CIP1 Promotes Mammary Cancer-Initiating Cells via Activation of Wnt/TCF1/CyclinD1 Signaling. Mol Cancer Res. 2019;17:1571-81
98. Gener P, Rafael D, Seras-Franzoso J, Perez A, Pindado LA, Casas G. et al. Pivotal Role of AKT2 during Dynamic Phenotypic Change of Breast Cancer Stem Cells. Cancers (Basel). 2019 11
99. Rosenthal DT, Zhang J, Bao L, Zhu L, Wu Z, Toy K. et al. RhoC impacts the metastatic potential and abundance of breast cancer stem cells. PLoS One. 2012;7:e40979
100. Gagliardi M, Pitner MK, Park J, Xie X, Saso H, Larson RA. et al. Differential functions of ERK1 and ERK2 in lung metastasis processes in triple-negative breast cancer. Sci Rep. 2020;10:8537
101. Shi L, Tang X, Qian M, Liu Z, Meng F, Fu L. et al. A SIRT1-centered circuitry regulates breast cancer stemness and metastasis. Oncogene. 2018;37:6299-315
102. Nguyen K, Yan Y, Yuan B, Dasgupta A, Sun J, Mu H. et al. ST8SIA1 Regulates Tumor Growth and Metastasis in TNBC by Activating the FAK-AKT-mTOR Signaling Pathway. Mol Cancer Ther. 2018;17:2689-701
103. Gong Z, Li A, Ding J, Li Q, Zhang L, Li Y. et al. OTUD7B Deubiquitinates LSD1 to Govern Its Binding Partner Specificity, Homeostasis, and Breast Cancer Metastasis. Adv Sci (Weinh). 2021;8:e2004504
104. Li M, Pan M, Wang J, You C, Zhao F, Zheng D. et al. miR-7 Reduces Breast Cancer Stem Cell Metastasis via Inhibiting RELA to Decrease ESAM Expression. Mol Ther Oncolytics. 2020;18:70-82
105. Lin C, Gao B, Yan X, Lei Z, Chen K, Li Y. et al. MicroRNA 628 suppresses migration and invasion of breast cancer stem cells through targeting SOS1. Onco Targets Ther. 2018;11:5419-28
106. Tang T, Yang Z, Zhu Q, Wu Y, Sun K, Alahdal M. et al. Up-regulation of miR-210 induced by a hypoxic microenvironment promotes breast cancer stem cells metastasis, proliferation, and self-renewal by targeting E-cadherin. FASEB J. 2018: fj201801013R.
107. Zeng L, Cen Y, Chen J. Long non-coding RNA MALAT-1 contributes to maintenance of stem cell-like phenotypes in breast cancer cells. Oncol Lett. 2018;15:2117-22
108. Hou L, Tu J, Cheng F, Yang H, Yu F, Wang M. et al. Long noncoding RNA ROR promotes breast cancer by regulating the TGF-beta pathway. Cancer Cell Int. 2018;18:142
109. Liu Y, Zhang P, Wu Q, Fang H, Wang Y, Xiao Y. et al. Long non-coding RNA NR2F1-AS1 induces breast cancer lung metastatic dormancy by regulating NR2F1 and DeltaNp63. Nat Commun. 2021;12:5232
110. Yang N, Wang C, Wang Z, Zona S, Lin SX, Wang X. et al. FOXM1 recruits nuclear Aurora kinase A to participate in a positive feedback loop essential for the self-renewal of breast cancer stem cells. Oncogene. 2017;36:3428-40
111. Verigos J, Karakaidos P, Kordias D, Papoudou-Bai A, Evangelou Z, Harissis HV. et al. The Histone Demethylase LSD1/KappaDM1A Mediates Chemoresistance in Breast Cancer via Regulation of a Stem Cell Program. Cancers (Basel). 2019 11
112. Tan W, Tang H, Jiang X, Ye F, Huang L, Shi D. et al. Metformin mediates induction of miR-708 to inhibit self-renewal and chemoresistance of breast cancer stem cells through targeting CD47. J Cell Mol Med. 2019;23:5994-6004
113. Wang N, Weng J, Xia J, Zhu Y, Chen Q, Hu D. et al. SIPA1 enhances SMAD2/3 expression to maintain stem cell features in breast cancer cells. Stem Cell Res. 2020;49:102099
114. Butti R, Gunasekaran VP, Kumar TVS, Banerjee P, Kundu GC. Breast cancer stem cells: Biology and therapeutic implications. Int J Biochem Cell Biol. 2019;107:38-52
115. Liu YP, Zheng CC, Huang YN, He ML, Xu WW, Li B. Molecular mechanisms of chemo- and radiotherapy resistance and the potential implications for cancer treatment. MedComm (2020). 2021;2:315-40
116. Yousefnia S, Seyed Forootan F, Seyed Forootan S, Nasr Esfahani MH, Gure AO, Ghaedi K. Mechanistic Pathways of Malignancy in Breast Cancer Stem Cells. Front Oncol. 2020;10:452
117. Dehghan Harati M, Rodemann HP, Toulany M. Nanog Signaling Mediates Radioresistance in ALDH-Positive Breast Cancer Cells. Int J Mol Sci. 2019 20
118. Bartucci M, Dattilo R, Moriconi C, Pagliuca A, Mottolese M, Federici G. et al. TAZ is required for metastatic activity and chemoresistance of breast cancer stem cells. Oncogene. 2015;34:681-90
119. Lai D, Ho KC, Hao Y, Yang X. Taxol resistance in breast cancer cells is mediated by the hippo pathway component TAZ and its downstream transcriptional targets Cyr61 and CTGF. Cancer Res. 2011;71:2728-38
120. Lin CH, Pelissier FA, Zhang H, Lakins J, Weaver VM, Park C. et al. Microenvironment rigidity modulates responses to the HER2 receptor tyrosine kinase inhibitor lapatinib via YAP and TAZ transcription factors. Mol Biol Cell. 2015;26:3946-53
121. Turunen SP, von Nandelstadh P, Ohman T, Gucciardo E, Seashore-Ludlow B, Martins B. et al. FGFR4 phosphorylates MST1 to confer breast cancer cells resistance to MST1/2-dependent apoptosis. Cell Death Differ. 2019;26:2577-93
122. Shima H, Yamada A, Ishikawa T, Endo I. Are breast cancer stem cells the key to resolving clinical issues in breast cancer therapy? Gland Surg. 2017;6:82-8
123. Jiao X, Velasco-Velazquez MA, Wang M, Li Z, Rui H, Peck AR. et al. CCR5 Governs DNA Damage Repair and Breast Cancer Stem Cell Expansion. Cancer Res. 2018;78:1657-71
124. Dey P, Rathod M, De A. Targeting stem cells in the realm of drug-resistant breast cancer. Breast Cancer (Dove Med Press). 2019;11:115-35
125. Bai X, Ni J, Beretov J, Graham P, Li Y. Cancer stem cell in breast cancer therapeutic resistance. Cancer Treat Rev. 2018;69:152-63
126. Smit L, Berns K, Spence K, Ryder WD, Zeps N, Madiredjo M. et al. An integrated genomic approach identifies that the PI3K/AKT/FOXO pathway is involved in breast cancer tumor initiation. Oncotarget. 2016;7:2596-610
127. Woo Y, Oh J, Kim JS. Suppression of Nrf2 Activity by Chestnut Leaf Extract Increases Chemosensitivity of Breast Cancer Stem Cells to Paclitaxel. Nutrients. 2017 9
128. Ryoo IG, Choi BH, Kwak MK. Activation of NRF2 by p62 and proteasome reduction in sphere-forming breast carcinoma cells. Oncotarget. 2015;6:8167-84
129. Ryoo IG, Choi BH, Ku SK, Kwak MK. High CD44 expression mediates p62-associated NFE2L2/NRF2 activation in breast cancer stem cell-like cells: Implications for cancer stem cell resistance. Redox Biol. 2018;17:246-58
130. Wang LL, Wan XY, Liu CQ, Zheng FM. NDR1 increases NOTCH1 signaling activity by impairing Fbw7 mediated NICD degradation to enhance breast cancer stem cell properties. Mol Med. 2022;28:49
131. Martin CE, List K. Cell surface-anchored serine proteases in cancer progression and metastasis. Cancer Metastasis Rev. 2019;38:357-87
132. Chang LH, Chen CH, Huang DY, Pai HC, Pan SL, Teng CM. Thrombin induces expression of twist and cell motility via the hypoxia-inducible factor-1alpha translational pathway in colorectal cancer cells. J Cell Physiol. 2011;226:1060-8
133. Shan Y, You B, Shi S, Shi W, Zhang Z, Zhang Q. et al. Hypoxia-Induced Matrix Metalloproteinase-13 Expression in Exosomes from Nasopharyngeal Carcinoma Enhances Metastases. Cell Death Dis. 2018;9:382
134. Najy AJ, Dyson G, Jena BP, Lin CY, Kim HR. Matriptase activation and shedding through PDGF-D-mediated extracellular acidosis. Am J Physiol Cell Physiol. 2016;310:C293-304
135. Devereaux S, Linch DC, Gribben JG, McMillan A, Patterson K, Goldstone AH. GM-CSF accelerates neutrophil recovery after autologous bone marrow transplantation for Hodgkin's disease. Bone Marrow Transplant. 1989;4:49-54
136. Jewer M, Lee L, Leibovitch M, Zhang G, Liu J, Findlay SD. et al. Translational control of breast cancer plasticity. Nat Commun. 2020;11:2498
137. Mathieu J, Zhang Z, Zhou W, Wang AJ, Heddleston JM, Pinna CM. et al. HIF induces human embryonic stem cell markers in cancer cells. Cancer Res. 2011;71:4640-52
138. Wang P, Wan WW, Xiong SL, Feng H, Wu N. Cancer stem-like cells can be induced through dedifferentiation under hypoxic conditions in glioma, hepatoma and lung cancer. Cell Death Discov. 2017;3:16105
139. Kim H, Lin Q, Glazer PM, Yun Z. The hypoxic tumor microenvironment in vivo selects the cancer stem cell fate of breast cancer cells. Breast Cancer Res. 2018;20:16
140. Zhang C, Samanta D, Lu H, Bullen JW, Zhang H, Chen I. et al. Hypoxia induces the breast cancer stem cell phenotype by HIF-dependent and ALKBH5-mediated m(6)A-demethylation of NANOG mRNA. Proc Natl Acad Sci U S A. 2016;113:E2047-56
141. Lu H, Samanta D, Xiang L, Zhang H, Hu H, Chen I. et al. Chemotherapy triggers HIF-1-dependent glutathione synthesis and copper chelation that induces the breast cancer stem cell phenotype. Proc Natl Acad Sci U S A. 2015;112:E4600-9
142. Ullah MS, Davies AJ, Halestrap AP. The plasma membrane lactate transporter MCT4, but not MCT1, is up-regulated by hypoxia through a HIF-1alpha-dependent mechanism. J Biol Chem. 2006;281:9030-7
143. Zhu P, He F, Hou Y, Tu G, Li Q, Jin T. et al. A novel hypoxic long noncoding RNA KB-1980E6.3 maintains breast cancer stem cell stemness via interacting with IGF2BP1 to facilitate c-Myc mRNA stability. Oncogene. 2021;40:1609-27
144. Kim R, Emi M, Tanabe K. Cancer cell immune escape and tumor progression by exploitation of anti-inflammatory and pro-inflammatory responses. Cancer Biol Ther. 2005;4:924-33
145. Seruga B, Zhang H, Bernstein LJ, Tannock IF. Cytokines and their relationship to the symptoms and outcome of cancer. Nat Rev Cancer. 2008;8:887-99
146. Chen W, Qin Y, Liu S. Cytokines, breast cancer stem cells (BCSCs) and chemoresistance. Clin Transl Med. 2018;7:27
147. Liu S, Lee JS, Jie C, Park MH, Iwakura Y, Patel Y. et al. HER2 Overexpression Triggers an IL1alpha Proinflammatory Circuit to Drive Tumorigenesis and Promote Chemotherapy Resistance. Cancer Res. 2018;78:2040-51
148. Wang D, Xu J, Liu B, He X, Zhou L, Hu X. et al. IL6 blockade potentiates the anti-tumor effects of gamma-secretase inhibitors in Notch3-expressing breast cancer. Cell Death Differ. 2018;25:330-9
149. Radharani NNV, Yadav AS, Nimma R, Kumar TVS, Bulbule A, Chanukuppa V. et al. Tumor-associated macrophage derived IL-6 enriches cancer stem cell population and promotes breast tumor progression via Stat-3 pathway. Cancer Cell Int. 2022;22:122
150. Choi HS, Kim JH, Kim SL, Lee DS. Disruption of the NF-kappaB/IL-8 Signaling Axis by Sulconazole Inhibits Human Breast Cancer Stem Cell Formation. Cells. 2019 8
151. Sorrentino C, Ciummo SL, D'Antonio L, Fieni C, Lanuti P, Turdo A. et al. Interleukin-30 feeds breast cancer stem cells via CXCL10 and IL23 autocrine loops and shapes immune contexture and host outcome. J Immunother Cancer. 2021 9
152. Shi P, Liu W, Tala, Wang H, Li F, Zhang H. et al. Metformin suppresses triple-negative breast cancer stem cells by targeting KLF5 for degradation. Cell Discov. 2017;3:17010
153. Chen W, Qin Y, Wang D, Zhou L, Liu Y, Chen S. et al. CCL20 triggered by chemotherapy hinders the therapeutic efficacy of breast cancer. PLoS Biol. 2018;16:e2005869
154. Sheng D, Ma W, Zhang R, Zhou L, Deng Q, Tu J. et al. Ccl3 enhances docetaxel chemosensitivity in breast cancer by triggering proinflammatory macrophage polarization. J Immunother Cancer. 2022;0:e003793
155. Ciummo SL, D'Antonio L, Sorrentino C, Fieni C, Lanuti P, Stassi G. et al. The C-X-C Motif Chemokine Ligand 1 Sustains Breast Cancer Stem Cell Self-Renewal and Promotes Tumor Progression and Immune Escape Programs. Front Cell Dev Biol. 2021;9:689286
156. Castiello L, Sestili P, Schiavoni G, Dattilo R, Monque DM, Ciaffoni F. et al. Disruption of IFN-I Signaling Promotes HER2/Neu Tumor Progression and Breast Cancer Stem Cells. Cancer Immunol Res. 2018;6:658-70
157. Sappino AP, Schurch W, Gabbiani G. Differentiation repertoire of fibroblastic cells: expression of cytoskeletal proteins as marker of phenotypic modulations. Lab Invest. 1990;63:144-61
158. De Wever O, Van Bockstal M, Mareel M, Hendrix A, Bracke M. Carcinoma-associated fibroblasts provide operational flexibility in metastasis. Semin Cancer Biol. 2014;25:33-46
159. Ohlund D, Elyada E, Tuveson D. Fibroblast heterogeneity in the cancer wound. J Exp Med. 2014;211:1503-23
160. Lappano R, Talia M, Cirillo F, Rigiracciolo DC, Scordamaglia D, Guzzi R. et al. The IL1beta-IL1R signaling is involved in the stimulatory effects triggered by hypoxia in breast cancer cells and cancer-associated fibroblasts (CAFs). J Exp Clin Cancer Res. 2020;39:153
161. Albrengues J, Bourget I, Pons C, Butet V, Hofman P, Tartare-Deckert S. et al. LIF mediates proinvasive activation of stromal fibroblasts in cancer. Cell Rep. 2014;7:1664-78
162. Shi Y, Gao W, Lytle NK, Huang P, Yuan X, Dann AM. et al. Targeting LIF-mediated paracrine interaction for pancreatic cancer therapy and monitoring. Nature. 2019;569:131-5
163. Comito G, Giannoni E, Segura CP, Barcellos-de-Souza P, Raspollini MR, Baroni G. et al. Cancer-associated fibroblasts and M2-polarized macrophages synergize during prostate carcinoma progression. Oncogene. 2014;33:2423-31
164. Kim JH, Oh SH, Kim EJ, Park SJ, Hong SP, Cheon JH. et al. The role of myofibroblasts in upregulation of S100A8 and S100A9 and the differentiation of myeloid cells in the colorectal cancer microenvironment. Biochem Biophys Res Commun. 2012;423:60-6
165. Salmon H, Franciszkiewicz K, Damotte D, Dieu-Nosjean MC, Validire P, Trautmann A. et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J Clin Invest. 2012;122:899-910
166. Lin C, Wang L, Wang H, Yang L, Guo H, Wang X. Tanshinone IIA inhibits breast cancer stem cells growth in vitro and in vivo through attenuation of IL-6/STAT3/NF-kB signaling pathways. J Cell Biochem. 2013;114:2061-70
167. Luga V, Zhang L, Viloria-Petit AM, Ogunjimi AA, Inanlou MR, Chiu E. et al. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell. 2012;151:1542-56
168. Chen Y, Zeng C, Zhan Y, Wang H, Jiang X, Li W. Aberrant low expression of p85alpha in stromal fibroblasts promotes breast cancer cell metastasis through exosome-mediated paracrine Wnt10b. Oncogene. 2017;36:4692-705
169. Donnarumma E, Fiore D, Nappa M, Roscigno G, Adamo A, Iaboni M. et al. Cancer-associated fibroblasts release exosomal microRNAs that dictate an aggressive phenotype in breast cancer. Oncotarget. 2017;8:19592-608
170. Kinugasa Y, Matsui T, Takakura N. CD44 expressed on cancer-associated fibroblasts is a functional molecule supporting the stemness and drug resistance of malignant cancer cells in the tumor microenvironment. Stem Cells. 2014;32:145-56
171. Su S, Chen J, Yao H, Liu J, Yu S, Lao L. et al. CD10(+)GPR77(+) Cancer-Associated Fibroblasts Promote Cancer Formation and Chemoresistance by Sustaining Cancer Stemness. Cell. 2018;172:841-56 e16
172. Jiralerspong S, Goodwin PJ. Obesity and Breast Cancer Prognosis: Evidence, Challenges, and Opportunities. J Clin Oncol. 2016;34:4203-16
173. Fujisaki K, Fujimoto H, Sangai T, Nagashima T, Sakakibara M, Shiina N. et al. Cancer-mediated adipose reversion promotes cancer cell migration via IL-6 and MCP-1. Breast Cancer Res Treat. 2015;150:255-63
174. Dirat B, Bochet L, Dabek M, Daviaud D, Dauvillier S, Majed B. et al. Cancer-associated adipocytes exhibit an activated phenotype and contribute to breast cancer invasion. Cancer Res. 2011;71:2455-65
175. Brock CK, Hebert KL, Artiles M, Wright MK, Cheng T, Windsor GO. et al. A Role for Adipocytes and Adipose Stem Cells in the Breast Tumor Microenvironment and Regenerative Medicine. Front Physiol. 2021;12:751239
176. Arendt LM, McCready J, Keller PJ, Baker DD, Naber SP, Seewaldt V. et al. Obesity promotes breast cancer by CCL2-mediated macrophage recruitment and angiogenesis. Cancer Res. 2013;73:6080-93
177. Iliopoulos D, Hirsch HA, Wang G, Struhl K. Inducible formation of breast cancer stem cells and their dynamic equilibrium with non-stem cancer cells via IL6 secretion. Proc Natl Acad Sci U S A. 2011;108:1397-402
178. Bowers LW, Rossi EL, McDonell SB, Doerstling SS, Khatib SA, Lineberger CG. et al. Leptin Signaling Mediates Obesity-Associated CSC Enrichment and EMT in Preclinical TNBC Models. Mol Cancer Res. 2018;16:869-79
179. Thiagarajan PS, Zheng Q, Bhagrath M, Mulkearns-Hubert EE, Myers MG, Lathia JD. et al. STAT3 activation by leptin receptor is essential for TNBC stem cell maintenance. Endocr Relat Cancer. 2017;24:415-26
180. Sabol RA, Bowles AC, Cote A, Wise R, O'Donnell B, Matossian MD. et al. Leptin produced by obesity-altered adipose stem cells promotes metastasis but not tumorigenesis of triple-negative breast cancer in orthotopic xenograft and patient-derived xenograft models. Breast Cancer Res. 2019;21:67
181. Gao Y, Chen X, He Q, Gimple RC, Liao Y, Wang L. et al. Adipocytes promote breast tumorigenesis through TAZ-dependent secretion of Resistin. Proc Natl Acad Sci U S A. 2020;117:33295-304
182. Qureshi R, Picon-Ruiz M, Aurrekoetxea-Rodriguez I, Nunes de Paiva V, D'Amico M, Yoon H. et al. The Major Pre- and Postmenopausal Estrogens Play Opposing Roles in Obesity-Driven Mammary Inflammation and Breast Cancer Development. Cell Metab. 2020;31:1154-72 e9
183. Taliaferro-Smith L, Nagalingam A, Zhong D, Zhou W, Saxena NK, Sharma D. LKB1 is required for adiponectin-mediated modulation of AMPK-S6K axis and inhibition of migration and invasion of breast cancer cells. Oncogene. 2009;28:2621-33
184. Wang Y, Lam JB, Lam KS, Liu J, Lam MC, Hoo RL. et al. Adiponectin modulates the glycogen synthase kinase-3beta/beta-catenin signaling pathway and attenuates mammary tumorigenesis of MDA-MB-231 cells in nude mice. Cancer Res. 2006;66:11462-70
185. Liu XZ, Rulina A, Choi MH, Pedersen L, Lepland J, Takle ST. et al. C/EBPB-dependent adaptation to palmitic acid promotes tumor formation in hormone receptor negative breast cancer. Nat Commun. 2022;13:69
186. Dai JZ, Wang YJ, Chen CH, Tsai IL, Chao YC, Lin CW. YAP Dictates Mitochondrial Redox Homeostasis to Facilitate Obesity-Associated Breast Cancer Progression. Adv Sci (Weinh). 2022;9:e2103687
187. Pollmann MA, Shao Q, Laird DW, Sandig M. Connexin 43 mediated gap junctional communication enhances breast tumor cell diapedesis in culture. Breast Cancer Res. 2005;7:R522-34
188. Taftaf R, Liu X, Singh S, Jia Y, Dashzeveg NK, Hoffmann AD. et al. ICAM1 initiates CTC cluster formation and trans-endothelial migration in lung metastasis of breast cancer. Nat Commun. 2021;12:4867
189. Kabir AU, Subramanian M, Lee DH, Wang X, Krchma K, Wu J. et al. Dual role of endothelial Myct1 in tumor angiogenesis and tumor immunity. Sci Transl Med. 2021 13
190. Blanco R, Gerhardt H. VEGF and Notch in tip and stalk cell selection. Cold Spring Harb Perspect Med. 2013;3:a006569
191. Hoschl C, Blahos J, Bednar J, Vondra V. [Administration of hypothalamic thyrotropin releasing hormone by inhalation (preliminary report)]. Vnitr Lek. 1976;22:987-9
192. Du R, Lu KV, Petritsch C, Liu P, Ganss R, Passegue E. et al. HIF1alpha induces the recruitment of bone marrow-derived vascular modulatory cells to regulate tumor angiogenesis and invasion. Cancer Cell. 2008;13:206-20
193. Xing P, Dong H, Liu Q, Zhao T, Yao F, Xu Y. et al. ALDH1 Expression and Vasculogenic Mimicry Are Positively Associated with Poor Prognosis in Patients with Breast Cancer. Cell Physiol Biochem. 2018;49:961-70
194. Izawa Y, Kashii-Magaribuchi K, Yoshida K, Nosaka M, Tsuji N, Yamamoto A. et al. Stem-like Human Breast Cancer Cells Initiate Vasculogenic Mimicry on Matrigel. Acta Histochem Cytochem. 2018;51:173-83
195. Byrd TT, Fousek K, Pignata A, Szot C, Samaha H, Seaman S. et al. TEM8/ANTXR1-Specific CAR T Cells as a Targeted Therapy for Triple-Negative Breast Cancer. Cancer Res. 2018;78:489-500
196. Lee E, Pandey NB, Popel AS. Lymphatic endothelial cells support tumor growth in breast cancer. Sci Rep. 2014;4:5853
197. Chen HC, Joalland N, Bridgeman JS, Alchami FS, Jarry U, Khan MWA. et al. Synergistic targeting of breast cancer stem-like cells by human gammadelta T cells and CD8(+) T cells. Immunol Cell Biol. 2017;95:620-9
198. Fang Y, Wang L, Wan C, Sun Y, Van der Jeught K, Zhou Z. et al. MAL2 drives immune evasion in breast cancer by suppressing tumor antigen presentation. J Clin Invest. 2021 131
199. Little AC, Pathanjeli P, Wu Z, Bao L, Goo LE, Yates JA. et al. IL-4/IL-13 Stimulated Macrophages Enhance Breast Cancer Invasion Via Rho-GTPase Regulation of Synergistic VEGF/CCL-18 Signaling. Front Oncol. 2019;9:456
200. Jovanovic IP, Pejnovic NN, Radosavljevic GD, Pantic JM, Milovanovic MZ, Arsenijevic NN. et al. Interleukin-33/ST2 axis promotes breast cancer growth and metastases by facilitating intratumoral accumulation of immunosuppressive and innate lymphoid cells. Int J Cancer. 2014;134:1669-82
201. Jiang G, Tu J, Zhou L, Dong M, Fan J, Chang Z. et al. Single-cell transcriptomics reveal the heterogeneity and dynamic of cancer stem-like cells during breast tumor progression. Cell Death Dis. 2021;12:979
202. Tsao LC, Crosby EJ, Trotter TN, Agarwal P, Hwang BJ, Acharya C. et al. CD47 blockade augmentation of trastuzumab antitumor efficacy dependent on antibody-dependent cellular phagocytosis. JCI Insight. 2019 4
203. Huang H, Wang C, Liu F, Li HZ, Peng G, Gao X. et al. Reciprocal Network between Cancer Stem-Like Cells and Macrophages Facilitates the Progression and Androgen Deprivation Therapy Resistance of Prostate Cancer. Clin Cancer Res. 2018;24:4612-26
204. Raghavan S, Mehta P, Xie Y, Lei YL, Mehta G. Ovarian cancer stem cells and macrophages reciprocally interact through the WNT pathway to promote pro-tumoral and malignant phenotypes in 3D engineered microenvironments. J Immunother Cancer. 2019;7:190
205. Mitchem JB, Brennan DJ, Knolhoff BL, Belt BA, Zhu Y, Sanford DE. et al. Targeting tumor-infiltrating macrophages decreases tumor-initiating cells, relieves immunosuppression, and improves chemotherapeutic responses. Cancer Res. 2013;73:1128-41
206. Strachan DC, Ruffell B, Oei Y, Bissell MJ, Coussens LM, Pryer N. et al. CSF1R inhibition delays cervical and mammary tumor growth in murine models by attenuating the turnover of tumor-associated macrophages and enhancing infiltration by CD8(+) T cells. Oncoimmunology. 2013;2:e26968
207. Wang Y, Jia A, Bi Y, Wang Y, Yang Q, Cao Y. et al. Targeting Myeloid-Derived Suppressor Cells in Cancer Immunotherapy. Cancers (Basel). 2020 12
208. Alshetaiwi H, Pervolarakis N, McIntyre LL, Ma D, Nguyen Q, Rath JA. et al. Defining the emergence of myeloid-derived suppressor cells in breast cancer using single-cell transcriptomics. Sci Immunol. 2020 5
209. Lee WC, Hsu PY, Hsu HY. Stem cell factor produced by tumor cells expands myeloid-derived suppressor cells in mice. Sci Rep. 2020;10:11257
210. Peng D, Tanikawa T, Li W, Zhao L, Vatan L, Szeliga W. et al. Myeloid-Derived Suppressor Cells Endow Stem-like Qualities to Breast Cancer Cells through IL6/STAT3 and NO/NOTCH Cross-talk Signaling. Cancer Res. 2016;76:3156-65
211. Kumar S, Wilkes DW, Samuel N, Blanco MA, Nayak A, Alicea-Torres K. et al. DeltaNp63-driven recruitment of myeloid-derived suppressor cells promotes metastasis in triple-negative breast cancer. J Clin Invest. 2018;128:5095-109
212. Stein RG, Ebert S, Schlahsa L, Scholz CJ, Braun M, Hauck P. et al. Cognate Nonlytic Interactions between CD8(+) T Cells and Breast Cancer Cells Induce Cancer Stem Cell-like Properties. Cancer Res. 2019;79:1507-19
213. Wu C, Alman BA. Side population cells in human cancers. Cancer Lett. 2008;268:1-9
214. Xu Y, Dong X, Qi P, Ye Y, Shen W, Leng L. et al. Sox2 Communicates with Tregs Through CCL1 to Promote the Stemness Property of Breast Cancer Cells. Stem Cells. 2017;35:2351-65
215. Stapelberg M, Zobalova R, Nguyen MN, Walker T, Stantic M, Goodwin J. et al. Indoleamine-2,3-dioxygenase elevated in tumor-initiating cells is suppressed by mitocans. Free Radic Biol Med. 2014;67:41-50
216. Prendergast GC, Smith C, Thomas S, Mandik-Nayak L, Laury-Kleintop L, Metz R. et al. Indoleamine 2,3-dioxygenase pathways of pathogenic inflammation and immune escape in cancer. Cancer Immunol Immunother. 2014;63:721-35
217. Koh SY, Moon JY, Unno T, Cho SK. Baicalein Suppresses Stem Cell-Like Characteristics in Radio- and Chemoresistant MDA-MB-231 Human Breast Cancer Cells through Up-Regulation of IFIT2. Nutrients. 2019 11
218. Nguyen YT, Moon JY, Ediriweera MK, Cho SK. Phenethyl Isothiocyanate Suppresses Stemness in the Chemo- and Radio-Resistant Triple-Negative Breast Cancer Cell Line MDA-MB-231/IR Via Downregulation of Metadherin. Cancers (Basel). 2020 12
219. Pal D, Kolluru V, Chandrasekaran B, Baby BV, Aman M, Suman S. et al. Targeting aberrant expression of Notch-1 in ALDH(+) cancer stem cells in breast cancer. Mol Carcinog. 2017;56:1127-36
220. Choi HS, Kim SL, Kim JH, Deng HY, Yun BS, Lee DS. Triterpene Acid (3-O-p-Coumaroyltormentic Acid) Isolated From Aronia Extracts Inhibits Breast Cancer Stem Cell Formation through Downregulation of c-Myc Protein. Int J Mol Sci. 2018 19
221. Yang Y, Hao E, Pan X, Tan D, Du Z, Xie J. et al. Gomisin M2 from Baizuan suppresses breast cancer stem cell proliferation in a zebrafish xenograft model. Aging (Albany NY). 2019;11:8347-61
222. Sengupta S, Nagalingam A, Muniraj N, Bonner MY, Mistriotis P, Afthinos A. et al. Activation of tumor suppressor LKB1 by honokiol abrogates cancer stem-like phenotype in breast cancer via inhibition of oncogenic Stat3. Oncogene. 2017;36:5709-21
223. Li YW, Xu J, Zhu GY, Huang ZJ, Lu Y, Li XQ. et al. Apigenin suppresses the stem cell-like properties of triple-negative breast cancer cells by inhibiting YAP/TAZ activity. Cell Death Discov. 2018;4:105
224. Wanandi SI, Limanto A, Yunita E, Syahrani RA, Louisa M, Wibowo AE. et al. In silico and in vitro studies on the anti-cancer activity of andrographolide targeting survivin in human breast cancer stem cells. PLoS One. 2020;15:e0240020
225. Wang N, Wang Q, Tang H, Zhang F, Zheng Y, Wang S. et al. Direct inhibition of ACTN4 by ellagic acid limits breast cancer metastasis via regulation of beta-catenin stabilization in cancer stem cells. J Exp Clin Cancer Res. 2017;36:172
226. Rajabi S, Shojaee M, Malmir A, Rezaei Tavirani M, Noori S. Anti-Breast Cancer Activities of 8-Hydroxydaidzein by Targeting Breast Cancer Stem-Like Cells. J Pharm Pharm Sci. 2020;23:47-57
227. Li Y, Zhang T, Korkaya H, Liu S, Lee HF, Newman B. et al. Sulforaphane, a dietary component of broccoli/broccoli sprouts, inhibits breast cancer stem cells. Clin Cancer Res. 2010;16:2580-90
228. Kim SH, Singh SV. Mammary cancer chemoprevention by withaferin A is accompanied by in vivo suppression of self-renewal of cancer stem cells. Cancer Prev Res (Phila). 2014;7:738-47
229. Kim SH, Sehrawat A, Singh SV. Dietary chemopreventative benzyl isothiocyanate inhibits breast cancer stem cells in vitro and in vivo. Cancer Prev Res (Phila). 2013;6:782-90
230. Jiang F, Li Y, Mu J, Hu C, Zhou M, Wang X. et al. Glabridin inhibits cancer stem cell-like properties of human breast cancer cells: An epigenetic regulation of miR-148a/SMAd2 signaling. Mol Carcinog. 2016;55:929-40
231. Wang N, Wang Z, Wang Y, Xie X, Shen J, Peng C. et al. Dietary compound isoliquiritigenin prevents mammary carcinogenesis by inhibiting breast cancer stem cells through WIF1 demethylation. Oncotarget. 2015;6:9854-76
232. Omene CO, Wu J, Frenkel K. Caffeic Acid Phenethyl Ester (CAPE) derived from propolis, a honeybee product, inhibits growth of breast cancer stem cells. Invest New Drugs. 2012;30:1279-88
233. Xu Z, Guo D, Jiang Z, Tong R, Jiang P, Bai L. et al. Novel HER2-Targeting Antibody-Drug Conjugates of Trastuzumab Beyond T-DM1 in Breast Cancer: Trastuzumab Deruxtecan(DS-8201a) and (Vic-)Trastuzumab Duocarmazine (SYD985). Eur J Med Chem. 2019;183:111682
234. Elshiaty M, Schindler H, Christopoulos P. Principles and Current Clinical Landscape of Multispecific Antibodies against Cancer. Int J Mol Sci. 2021 22
235. Stivarou T, Stellas D, Vartzi G, Thomaidou D, Patsavoudi E. Targeting highly expressed extracellular HSP90 in breast cancer stem cells inhibits tumor growth in vitro and in vivo. Cancer Biol Ther. 2016;17:799-812
236. Sharma A, Paranjape AN, Rangarajan A, Dighe RR. A monoclonal antibody against human Notch1 ligand-binding domain depletes subpopulation of putative breast cancer stem-like cells. Mol Cancer Ther. 2012;11:77-86
237. Masiero M, Li D, Whiteman P, Bentley C, Greig J, Hassanali T. et al. Development of Therapeutic Anti-JAGGED1 Antibodies for Cancer Therapy. Mol Cancer Ther. 2019;18:2030-42
238. Kaur S, Elkahloun AG, Singh SP, Chen QR, Meerzaman DM, Song T. et al. A function-blocking CD47 antibody suppresses stem cell and EGF signaling in triple-negative breast cancer. Oncotarget. 2016;7:10133-52
239. Ly S, Anand V, El-Dana F, Nguyen K, Cai Y, Cai S. et al. Anti-GD2 antibody dinutuximab inhibits triple-negative breast tumor growth by targeting GD2(+) breast cancer stem-like cells. J Immunother Cancer. 2021 9
240. Zhang S, Zhang H, Ghia EM, Huang J, Wu L, Zhang J. et al. Inhibition of chemotherapy resistant breast cancer stem cells by a ROR1 specific antibody. Proc Natl Acad Sci U S A. 2019;116:1370-7
241. Chen JH, Huang WC, Bamodu OA, Chang PM, Chao TY, Huang TH. Monospecific antibody targeting of CDH11 inhibits epithelial-to-mesenchymal transition and represses cancer stem cell-like phenotype by up-regulating miR-335 in metastatic breast cancer, in vitro and in vivo. BMC Cancer. 2019;19:634
242. Bender KW, Zipfel C. Plant G-protein activation: connecting to plant receptor kinases. Cell Res. 2018;28:697-8
243. Ohlfest JR, Zellmer DM, Panyam J, Swaminathan SK, Oh S, Waldron NN. et al. Immunotoxin targeting CD133(+) breast carcinoma cells. Drug Deliv Transl Res. 2013;3:195-204
244. Swaminathan SK, Roger E, Toti U, Niu L, Ohlfest JR, Panyam J. CD133-targeted paclitaxel delivery inhibits local tumor recurrence in a mouse model of breast cancer. J Control Release. 2013;171:280-7
245. Bao H, Wu D. Current Status of Leukemia Cytotherapy - Exploitation with Immune Cells. Curr Stem Cell Res Ther. 2017;12:188-96
246. Wang Z, Cao YJ. Adoptive Cell Therapy Targeting Neoantigens: A Frontier for Cancer Research. Front Immunol. 2020;11:176
247. Chen Q, Cui XX, Liang PF, Dou JX, Liu ZY, Sun WW. Immunotherapy with dendritic cells and cytokine-induced killer cells for MDA-MB-231 breast cancer stem cells in nude mice. Am J Transl Res. 2016;8:2947-55
248. Battula VL, Shi Y, Evans KW, Wang RY, Spaeth EL, Jacamo RO. et al. Ganglioside GD2 identifies breast cancer stem cells and promotes tumorigenesis. J Clin Invest. 2012;122:2066-78
249. Seitz CM, Schroeder S, Knopf P, Krahl AC, Hau J, Schleicher S. et al. GD2-targeted chimeric antigen receptor T cells prevent metastasis formation by elimination of breast cancer stem-like cells. Oncoimmunology. 2020;9:1683345
250. Wang T, Wu X, Guo C, Zhang K, Xu J, Li Z. et al. Development of Inhibitors of the Programmed Cell Death-1/Programmed Cell Death-Ligand 1 Signaling Pathway. J Med Chem. 2019;62:1715-30
251. Baah S, Laws M, Rahman KM. Antibody-Drug Conjugates-A Tutorial Review. Molecules. 2021 26
252. Murphy AG, Zheng L. Small molecule drugs with immunomodulatory effects in cancer. Hum Vaccin Immunother. 2015;11:2463-8
253. Bonuccelli G, Peiris-Pages M, Ozsvari B, Martinez-Outschoorn UE, Sotgia F, Lisanti MP. Targeting cancer stem cell propagation with palbociclib, a CDK4/6 inhibitor: Telomerase drives tumor cell heterogeneity. Oncotarget. 2017;8:9868-84
254. Hu D, Gur M, Zhou Z, Gamper A, Hung MC, Fujita N. et al. Interplay between arginine methylation and ubiquitylation regulates KLF4-mediated genome stability and carcinogenesis. Nature Communications. 2015 6
255. Wang X, Qiu T, Wu Y, Yang C, Li Y, Du G. et al. Arginine methyltransferase PRMT5 methylates and stabilizes KLF5 via decreasing its phosphorylation and ubiquitination to promote basal-like breast cancer. Cell Death Differ. 2021;28:2931-45
256. Zhou Z, Feng Z, Hu D, Yang P, Gur M, Bahar I. et al. A novel small-molecule antagonizes PRMT5-mediated KLF4 methylation for targeted therapy. EBioMedicine. 2019;44:98-111
257. Cuyas E, Gumuzio J, Verdura S, Brunet J, Bosch-Barrera J, Martin-Castillo B. et al. The LSD1 inhibitor iadademstat (ORY-1001) targets SOX2-driven breast cancer stem cells: a potential epigenetic therapy in luminal-B and HER2-positive breast cancer subtypes. Aging (Albany NY). 2020;12:4794-814
258. Zhou H, Yu C, Kong L, Xu X, Yan J, Li Y. et al. B591, a novel specific pan-PI3K inhibitor, preferentially targets cancer stem cells. Oncogene. 2019;38:3371-86
259. Dattilo R, Mottini C, Camera E, Lamolinara A, Auslander N, Doglioni G. et al. Pyrvinium Pamoate Induces Death of Triple-Negative Breast Cancer Stem-Like Cells and Reduces Metastases through Effects on Lipid Anabolism. Cancer Res. 2020;80:4087-102
260. Sand A, Piacsek M, Donohoe DL, Duffin AT, Riddell GT, Sun C. et al. WEE1 inhibitor, AZD1775, overcomes trastuzumab resistance by targeting cancer stem-like properties in HER2-positive breast cancer. Cancer Lett. 2020;472:119-31
261. Kim DA, Choi HS, Ryu ES, Ko J, Shin HS, Lee JM. et al. Tannic acid attenuates the formation of cancer stem cells by inhibiting NF-kappaB-mediated phenotype transition of breast cancer cells. Am J Cancer Res. 2019;9:1664-81
262. Zhang X, Li F, Zheng Y, Wang X, Wang K, Yu Y. et al. Propofol Reduced Mammosphere Formation of Breast Cancer Stem Cells via PD-L1/Nanog In Vitro. Oxid Med Cell Longev. 2019;2019:9078209
263. De Francesco EM, Ozsvari B, Sotgia F, Lisanti MP. Dodecyl-TPP Targets Mitochondria and Potently Eradicates Cancer Stem Cells (CSCs): Synergy With FDA-Approved Drugs and Natural Compounds (Vitamin C and Berberine). Front Oncol. 2019;9:615
264. Safi R, Hamade A, Bteich N, El Saghir J, Assaf MD, El-Sabban M. et al. A ferutinin analogue with enhanced potency and selectivity against ER-positive breast cancer cells in vitro. Biomed Pharmacother. 2018;105:267-73
265. Cai P, Xiao Z, Pan T, Wen X, Cao J, Ouyang B. Lx2-32c inhibits the formation of mammosphere from MDA-MB-231 cells and induces apoptosis involving in down-regulating FoxM1. Biomed Pharmacother. 2018;102:1176-81
266. Sato K, Padgaonkar AA, Baker SJ, Cosenza SC, Rechkoblit O, Subbaiah D. et al. Simultaneous CK2/TNIK/DYRK1 inhibition by 108600 suppresses triple negative breast cancer stem cells and chemotherapy-resistant disease. Nat Commun. 2021;12:4671
267. Scatena C, Roncella M, Di Paolo A, Aretini P, Menicagli M, Fanelli G. et al. Doxycycline, an Inhibitor of Mitochondrial Biogenesis, Effectively Reduces Cancer Stem Cells (CSCs) in Early Breast Cancer Patients: A Clinical Pilot Study. Front Oncol. 2018;8:452
268. Pushpakom S, Iorio F, Eyers PA, Escott KJ, Hopper S, Wells A. et al. Drug repurposing: progress, challenges and recommendations. Nat Rev Drug Discov. 2019;18:41-58
269. Yi H, Wu M, Zhang Q, Lu L, Yao H, Chen S. et al. Reversal of HER2 Negativity: An Unexpected Role for Lovastatin in Triple-Negative Breast Cancer Stem Cells. J Cancer. 2020;11:3713-6
270. An H, Kim JY, Lee N, Cho Y, Oh E, Seo JH. Salinomycin possesses anti-tumor activity and inhibits breast cancer stem-like cells via an apoptosis-independent pathway. Biochem Biophys Res Commun. 2015;466:696-703
271. Lu Y, Ma W, Mao J, Yu X, Hou Z, Fan S. et al. Salinomycin exerts anticancer effects on human breast carcinoma MCF-7 cancer stem cells via modulation of Hedgehog signaling. Chem Biol Interact. 2015;228:100-7
272. Versini A, Colombeau L, Hienzsch A, Gaillet C, Retailleau P, Debieu S. et al. Salinomycin Derivatives Kill Breast Cancer Stem Cells by Lysosomal Iron Targeting. Chemistry. 2020;26:7416-24
273. Liu Z, Mi M, Zheng X, Zhang C, Zhu F, Liu T. et al. miR-30a/SOX4 Double Negative Feedback Loop is modulated by Disulfiram and regulates EMT and Stem Cell-like properties in Breast Cancer. J Cancer. 2021;12:5053-65
274. Lee H, Park HJ, Park CS, Oh ET, Choi BH, Williams B. et al. Response of breast cancer cells and cancer stem cells to metformin and hyperthermia alone or combined. PLoS One. 2014;9:e87979
275. Liu R, Shi P, Nie Z, Liang H, Zhou Z, Chen W. et al. Mifepristone Suppresses Basal Triple-Negative Breast Cancer Stem Cells by Down-regulating KLF5 Expression. Theranostics. 2016;6:533-44
276. Liu R, Chen H, Zhao P, Chen CH, Liang H, Yang C. et al. Mifepristone Derivative FZU-00,003 Suppresses Triple-negative Breast Cancer Cell Growth partially via miR-153-KLF5 axis. Int J Biol Sci. 2020;16:611-9
277. Lin Y, Liu R, Zhao P, Ye J, Zheng Z, Huang J. et al. Discovery of novel mifepristone derivatives via suppressing KLF5 expression for the treatment of triple-negative breast cancer. Eur J Med Chem. 2018;146:354-67
278. Oh E, Kim YJ, An H, Sung D, Cho TM, Farrand L. et al. Flubendazole elicits anti-metastatic effects in triple-negative breast cancer via STAT3 inhibition. Int J Cancer. 2018;143:1978-93
279. Kulkarni JA, Witzigmann D, Thomson SB, Chen S, Leavitt BR, Cullis PR. et al. The current landscape of nucleic acid therapeutics. Nat Nanotechnol. 2021;16:630-43
280. Yamakawa K, Nakano-Narusawa Y, Hashimoto N, Yokohira M, Matsuda Y. Development and Clinical Trials of Nucleic Acid Medicines for Pancreatic Cancer Treatment. Int J Mol Sci. 2019 20
281. Witt K, Ligtenberg MA, Conti L, Lanzardo S, Ruiu R, Wallmann T. et al. Cripto-1 Plasmid DNA Vaccination Targets Metastasis and Cancer Stem Cells in Murine Mammary Carcinoma. Cancer Immunol Res. 2018;6:1417-25
282. Donofrio G, Tebaldi G, Lanzardo S, Ruiu R, Bolli E, Ballatore A. et al. Bovine herpesvirus 4-based vector delivering the full length xCT DNA efficiently protects mice from mammary cancer metastases by targeting cancer stem cells. Oncoimmunology. 2018;7:e1494108
283. Wang T, Gantier MP, Xiang D, Bean AG, Bruce M, Zhou SF. et al. EpCAM Aptamer-mediated Survivin Silencing Sensitized Cancer Stem Cells to Doxorubicin in a Breast Cancer Model. Theranostics. 2015;5:1456-72
284. Ding AJ, Zheng SQ, Huang XB, Xing TK, Wu GS, Sun HY. et al. Current Perspective in the Discovery of Anti-aging Agents from Natural Products. Nat Prod Bioprospect. 2017;7:335-404
Author contact
Corresponding authors: Ceshi Chen, E-mail: chenckiz.ac.cn or Suling Liu, E-mail: sulingedu.cn.

 Global reach, higher impact
Global reach, higher impact