ISSN: 1449-2288
Int J Biol Sci 2023; 19(4):1009-1023. doi:10.7150/ijbs.73570 This issue Cite
Research Paper
N6-Methyladenosine Modification of ANLN Enhances Hepatocellular Carcinoma Bone Metastasis
1. Third Department of Hepatic Surgery, Third Affiliated Hospital, Naval Medical University, Shanghai 200438, China.
2. Key Laboratory of Signaling Regulation and Targeting Therapy of Liver Cancer (SMMU), Ministry of Education, Shanghai 200438, China.
3. Department of Organization Sample Bank, Shanghai Key Laboratory of Hepatobiliary Tumor Biology (EHBH), Shanghai 200438, China.
4. Department of Hepato-Pancreato-Biliary Centers, Zhong Da Hospital, School of Medicine, Southeast University, Nanjing 210009, China.
5. Department of General Surgery, The Second Affiliated Hospital of Nanchang University, Nanchang, 330000, China.
6. Department of Hepatobiliary and Pancreatic Surgery, The 10th People's Hospital, Tongji University, Shanghai 200433, China.
7. Division of Hematology and Medical Oncology, Perlmutter Cancer Center, NYU Langone Health, New York, NY, USA.
8. Division of Gastroenterology, Department of Medicine, Kurume University School of Medicine, Kurume, Japan.
9. Hrishikesh Samant, Division of Gastroenterology and Hepatology, LSU Health Science Center, Shreveport, LA, USA.
10. State Key Laboratory of Oncogenes and Related Genes, Shanghai Cancer Institute, Renji Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200032, China.
*Equal contributions to this work.
Abstract
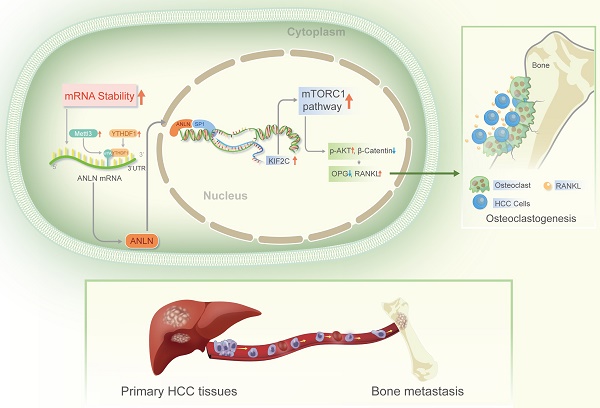
Bones are categorized as the second most prevalent location of extra-hepatic metastasis in Hepatocellular Carcinoma (HCC), which is linked to an extremely poor prognosis due to limited therapeutic options. N6-methyladenosine (m6A) is a prominent modification involved in HCC, but the exact mechanisms on how m6A modifications induce HCC bone metastases (BM) remain unclear. The key modulators responsible for the abundant m6A RNA modification-induced HCC BM was found to be the METTL3 and YTHDF1. The expression of Anillin actin-binding protein (ANLN) was dramatically higher in HCC with BM tissues, and its messenger RNA (mRNA) stability was enhanced via m6A epitranscriptomic regulation by METTL3 and YTHDF1. High METTL3 and YTHDF1 expression along with nuclear ANLN protein was clinically correlated with BM in HCC patients. Furthermore, HCC BM was attributed to over-expression of nuclear ANLN forming a transcriptional complex with SP1 which enhanced KIF2C transcriptional activity to activate the mTORC1 pathway, therefore increased the expression of RANKL and disproportionated RANKL-OPG expression in bone microenvironment leading to malignant neoplasms invade bone tissue. In addition, inhibition of ANLN m6A modification by DZNeP attenuated HCC BM. This data provides meaningful understanding of the modulation and association of m6A epitranscriptomic-regulated BM in HCC, and moreover, defines potentially valuable therapeutic targets.
Keywords: hepatocellular carcinoma, bone metastasis, N6-methyladenosine, anillin actin binding protein, DZNeP

 Global reach, higher impact
Global reach, higher impact