Impact Factor ISSN: 1449-2288
Int J Biol Sci 2011; 7(7):1027-1036. doi:10.7150/ijbs.7.1027 This issue Cite
Research Paper
Circulating and Tumor-Infiltrating Foxp3+ Regulatory T Cell Subset in Chinese Patients with Extranodal NK/T Cell Lymphoma
1. State Key Laboratory of Oncology in South China, Sun Yat-Sen University Cancer Center, Guangzhou, 510060, China
2. Department of Medical Oncology, Sun Yat-Sen University Cancer Center, Guangzhou, 510060, China
3. Department of Biotherapy Center, Sun Yat-Sen University Cancer Center, Guangzhou, 510060, China
4. Department of Experimental Research, Sun Yat-Sen University Cancer Center, Guangzhou, 510060, China
Received 2011-6-29; Accepted 2011-8-11; Published 2011-8-19
Abstract
Foxp3+ regulatory T lymphocytes (Tregs) usually act as an immune suppressor and correlate with poorer survival in malignancies. This study aims to investigate the distribution and characterization of Foxp3+ subset in peripheral blood mononuclear cells (PBMCs) and tumor tissues from extranodal NK/T cell lymphoma (ENKTL). Our study showed the percentage of Foxp3+ subset from PBMC was significantly higher than that of healthy individuals (P<0.001). The Foxp3+ subset from PBMCs expressed CD45RO, CTLA4, GITR, CCR7, and had an IL-10highIFNγ+TGFβ+IL-2lowIL-17low cytokine secreting phenotype. Interestingly, the existence of EBV antigen-specific CD8+Foxp3+ Tregs was discovered in ENKTL. Furthermore, the high density of Foxp3+ TILs was associated with improved progression-free survival (PFS) in ENKTL patients (P<0.05). Collectively, our study implicates that EBV antigens could induce antigen-specific CD8+Foxp3+ Tregs in ENKTL, and Foxp3+ TILs is an independent factor for PFS in ENKTL.
Keywords: Foxp3+ regulatory T cells, Extranodal NK/T cell lymphoma, tumor infiltrating lymphocytes, EBV
Introduction
Extranodal NK/T-cell lymphoma (ENKTL), nasal type referred to as angiocentric lymphoma according to the REAL classification[1], is recognized as a distinctive clinicopathologic entity and Epstein-Barr virus (EBV) associated disease. A typical phenotype of ENKTL is CD2+, CD3ε+, CD56+, cytotoxic molecules positive and EBV encoded RNA (EBERs) positive[2]. ENKTL patients are often refractory to radiotherapy and chemotherapy, and show aggressive behaviors with poor outcomes. EBV is presented in the malignant cells of ENKTL as latent type II infection, irrespective of geographical origin[3], and infiltration of reactive inflammatory cells, including small lymphocytes, plasma cells, and histiocytes for the neo-virus antigens expressed on malignant cells are dectected[2]. However, it is still poorly understood the role of specific circulating and tumor infiltrating lymphocyte subsets in the disease progression of ENKTL.
CD8+ cytotoxic T lymphocytes (CTLs) play a major role in controlling and eradicating the virus-infected cells, and the density of CD8+ TILs is associated with prognosis of patients with cancers[4-8]. Furthermore, the responses of CD4+ T cell to viral infections are critical to maintain virus-specific memory CTLs[9]. CD4+ T cells are comprised of the traditional Th1, Th2 helper T cell subsets, Foxp3+ regulatory T cell (Tregs) and IL-17-producing Th17 cells[9]. For the suppressive function to immune response and maintenance the immune tolerance, Foxp3+ Tregs is usually identified as a suppressor to anti-tumor immunity and associated with poorer clinical outcome of patients with some solid tumors[10-12]. However, recently studies suggested that the density of tumor infiltrating Foxp3+ Tregs was associated with improved outcome of patients with EBV positive malignancies including Hodgkin lymphoma, nasopharyngeal carcinoma (NPC) and other cancer, such as follicular lymphoma[8, 13, 14], but it is still unclear for the distribution and characterization of Tregs and its association with clinical outcome in patients with EBV positive ENKTL. In this study, to identify the Foxp3+ Tregs subset in circulating lymphocytes and tumor tissues in patients with ENKTL, we investigated the biological features of Foxp3+ Tregs subset in PBMCs, and the correlation with the density of different lymphocyte subset including CD4+ TILs, CD8+ TILs and Foxp3+ TILs in tumor tissues and clinicopathological characteristics and survival of ENKTL patients.
Materials and Methods
Patients
Thirty-five newly diagnosed ENKTL cases were included in this study, peripheral blood was collected from 10 patients and 10 age-matched healthy donors for isolating lymphocytes, and paraffin-embedded specimen were collected from 27 cases at Sun Yat-Sen University Cancer Center from 2001 to 2009. Tumor tissue and paired blood were obtained from two cases. Informed consent was obtained from each subject. This study was conducted in accordance with the Helsinki Declaration, and approved by the Research Ethic Committee of Sun Yat-Sen University Cancer Center. All healthy donors provided written informed consent for their participation.
All patients followed the criteria described in the previous report[15]. The demographic characteristic of all eligible patients was showed in Table 1 and Supplementary Material: Table S1. There were 25 males and 10 females in 35 eligible cases. The median age was 41 years (Range 13-68 years). Twelve patients (34.3%) had B symptom and 22 cases were localized disease (Ann Arbor stage I and II, 62.8%). Most cases' performance status was 0-1 (n=26, 74.3%) and the remains' were 2-4 (n=9, 25.7%). The disease primarily involved the upper aerodigestive tract (UAT) in 24 cases (68.6%) and non-UAT in 11 cases (31.4%); and 16 cases (45.7%) had elevated serum lactate dehydrogenase (LDH). Seventeen cases were categorized as 0-1 and 18 cases as 2-5 for International Prognostic Index (IPI). Thirteen cases (37.1%) were low risk, the other 22 cases (62.9%) were in intermediated or high-risk group, based on Korean Prognostic Index (KPI); while 25 cases (71.4%) were low risk, and the other 10 cases (28.6%) were intermediated to high risk, according to peripheral T cell prognostic index (PIT) model. All cases in this study were EBV positive by EBERs (EBV encoding RNAs, EBERs) in situ hybridization.
Clinical charactertics of 35 cases of newly diagnosed extranodal NK/T cell lymphoma
| Characteristics | Case (%) |
|---|---|
| Gender | |
| Male | 25 (71.4) |
| Female | 10 (28.6) |
| Age | |
| Median | 41 |
| Range | 13-68 |
| B symptom | |
| No | 23 (65.7) |
| Yes | 12 (34.3) |
| Performance status | |
| 0-1 | 26 (74.3) |
| ≥2 | 9 (25.7) |
| Involved field | |
| UAT | 24 (68.6) |
| non-UAT | 11 (31.4) |
| Ann-Arbor Stage | |
| I | 7 (20.0) |
| II | 15 (42.8) |
| III | 5 (14.3) |
| IV | 8 (22.9) |
| LDH | |
| Normal | 19 (54.3) |
| Elevated | 16 (45.7) |
| IPI score | |
| 0-1 | 17 (48.5) |
| 2-5 | 18 (51.5) |
| KPI score | |
| 0-1 | 13 (37.1) |
| 2-4 | 22 (62.9) |
| PIT score | |
| 0-1 | 25 (71.4) |
| 2-4 | 10 (28.6) |
Note: UAT, upper aerodigestive tract; LDH, lactate dehydrogenase; IPI, International Prognostic Index; KPI, Korean Prognostic Index; PTI: Peripheral T-cell lymphoma Prognostic Index
Antibodies and flow cytometer analysis (FACS)
The following antibodies were used for staining single cell suspension ex vivo: anti-CD4 (Clone RPA-T4), anti-CD25 (BC96), anti-CCR6 (R6H1), anti-IL17 (SCPL1362), anti-FOXP3 (236A/E7), anti-IFNγ (B27), anti-IL-2 (M11-17H12), anti-IL-10 (JES3-19F1), anti-CCR7 (3D12), anti-CD45RO (HI100), anti-CTLA-4 (eBio20A) and anti-GITR (eBioAITR) antibodies conjugated with FITC, PE, APC or PE-Cy7 were purchased from BD Biosciences (San Jose, CA, USA) or eBioscience (San Diego, CA, USA). Recombinant human IL-2 was purchased from R & D System (Boston, MS, USA).
The expression of markers on T cells was investigated by FACS analysis after surface staining or intracellular staining with anti-human-specific Abs conjugated with different fluorescence dyes. Intracellular staining for cytokines was performed on T cells stimulated by phorbol12-myristate13-acetate (PMA) and inomycin in the presence of brefeldin A (10 μg/ml, Sigma-Aldrich) for 4 hours. Cells were stained with fluorescent antibodies in 0.1% saponin permeablization buffer and detected on a FC500 flow cytometer machine. All data were analyzed using CXP analysis software (Beckman Coulter, Inc. Fullerton, CA, USA).
The frequency of T cell specific for HLA-A2 restricted epitopes in LMP1 and LMP2 was analyzed by staining with HLA-A2 tetramers assembled with synthetic peptides origin from LMP1 YLQQNWWTL and LMP2 FLYALALLL, LLWTLVVLL, GLGTLGAAI (Guangzhou Taimo Corporation, Guangzhou, China). The samples were stained with 1 μg/ml phycoerythrin (PE)-labeled tetrameric complex firstly, and then co-stained with anti-CD8-FITC and anti-Foxp3-APC antibodies followed by fixation in 0.5% paraformaldehyde for 20 mins. For each sample, 105 cells were analyzed using the FC500 flow cytometer machine and CXP analysis software.
CD4-positive and CD8-positive Tregs sorting and T cell proliferation
CD4+CD25+ Tregs or CD8+CD25+ Tregs subsets from PBMCs of ENKTL were isolated with the anti-CD4, anti-CD8 and anti-CD25 staining and FACS sorting on FACS sorting machine MoFlo XDP Cell Sorter (Beckman Coulter, Inc. Fullerton, CA, USA). The sorted cells were expanded in IL-2 medium in vitro, and T cell proliferation assays were performed as described previously[16]. In brief, 105 CD4+ naïve T cells were labelled with 5- or 6-(N-Succinimidyloxycarbonyl)-3',6'-O,O'-diacetylfluorescein (CSFE, Molecular Probes/Invitrogen, Burlington, CA) and cocultured with different ratios of CD4+ Tregs cells in u-bottom 96-well human OKT-3 (1ug/ml) coated plate for 5 days in no IL-2 medium. The proliferation of CSFE-labeled CD4+ naïve T cells was detected by FACS analyses gating on the CSFE-labelled cell population.
Immunohistochemical staining (IHC)
The paraffin-embedded tumor sections were used for IHC. After antigen retrieval by microwave heating (95°C for 20 min), tissue sections were incubated with primary antibodies including mouse monoclonal anti-human CD4 (Clone 4B12, DAKO, Glostrup, Denmark; diluted at 1:80), mouse monoclonal anti-human CD8 (Clone CD8/144B, DAKO, Glostrup, Denmark; diluted at 1:40), mouse monoclonal anti-human Foxp3 (Clone 221D/D3, Santa Cruz Biotechnology, CA, USA, diluted at 1:100), mouse monoclonal anti-LMP2A was provided by Prof. Zeng M (diluted at 1:200)[17] were incubated at 4°C overnight. Then bind to non-biotin horseradish peroxidase detection system (Dako, Glostrup, Denmark). Data was obtained by counting the positively stained lymphocytes in 5 separate 400× high-power microscopic fields (HPFs) and calculating the mean number of positively stained cells per HPF. Mouse IgG1 (DAKO, Glostrup, Denmark) negative control staining was generated and evaluated. LMP2A expression was defined as negative for score 0-2, positive for score 3-5, according to the number and intensity of stained cell.
Statistical analysis
All data was analyzed with SPSS 16.0 software (SPSS, Chicago, IL, USA). The median value was applied to cut off the subgroups of all immunohistochemical variants in our data. The correlation with lymphocytic variables and clinical characteristics was estimated by Pearson chi test or Fisher's Exact test. Two-tailed P<0.05 was set as statistical significance. Survival rate was calculated by the Kaplan-Meier method and tested by the log-rank analysis. Cox regression analysis was applied to adjusted analysis.
Results
Increased proportion of circulating CD4+Foxp3+ Tregs and CD8+Foxp3+ Tregs was determined in ENKTL patients
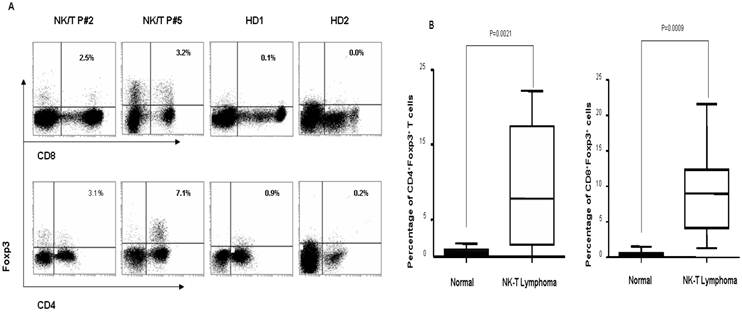
To identify the Foxp3+ Treg subset in peripheral blood of ENKTL patients, we firstly compared the proportion of Foxp3+ Tregs in CD4+ and CD8+ subsets in PBMCs from ENKTL patients and controls (healthy individuals) by FACS analyses based on the expression of Foxp3 protein. As shown in Fig 1A and 1B, the percentage of CD4+Foxp3+ Tregs (mean=9.00±7.61%; n=10) and of CD8+Foxp3+ Tregs (mean=8.97±6.17%; n=10) in PBMCs from ENKTL patients was significantly higher compared to that of in healthy individuals (mean=0.75±0.43% and mean=0.48±0.44%, respectively; n=20, both P<0.001).
The distribution of CD4+Foxp3+ Tregs and CD8+Foxp3+ Tregs in PBMCs of ENKTL patients and healthy donors. A. Representative dot plots for CD4, Foxp3 and CD8, Foxp3 in PBMCs from 2 ENKTL patients and 2 health donors. B. The dramatically increase of CD4+Foxp3+ Tregs and CD8+Foxp3+ Tregs was found out in PBMCs of ENKTL (n=10) compared with that of in PBMCs of healthy donors (n=10, P<0.005).
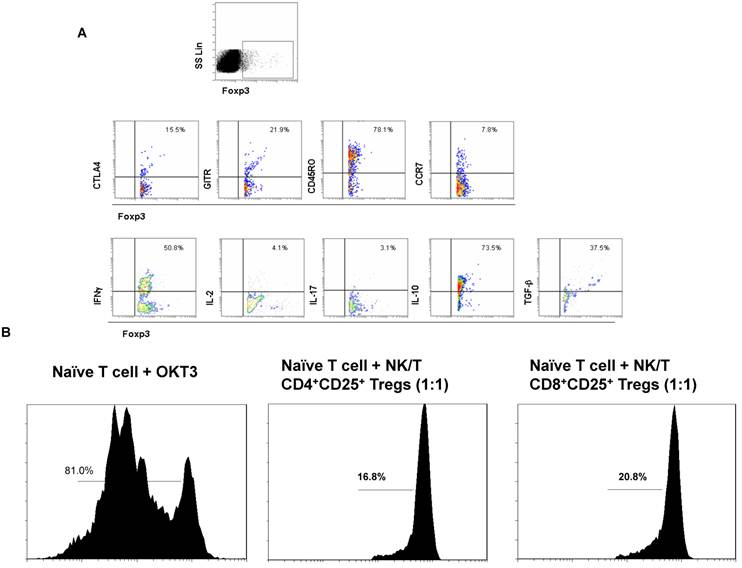
Next we sought to explore the phenotypic and functional features of Foxp3+ Tregs subset in PBMCs of ENKTL patients. The Foxp3+ Tregs subset expressed high level of CD45RO and middle levels of CCR7, CTLA4 and GITR that indicated most Foxp3+ Tregs in ENKTL were from memory cell population and expressed the special markers CTLA4 and GITR for Tregs (Fig 2A). The cytokine expression profiles of the Foxp3+ Tregs subset expressed high levels of IL-10 and TGFβ, low levels of IL-2 and IL-17 (Tregs phenotype). Interestingly, the level of IFN-γ in Foxp3+ Tregs subset in PBMCs from ENKTL was much higher than that of in PBMCs from healthy donors (data not shown). In order to test the suppressive function of Tregs subset in PBMCs from ENKTL patients, we sorted the CD4+CD25high and CD8+CD25high subsets and expanded these two cell subsets in vitro. Cells expressing high levels of CD25 in PBMCs were usually qualified as Tregs in this study followed others' report[18]. The suppressive ability of these two subsets to CD4+ naïve T cells in vitro was detected after expansion in vitro. Both CD4+CD25+ T cell subset and CD8+CD25+ T cell subset could inhibit the proliferation of CD4+ naïve T cells shown in Fig 2B.
The biological characteristics and function of Foxp3+ Tregs subset. A. The flow cytometer profiles are representatives from the stainings for CTLA, GITR, CD45RO, CCR7, IFNγ, IL-2, IL-17, IL-10 and TGFβ in Foxp3+ Treg subset. B. The suppressive ability of CD4+ Tregs and CD8+ Tregs to naïve CD4+ T cells in vitro was measured by the T cell proliferation experiment. CSFE-labeled naïve CD4+ T cells were alone or co-cultured with CD8+ Tregs cells at ratio of 1:1 in the in complete RPMI1640 medium without IL-2 in OKT3-coated 96-well plate for 5 days and analyzed by FACS detection.
The EBV LMP1 and LMP2 epitopic antigen-specific CD8+ effectors and CD8+Foxp3+ Tregs was identified in ENKTL patients
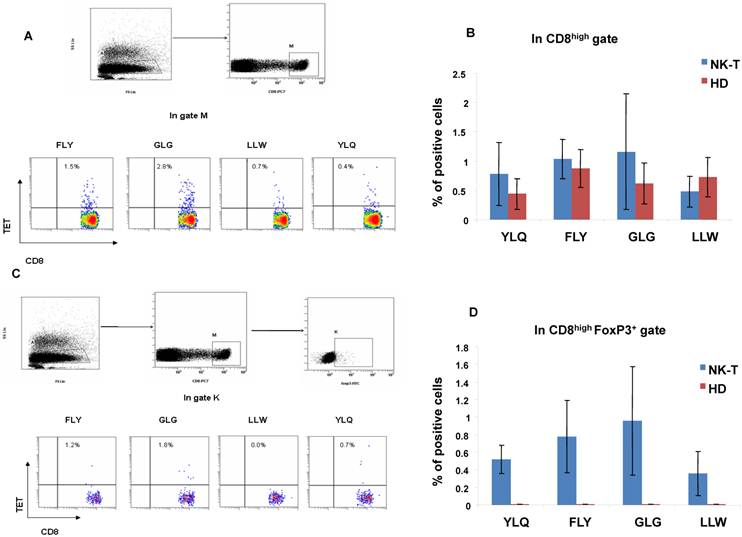
The ENKTL tumor cells can express EBV latent type II antigens including LMP1, LMP2, EBNA1 and BARF0 as nasopharyngeal carcinoma or EBV positive B cell lymphoma[19, 20]. In order to address if there are pathogenic CD8+ Treg cells as well as CD8+ effector T cells induced by EBV antigens in ENKTL, we detected the HLA-A2 restricted EBV LMP1 (YLQ) and LMP2 (FLY, GLG and LLW) epitopic antigen-specific effector T cells and Foxp3+ Tregs in PBMCs from five HLA-A2 positive ENKTL patients and five HLA-A2 positive healthy controls by CD8 with tetramer staining together and CD8 and Foxp3 with tetramer staining together. We found out the different frequency of CD8+tetramer+ LMP1 (YLQ) or LMP2 (FLY, GLG and LLW) epitopic effector T cells in circulating blood from five ENKTL patients and five healthy controls as shown in Fig 3A and B; Interestingly, we also measured that the different frequency of CD8+Foxp3+tetramer+ Treg cells in circulating blood from the same five ENKTL patients but not in healthy controls as shown in Fig 3C and 3D.
The identification of EBV-LMP1 and LMP2 epitopic antigen-specific CD8+ T cells and CD8+Foxp3+ Tregs in ENKTL patients. A. FACS analysis of HLA-A2 restricted EBV epitopic antigen LLW, YLQ (LMP1) and GLG (LMP2) antigen specific CD8+ T cells; B. The histogram showed different percentage of CD8+tetramer+ T cells in PBMCs from HLA-A2 positive ENKTL patients (n=5) and healthy controls (n=5); C. FACS analysis of HLA-A2 restricted EBV epitopic antigen LLW, YLQ (LMP1) and GLG (LMP2) antigen-specific CD8+Foxp3+ Tregs; D. The histogram showed different percentage of CD8+Foxp3+tetramer+ Tregs in PBMCs from HLA-A2 positive ENKTL patients (n=5) and healthy controls (n=5).
The correlation of different tumor infiltrating lymphocyte subset and clinicopathological parameters was analyzed in ENKTL patients
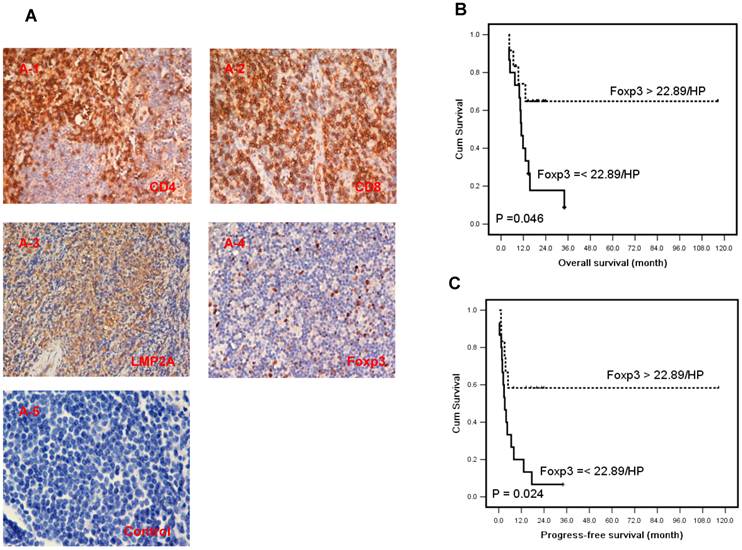
There are large amount of inflammatory cells around the tumor tissues in ENKTL for the presentation of neo-virus antigens on EBV infected tumor cells in ENKTL[2]. To characterize the lymphocyte subsets of TILs in ENKTL tumor tissues, we defined the different lymphocyte subset by specific antibodies: CD4 for helper T lymphocytes, CD8 for effector T cells and Foxp3 for Tregs (Fig 4A). The median number of CD4+, CD8+ and Foxp3+ cells in tumor tissues was 15.28±3.33 cells/HPF (range 0-62), 54.31±8.60 cells/HPF (range 0-212), and 22.89±4.69 cells/HPF (range 0-100) respectively (Supplementary Material: Table S2).
The expression of CD4, CD8 and Foxp3 in TILs and LMP2 in tumor cells of ENKTL patients and results of survival rate with Foxp3+ TILs in ENKTL patients. A, Immunohistochemical staining of CD4, CD8, Foxp3 in TILs and LMP2A was performed in tumor section of ENKTL patients (n=27). CD4 (A-1) and CD8 (A-2), LMP2A (A-3), Foxp3 (A-4) and isotypic antibody negative control (A-5). The number of Foxp3+ TILs are significantly associated with the overall survival (C, P=0.046) and progression-free survival (D, P=0.024) in ENKTL patients.
The correlations of the number of TILs subsets and clinicopathological parameters were summarized in Table 2. In brief, the density of CD4 was significantly negatively related to Korean Prognostic Index (KPI) and International Prognostic Index (IPI) (P<0.05), and marginally related to Ann Arbor stage (P=0.09). Interestingly, the density of Foxp3+ Tregs was negatively correlated with Ann Arbor stage and serum LDH (P<0.05). And it seemed relate to KPI, though it was not significant (P=0.06). These data indicated that the density of CD4+ T cells and Foxp3+ Tregs both was negatively associated to other risk prognostic factors in ENKTL. No significant relationship was observed between the density of CD8 and ENKTL clinical characteristics (P>0 .05). And no association was found between the density of different TILs subsets and the expression of LMP2A on tumor cells in ENKTL (P>0.05).
The association of immunohistochemical variants and clinicpathological characteristics of 27 cases of ENKTL with tissue sections
| Characteristics | CD4 | P value | CD8 | P value | FOXP3 | P value | |||
|---|---|---|---|---|---|---|---|---|---|
| ≤ 15.28 | > 15.28 | ≤ 54.31 | > 54.31 | ≤ 22.89 | > 22.89 | ||||
| Age | 1.00 | 0.50 | 0.19 | ||||||
| < 60 | 17 (63.0) | 8 (29.6) | 14 (51.9) | 11 (40.7) | 15 (55.6) | 10 (37.0) | |||
| ≥ 60 | 2 (7.4) | 0 | 2 (7.4) | 0 | 0 | 2 (7.4) | |||
| Gender | 1.00 | 1.00 | 0.63 | ||||||
| Male | 15 (55.6) | 7 (25.9) | 13 (48.1) | 9 (33.3) | 13 (48.1) | 9 (33.3) | |||
| Female | 4 (14.8) | 1 (3.7) | 3 (11.1) | 2 (7.4) | 2 (7.4) | 3 (11.1) | |||
| Stage | 0.09 | 0.43 | *0.005 | ||||||
| I-II | 9 (33.3) | 7 (25.9) | 8 (29.6) | 8 (29.6) | 5 (18.5) | 11 (40.7) | |||
| III-IV | 10 (37.0) | 1 (3.7) | 8 (29.6) | 3 (11.1) | 10 (37.0) | 1 (3.7) | |||
| B symptom | 1.00 | 0.24 | 0.44 | ||||||
| No | 11 (40.7) | 4 (14.8) | 7 (25.9) | 8 (29.6) | 7 (25.9) | 8 (29.6) | |||
| Yes | 8 (29.6) | 4 (14.8) | 9 (33.3) | 3 (11.1) | 8 (29.6) | 4 (14.8) | |||
| LDH | 0.21 | 0.70 | *0.05 | ||||||
| Normal | 8 (29.6) | 6 (22.2) | 9 (33.3) | 5 (18.5) | 5 (18.5) | 9 (33.3) | |||
| Elevated | 11 (40.7) | 2 (7.4) | 7 (25.9) | 6 (22.2) | 10 (37.0) | 3 (11.1) | |||
| PS | 0.68 | 0.69 | 0.22 | ||||||
| 0-1 | 12 (44.4) | 6 (22.2) | 10 (37.0) | 8 (29.6) | 8 (29.6) | 10 (37.0) | |||
| 2-5 | 7 (25.9) | 2 (7.4) | 6 (22.2) | 3 (11.1) | 7 (25.9) | 2 (7.4) | |||
| IPI score | *0.03 | 0.71 | 0.13 | ||||||
| 0-1 | 5 (18.5) | 6 (22.2) | 6 (22.2) | 5 (18.5) | 4 (14.8) | 7 (25.9) | |||
| 2-5 | 14 (51.9) | 2 (7.4) | 10 (37.0) | 6 (22.2) | 11 (40.7) | 5 (18.5) | |||
| KPI score | *0.03 | 0.45 | 0.06 | ||||||
| 0-1 | 4 (14.8) | 6 (22.2) | 7 (25.9) | 3 (11.1) | 3 (11.1) | 7 (25.9) | |||
| 2-4 | 15 (55.6) | 2 (7.4) | 9 (33.3) | 8 (29.6) | 12 (44.4) | 5 (18.5) | |||
| PIT score | 0.67 | 1.00 | 0.42 | ||||||
| 0-1 | 11 (40.7) | 6 (22.2) | 10 (37.0) | 7 (25.9) | 8 (29.6) | 9 (33.3) | |||
| 2-4 | 8 (29.6) | 2 (7.4) | 6 (22.2) | 4 (14.8) | 7 (25.9) | 3 (11.1) | |||
| LMP2A | 0.36 | 1.00 | 1.00 | ||||||
| Negative | 5 (20.8) | 4 (16.7) | 5 (20.8) | 4 (16.7) | 4 (16.70 | 5 (20.8) | |||
| Positive | 12 (50.0) | 3 (12.5) | 9 (37.5) | 6 (25.0) | 8 (33.3) | 7 (29.2) | |||
Note: UAT, upper aerodigestive tract; LDH, lactate dehydrogenase; IPI, International Prognostic Index; KPI, Korean Prognostic Index; PTI: Peripheral T-cell lymphoma Prognostic Index; PS: performance status.
Increased Foxp3+ TILs predicts a favorable progression-free survival in ENKTL patients
Twenty-seven newly diagnosed cases with tissue sections were performed for survival analysis. The estimated 3-year survival rate was 20.0% and median survival time was 13.1 months (4.1-116.5 months) in this study group. Seventeen cases died of cancer at the last follow-up and 19 cases relapsed (data listed in Supplementary Material: Table S2). A higher density of Foxp3 Tregs (>22.89 cells/HPF) correlated with better overall survival (OS) (HR, 0.32; 95% CI, 0.10-0.98; P=0.046, Table 3 and Fig 4B), and longer progression-free survival (PFS) by univariate analysis (HR, 0.31; 95% CI, 0.11-0.85; P=0.024, Table 3 and Fig 4C). Moreover the number of Foxp3 remained an independent predictor for PFS (HR, 0.12; 95% CI, 0.02-0.91; P=0.040, Table 3) after adjusted multiple variates analysis combined with other classical risk prognostic factors including Ann Arbor stage, B symptom, performance status, serum LDH level, IPI, KPI and PIT. But Foxp3 failed to predict OS of ENKTL patients (HR, 0.18; 95% CI, 0.05-1.75; P=0.997, Table 3) in adjusted analysis. The density of CD4 and CD8 didn't predict the OS and PFS in this study (P>0.05). Data was outlined in Table 3.
Prognostic value of TIL variants for OS and PFS in 27 cases of newly diagnosed ENKTL with tissue sections
| Parameter | Overall survival | Progress-free survival | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Univariate analysis | Adjusted analysis | Univariate analysis | Adjusted analysis | ||||||||||||
| HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value | HR | 95% CI | P-value | ||||
| CD4 | 0.67 | 0.22-2.06 | 0.480 | 1.00 | 0.15-6.63 | 0.626 | 0.44 | 0.14-1.32 | 0.142 | 0.54 | 0.10-3.01 | 0.484 | |||
| CD8 | 0.57 | 0.20-1.66 | 0.300 | 1.70 | 0.20-14.52 | 0.182 | 0.51 | 0.19-1.38 | 0.188 | 0.47 | 0.06-3.56 | 0.465 | |||
| FOXP3 | 0.32 | 0.10-0.98 | *0.046 | 0.18 | 0.05-1.75 | 0.997 | 0.31 | 0.11-0.85 | *0.024 | 0.12 | 0.02-0.91 | *0.040 | |||
Note: Adjusted analysis included Ann-Arbor stage, performance status, LDH, B symptom, IPI, KPI and PIT
Discussion
Increased CD4+CD25+ Tregs or CD4+Foxp3+ Tregs in peripheral blood has been reported in patients with cancers including gastric cancer, colorectal cancer, glioma and NPC[8, 21-23]. We not only investigated the significantly increased frequency of CD4+Foxp3+ Tregs in circulating lymphocyte, but also detected the dramatically increased frequency of CD8+Foxp3+ Tregs in circulating lymphocytes of patients with EBV positive ENKTL compared to healthy donors (P<0.001, Fig 1A and B). The expansion of CD8+ Tregs in peripheral blood has been reported in lung cancer and pleural mesothelioma[24], our recently work also showed the significantly increased frequency of CD8+ Tregs in peripheral blood of patients with EBV positive NPC (un-published data). The phenotypes and functional features of Foxp3+ Tregs subset were observed in this study. The Foxp3+ Tregs subset highly expressed CD45RO molecule and some levels of CCR7, GITR and CTLA4, and expressed IL-10highIFNγ+TGFβ+IL-2lowIL-17low cytokine secreting phenotype (Fig 2A), indicating that Foxp3+ Tregs subset in PBMCs from ENKTL patients contained different Treg cell subset such as natural Tregs (nTreg) and T regulatory Type 1 cells (Tr1 cells) which usually with a IL-10highTGFβ+IFNγ+IL-2- cytokine secreting phenotype. The suppressive function of Foxp3+ Tregs in ENKTL was determined in this study by inhibiting the proliferation of naïve T cells using the CD4+CD25high cells and CD8+CD25high sorting cells population in vitro (Fig 2B). However, here we couldn't find out the correlation of increased proportion of Foxp3+ Tregs subset in PBMCs and the tumor progression of ENKTL for limitation of sample number (only 10 ENKTL patients). The frequency of Tregs in peripheral blood increased with tumor stage has been reported in gastrointestinal malignancies[22].
EBV infection is associated with lymphoid and epithelial origin cancers including Burkitt lymphoma, Hodgkin lymphoma, nasopharyngeal carcinoma and ENKTL[25, 26]. The presentation of neo-virus antigens by virus-infected tumor cells could trigger an enlarged cellular immune response in cancer patients[27-31]. However, the tumor cells still escape the supervision of immune system, and the pathogenic induced Tregs has been identified in cancer in addition to the pathogenic specific cytotoxic T cell[32]. Here, we discovered for the first time that the existence of EBV LMP1 and LMP2 HLA-A2 restricted epitopic antigen-specific CD8+ Tregs in PBMCs from ENKTL patients (Fig 3C and 3D) in addition to the existence of EBV LMP1 and LMP2 HLA-A2 restricted epitopic antigen-specific cytotoxic T cells (Fig 3A and 3B); but we only found out the EBV antigen-specific effector T cells not the EBV antigen-specific CD8+Foxp3+ Tregs in healthy donors. The similar results were also found in NPC patients (data not shown) recently. These data suggest that the EBV can induce virus antigen-specific Tregs besides the EBV antigen-specific effector T cells in ENKTL patients. But we failed to find out the correlation of the density of TILs subsets including Foxp3+ Tregs and the expression of LMP2 antigen on tumor cell in this study.
Foxp3+ Tregs subset as suppressors of activated T effector cells usually have suppression to anti-tumor immunity in malignancies and correlated with poorer survival in many kinds of solid cancers[33]. However, recently in EBV associated malignancies including nasopharyngeal carcinoma, EBV positive classic Hodgkin lymphoma and follicular lymphoma, higher number of Foxp3+ TILs was associated with better survival[8, 13, 14]. ENKTL is an EBV positive malignancy with distinctive clinical symptoms and aggressive progression. Several classical prognostic factors including LDH level, B symptoms, IPI, PIT, and KPI could predict the prognosis of ENKTL patients. Recently one research group observed that the increasing number of absolute lymphocytes count (ALC) is a novel favorable prognostic index for ENKTL[15]. But it is still unclear the association of specific lymphocytes subset and clinical features and outcome, and their biological function in ENKTL. Here, our results showed that the density of CD4+ TILs was negatively associated with IPI and KPI score that suggested the CD4+ TILs decreased in high risk patients. Moreover the number of Foxp3+ TILs was negatively correlated with Ann Arbor stage and LDH level that implicated the frequency of Foxp3+ TILs was high in early stage and low tumor burden of ENKTL patients (Table 2). We cut off 27 newly diagnosed ENKTL patients into two subgroups by the median density of Foxp3+ TILs/HPF (22.89 cells/HPF), the ENKTL group with higher number of Foxp3+ TILs has longer OS and PFS, and moreover Foxp3 is an independent factor for PFS by adjusted multiple variates survival analysis (Table 3). Our findings were consistent with Kim's report[34]. Until now it is difficult to explain the clinical correlation and biological function of Foxp3+ Tregs in tumor site. The impact of Foxp3+ Tregs on tumor cells' behavior and on patients' outcome was conflicting in different type of malignancies. In EBV positive NPC patients we also found the Foxp3+TILs was an independent factor for prolonged OS and PFS[8], and moreover the Foxp3+ Tregs from tumor tissues could secret a large amount of IFNγ compared to that of PBMCs in the paired NPC patients (un-published data). Here, we also found that the Foxp3+Tregs subset in PBMCs of ENKTL patients contained a high proportion of IFNγ-secreting cells; the IFNγ is an inflammatory cytokine which could inhibit the growth of tumor cells, maybe Foxp3+ TILs take a protective function through secreting Th1 cytokine IFNγ. However, we couldn't detect this function of Tregs subset from TILs for the limitation of the small biopsy tumor tissues of ENKTL.
In summary, this study revealed for the first time the increased proportion of Foxp3+ Tregs in CD4+ and CD8+ cells population in peripheral blood of ENKTL patients compared with healthy individuals, and the Foxp3+ Tregs subset in PBMCs expressing IL-10highIFNγ+TGFβ+IL-2lowIL-17low cytokine secreting phenotype. Interestingly, we identified for the first time that the EBV could induce pathogenic antigen specific Tregs subset in ENKTL patients. At last, we illustrated that the increase of Foxp3 Trges in tumor tissues was an independent favorable prognostic index for PFS in ENKTL.
Supplementary Material
Table S1. Clinical characteristics of newly diagnosis extranodal NK/T cell lymphoma. Table S2. Immunohistochemical variants and survival in 27 newly diagnosis patients of extranodal NK/T cell lymphoma.
Acknowledgements
We thank all members of Dr. Zeng's laboratory for invaluable advice and discussions and Prof Mu-sheng Zeng for providing the LMP2 antibody.
Funding
This work was supported by grants from the National Natural Science Foundation of China (Grant No. 224 (30872981), author Jiang Li) and Guangdong Province Natural Science Foundation (Grant No.10151008901000156, author Jiang Li).
Conflict of Interests
No potential conflicts of interest were disclosed. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
1. Harris NL, Jaffe ES, Diebold J. et al. The World Health Organization classification of neoplastic diseases of the hematopoietic and lymphoid tissues. Report of the Clinical Advisory Committee meeting, Airlie House, Virginia, November, 1997. Ann Oncol. 1999;10:1419-1432
2. Swerdlow SH, Campo E, Harris NL. et al. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. France: International Agency for Research on Cancer. 2008
3. Hsieh PP, Tung CL, Chan AB. et al. EBV viral load in tumor tissue is an important prognostic indicator for nasal NK/T-cell lymphoma. Am J Clin Pathol. 2007;128:579-584
4. Mahmoud SM, Paish EC, Powe DG. et al. Tumor-infiltrating CD8+ lymphocytes predict clinical outcome in breast cancer. J Clin Oncol. 2011;29:1949-1955
5. Miotto D, Cascio NL, Stendardo M. et al. CD8+ T cells expressing IL-10 are associated with a favourable prognosis in lung cancer. Lung Cancer. 2010;69:355-360
6. Sorbye SW, Kilvaer T, Valkov A. et al. Prognostic impact of lymphocytes in soft tissue sarcomas. PLoS One. 2011;6:e14611
7. Suzuki H, Chikazawa N, Tasaka T. et al. Intratumoral CD8(+) T/FOXP3 (+) cell ratio is a predictive marker for survival in patients with colorectal cancer. Cancer Immunol Immunother. 2010;59:653-661
8. Zhang YL, Li J, Mo HY. et al. Different subsets of tumor infiltrating lymphocytes correlate with NPC progression in different ways. Mol Cancer. 2010;9:4
9. Kalams SA, Walker BD. The critical need for CD4 help in maintaining effective cytotoxic T lymphocyte responses. J Exp Med. 1998;188:2199-2204
10. Wolf AM, Rumpold H, Wolf D. et al. Role of forkhead box protein 3 expression in invasive breast cancer. J Clin Oncol. 2007;25:4499-4500
11. Curiel TJ, Coukos G, Zou L. et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10:942-949
12. Salama P, Phillips M, Grieu F. et al. Tumor-infiltrating FOXP3+ T regulatory cells show strong prognostic significance in colorectal cancer. J Clin Oncol. 2009;27:186-192
13. Alvaro T, Lejeune M, Salvado MT. et al. Outcome in Hodgkin's lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells. Clin Cancer Res. 2005;11:1467-1473
14. Tzankov A, Meier C, Hirschmann P. et al. Correlation of high numbers of intratumoral FOXP3+ regulatory T cells with improved survival in germinal center-like diffuse large B-cell lymphoma, follicular lymphoma and classical Hodgkin's lymphoma. Haematologica. 2008;93:193-200
15. Huang JJ, Jiang WQ, Lin TY. et al. Absolute lymphocyte count is a novel prognostic indicator in extranodal natural killer/T-cell lymphoma, nasal type. Ann Oncol. 2011;22:149-155
16. Wang HY, Peng G, Guo Z. et al. Recognition of a new ARTC1 peptide ligand uniquely expressed in tumor cells by antigen-specific CD4+ regulatory T cells. J Immunol. 2005;174:2661-2670
17. Kong QL, Hu LJ, Cao JY. et al. Epstein-Barr virus-encoded LMP2A induces an epithelial-mesenchymal transition and increases the number of side population stem-like cancer cells in nasopharyngeal carcinoma. PLoS Pathog. 2010;6:e1000940
18. Trzonkowski P, Szmit E, Mysliwska J, Mysliwski A. CD4+CD25+ T regulatory cells inhibit cytotoxic activity of CTL and NK cells in humans-impact of immunosenescence. Clin Immunol. 2006;119:307-316
19. Li J, Qian CN, Zeng YX. Regulatory T cells and EBV associated malignancies. Int Immunopharmacol. 2009;9:590-592
20. Jones K, Gandhi MK. Epstein-Barr virus DNA as a biomarker for Epstein-Barr virus-positive lymphomas: are we there yet? Leuk Lymphoma. 2009;50:684-686
21. Wolf AM, Wolf D, Steurer M. et al. Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res. 2003;9:606-612
22. Shen LS, Wang J, Shen DF. et al. CD4(+)CD25(+)CD127(low/-) regulatory T cells express Foxp3 and suppress effector T cell proliferation and contribute to gastric cancers progression. Clin Immunol. 2009;131:109-118
23. Heimberger AB, Abou-Ghazal M, Reina-Ortiz C. et al. Incidence and prognostic impact of FoxP3+ regulatory T cells in human gliomas. Clin Cancer Res. 2008;14:5166-5172
24. Meloni F, Morosini M, Solari N. et al. Foxp3 expressing CD4+ CD25+ and CD8+CD28- T regulatory cells in the peripheral blood of patients with lung cancer and pleural mesothelioma. Hum Immunol. 2006;67:1-12
25. Hippocrate A, Oussaief L, Joab I. Possible role of EBV in breast cancer and other unusually EBV-associated cancers. Cancer Lett. 2011;305:144-149
26. Peh SC, Kim LH, Poppema S. Frequent presence of subtype A virus in Epstein-Barr virus-associated malignancies. Pathology. 2002;34:446-450
27. Rickinson AB. Immune intervention against virus-associated human cancers. Ann Oncol. 1995;6(Suppl 1):69-71
28. Cevenini R, Donati M, Moroni A. et al. Specific Epstein-Barr virus serological response in patients with nasopharyngeal carcinoma detected by immunoblotting. Eur J Epidemiol. 1988;4:301-305
29. Rickinson AB, Gregory CD, Young LS. Viruses and cancer risks: outgrowth of Epstein-Barr virus-positive Burkitt's lymphoma in the immune host. Med Oncol Tumor Pharmacother. 1987;4:177-186
30. Houston KJ, Blasecki JW. Assessment of host immune status during progressive growth and after excision of DNA virus (simian virus 40) tumors in hamsters: comparison of tumor-specific and tumor-unrelated parameters of immune responsiveness. J Natl Cancer Inst. 1979;63:665-673
31. Bowen JM, Dmochowski L, Miller MF. et al. Implications of humoral antibody in mice and humans to breast tumor and mouse mammary tumor virus-associated antigens. Cancer Res. 1976;36:759-764
32. Wang RF. Immune suppression by tumor-specific CD4+ regulatory T-cells in cancer. Semin Cancer Biol. 2006;16:73-79
33. Curiel TJ. Regulatory T cells and treatment of cancer. Curr Opin Immunol. 2008;20:241-246
34. Kim WY, Jeon YK, Kim TM. et al. Increased quantity of tumor-infiltrating FOXP3-positive regulatory T cells is an independent predictor for improved clinical outcome in extranodal NK/T-cell lymphoma. Ann Oncol. 2009;20:1688-1696
Author contact
Corresponding author: Prof. J. Li. Department of Biotherapy Center & Department of Experimental Research, Sun Yat-Sen University Cancer Center, 651 Dongfeng Road East, Guangzhou, 510060. Tel: +86-20-87343174; Fax: +86-20-87343392. E-mail: lijiang2sysu.edu.cn & Prof. Y.X. Zeng, Department of Experimental Research, Sun Yat-Sen University Cancer Center, 651 Dongfeng Road East, Guangzhou, 510060. Tel: +86-20-87343333; Fax: +86-20-87343392; E-mail: zengyxorg.cn

 Global reach, higher impact
Global reach, higher impact