Impact Factor ISSN: 1449-2288
Int J Biol Sci 2014; 10(2):136-141. doi:10.7150/ijbs.7515 This issue Cite
Research Paper
Micrometastasis Expressing Insulin Arise in Lung and Spleen at Advanced Stage of Rip1-Tag2 Transgenic Mice
1. Vascular Biology Research Institute, Guangdong Pharmaceutical University, Guangzhou 510006, China;
2. School of Basic Courses, Guangdong Pharmaceutical University, Guangzhou, 510006, Guangdong, China;
Received 2013-8-28; Accepted 2013-12-17; Published 2014-1-10
Abstract
Rip1-Tag2 mice is one overt pancreatic β-cell tumor model, which is widely used for studying pancreas tumor angiogenesis and tumor development. However, tumor metastasis in Rip1-Tag2 mice had rarely been reported, in this present study, we find some micrometastasis in lung and spleen of the Rip1-Tag2 mice at advanced stage, which is important for uncovering metastasis cell characteristics and exploring how to survive in cancer microenvironment. To study the micrometastasis of Rip1-Tag2 mice in advanced pancreatic cancer, we first observed the pathology process of β cell tumor in Rip1-Tag2 mice through HE staining, then we performed immunohistochemistry with insulin antibody, T-antigen antibodies and C-petide antibody on lung and spleen tissues sections from advanced stage, comparing with background wild-type C57BL/6 mice sections. The results indicated that micrometastasis expressing insulin was found in the Rip1-Tag2 mice lung, and spleen. Further evidences demonstrate pathology structure of lung and spleen are damaged. Interestingly and importantly, the expression of T antigen and insulin antibodies are all decreased in advanced stage of primary β cell tumor, which suggest that the at least partly micrometastasis is derived from the early stage or from advanced stage of β cell tumor then return to undifferentiated state like cancer stem cell. The findings contributed to the study of cancer metastasis and cancer stem cell.
Keywords: micrometastasis, insulin, Rip1-Tag2 transgenic mice
INTRODUCTION
SV40 T antigen is a well-known oncogene, and tumor cell would emerged in different kinds mice model of SV40 T antigen transgenic mice [1-4]. SV40 T antigen (T antigen) under the control of the rat insulin gene promoter region is used to produce Rip1-Tag2 mice, which is overt pancreatic β-cell tumor model [5]. This mice model is used to study tumor development and tumor angiogenesis. Even pancreatic tumor pathology of those model mice was standardized nomenclature with definitions [6, 7]. Generally the Rip1-Tag2 mice is not regard as metastasis animal model, however, we unexpectedly observed the micrometastasis in Rip1-Tag2 mice in tumor advanced stage that is not reported before. It is of great significance for studying the process of metastasis of cancer cell or the characteristics of metastasis cell.
As well known, in Rip1-Tag2 transgenic mice, T antigen and insulin are linked together in gene location and expression simultaneously cue to under the same promoter. T antigen inactivate of the Rb protein and P53 [8-14], targeting multiple cell function including regulate cell proliferation, death, immune responses and even the tumor microenvironment involved in that process [15-17]. With tumor development, T-antigen expression level is down-regulated as previous reports had said [18]. Although the tumorigenesis process is triggered by Tag, it remains unknown whether continuous T antigen expression is necessary for growth or metastasis cell in Rip1-Tag2 mice.
In this study, our results demonstrated that micrometastasis express insulin and T antigen, but their expression in primary tumor was down-regulated in advanced stage in Rip1-Tag2 mice. The contribution of this study is that micrometastasis character in Rip1-tag2 mice, expressing T antigen and insulin, was established. Interestingly and importantly, the findings suggested that micrometastasis return back to undifferentiated state like cancer stem cell expressing insulin again. In addition, insulin-producing cells is important for diabetic patients [19, 20].
MATERIALS AND METHODS
Transgenic mice
The Rip1-Tag2 (C57BL/6 mice as background mice) mice were purchased from The Jackson Laboratory, and C57/BL6 mice were obtained from Animal Center of Guangdong province (Guangzhou, China). The advanced stage of mice refers to about 14 weeks of Rip-Tag2 mice. The studies of transgenic mice and C57BL/6 mice were reviewed and approved by the Committees for Ethical Review of Research involving Animal Subjects at Guangdong Pharmaceutical University, China.
HE staining
Mice pancreases and other tissues were fixed overnight in 4% formalin. Fixed tissues were rinsed in 1×PBS, subsequently dehydrated in 35, 50, and 75% ethanol, then embedded in paraffin. 10 μm sections were deparrafinized in xylene and rehydrated in 100, 95, 70, and 50% ethanol then 1×PBS. H&E staining of sections was carried out as previously described [21].
Immunohistochemistry
Immunohistochemistry (IHC) was performed as described on the web of Cell Signaling Technology, Inc (MA, USA), Primary antibody include T-antigen (1:100, Abcam), C-petide antibody (1:100, Abcam), and Insulin antibody (1:200, Fitzgerald), and second antibody system was purchased from Genetech (Shanghai, China). The slides for T-antigen and C-petide antibodies were treated with EDTA solution (pH=9.0) for antigen retrieval, while the slides corresponding insulin antibody was no treated for antigen retrieval.
Statistical Analysis
SPSS 16.0 (SPSS Inc., Chicago, USA) was adopted to analyze our data. The IHC results of C57BL/6 and Rip1-Tag2 mice were compared using Pearson's Chi-square test. A P value less than 0.05 was considered statistically significant.
RESULTS
Identification of Rip1-Tag2 transgenic mice
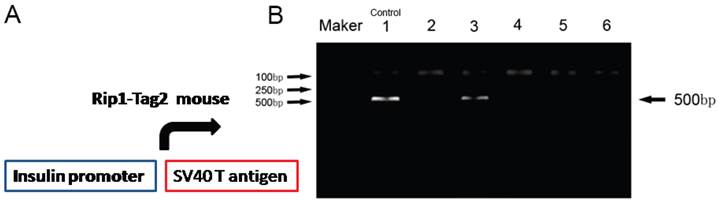
To obtain the Rip-Tag2 transgenic mice, we identified the transgenic mice by detecting T antigen with PCR, which located downstream of mouse insulin promoter (Fig.1A). Primer sequences used are shown: the forwards primer: 5'-GGACAAACCACAACTAGAATGCAG-3' and reverse primer: 5'-CAGAGCAGAATTGTGGAGTGG-3'. PCR conditions were showed as: 94℃ 2min; 94℃ 30sec, 56℃ 30s, 72℃ 30s (35 cycles); 72℃10min. PCR product size is about 500bp. Rip1-Tag2 transgenic mice were shown as lane 1, 3 and lane 6 (shown in Fig.1B).
Identification of the gene-type of Rip1-Tag2 mice. T-antigen gene was identified by using PCR assay. The lanes 2, 4, 5 indicated Tag2 were negative (-), but the lanes 3, and 6 show Tag2 were positive (+). Lane 1 was regard as positive control. M was indicated the Marker DL2000.
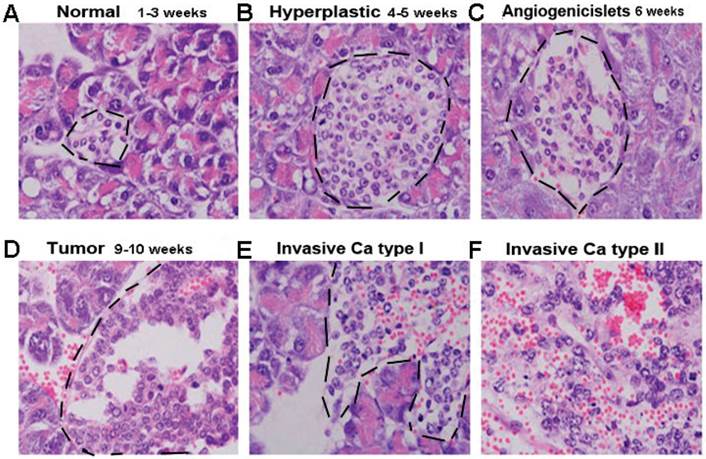
Pathological Progress of islet β cell tumor in Rip1-Tag2
β cells of Rip1-Tag2 transgenic mice secrete insulin, and SV40 T antigen (T antigen) under insulin promoter express simultaneously. β cells developed a multi-stage pancreatic tumor (As shown in Fig.2), about 1-3 weeks, it was known as “Normal stage”, in which Islet cell had no difference with C57BL/6 mice (Fig 1A); At 4-5weeks of age, hyperplastic islet/dysplastic began to appear, most of cell nuclear/cytoplasm ratio increases, but the vascular system remained quiescent at this stage (Fig 2B). From about 6 weeks, it began to appear angiogenic Islets and different sizes of angiogenic Islets were formed, We could see the nuclear/plasma ratio remained increasing, and the vascular system was activated as well as branch multiplied (Fig 2C). In 9-10 weeks tumor appeared, nuclear/cytoplasm ratio and cell density reached the highest, and the vascular network was densely developed (Fig 2D), In the advanced stage about 14 weeks (Fig 2E and 2F), about half of the tumors infiltrated into the surrounding acinar tissue.
The process of Pancreatic βcell tumor in Rip1-Tag2 transgenic mice. A. Normal stage, the Islet cell is normal and same as C57BL/6 mice(Fig 2A); B. Hyperplastic islet stage, dysplastic begin to appear, most of cell nuclear/cytoplasm ratio increases(Fig 2B), C. Angiogenic islets stage, angiogenic Islets began to appear(Fig 2C), D. Tumor stage, the vascular network is highly developed, solid tumors formed(Fig 3D). E. Invasion stage. Tumor infiltrate into the surrounding acinar tissue (Fig 2E and 2F).
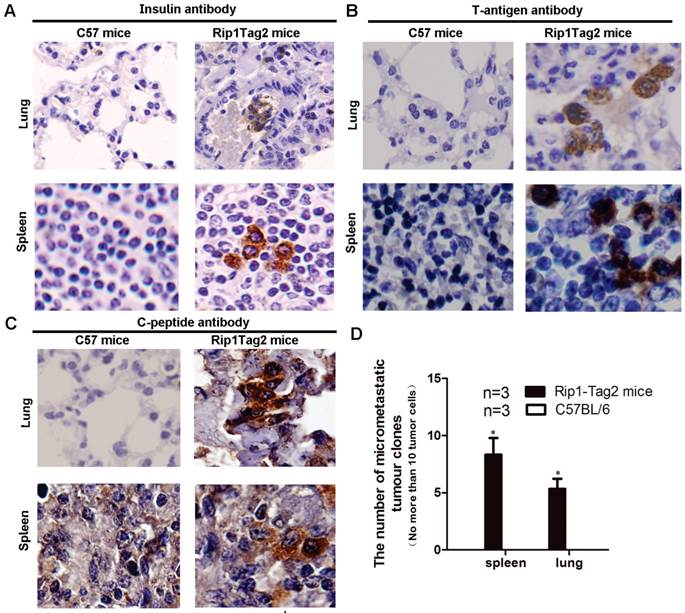
Micrometastasis arise in lung and spleen expressing insulin and T antigen
Metastasis was few reported in Rip1-Tag2 mice. To validate if the micrometastasis had existed in multiple organ systems in advanced stage referring to 14 weeks of β cell tumor, we performed IHC to detect lung and spleen tissue with insulin, C-peptide, and T-antigen antibody. Insulin was known as a peptide hormone produced only by β cells, and C-peptide is a peptide composed of 31 amino acids which is released from the pancreatic beta-cells. T-antigen antibody, which gene was located link together with insulin gene in this transgenic mice as shown in Fig.1. By comparing the two results of Rip1-Tag2 mice and background wild type C57BL/6 mice, we found that the insulin expression positive cells were arising obviously in the lung and spleen, as shown in Fig. 3A. Further C-peptide and T-antigen expressing cell also were detected in lung and spleen of Rip1-Tag2 mice (Fig 3B and 3C), the results showed C-peptide and T-antigen all present in micrometastasis of lung and spleen. In addition, most micrometastassis size is less than 10 tumor cells. We further counted the number of micrometastasis in lung and spleen of Rip1-Tag2 mice in five consecutive sides not repeating count the same micrometastasis, not finding the micrometastasis in C57BL/6 mice. The number of micrometastsis were significantly statistical differences in lung and spleen comparing with the corresponding to mice, as shown in Fig. 3D, *P<0.0001.
Micrometastasis appears in lung and spleen in Rip1-Tag2 mice. A. Micrometastasis size was defined as no more than 10 cells per section (10μm). Micrometastasis tumor cell expressing insulin rise in lung and spleen as shown in Fig.3A (magnification: 600×). B. Micrometastasis was further confirmed by IHC assay, C-peptide antibody was used to verify above IHC results, T-antigen antibody was also used in IHC to show that micrometastsis expressed T-antigen as show in Fig 3B (magnification: 1000×). C. Likely, C-peptide was also expression in micrometastasis shown as Fig 3C (magnification: 1000×). The results suggested that micrometastasis was derived from insulinoma. D. Micrometastasis are not find in lung and spleen in C57BL/6 mice but was observed in Rip1-Tag2 mice (P<0.0001, n=3, the number of section is 10μm/slide), shown as Fig.3D. Note: micrometastassis refer to the number of tumor cell was less than 10, and advanced stage was 14 weeks.
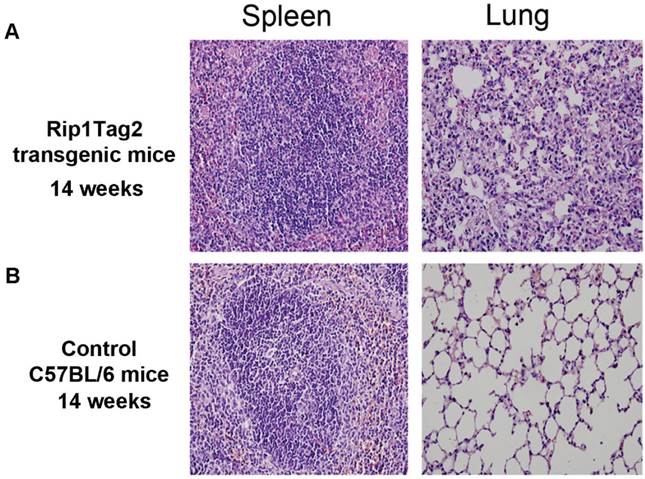
Pathological structure of lung and spleen in advanced stage were damaged
To clarity if micrometastasis of β cell tumor had damaged and affected architecture of lung and spleen in Rip1-Tag2 mice, HE staining was used to detect the pathological structure of lung, and spleen. As shown in Fig.4A, RIP1-Tag2 mice's lung and spleen were damaged obviously, Reactive hyperplasia of lymphoid tissue were seen in spleen, and thickened and bleeding of the alveolar septa, congestion of capillary, hemorrhagic foci were visible in lung, all the features as shown in Fig 4A. But the pathological structure of lung and spleen of C57BL/6 mice was no altering (Fig. 4B).
Pathological structures altering in lung and spleen in advantage stage. A. Reactive hyperplasia of lymphoid tissue in spleen in Rip-Tag2 mice and C57BL/6 mice (background) as control. B. Thickening and bleeding of the alveolar septa, congestion of capillary, hemorrhagic foci (pink dot) was observed in lung of Rip1-Tag2 mice. Microscope magnification: 400×.
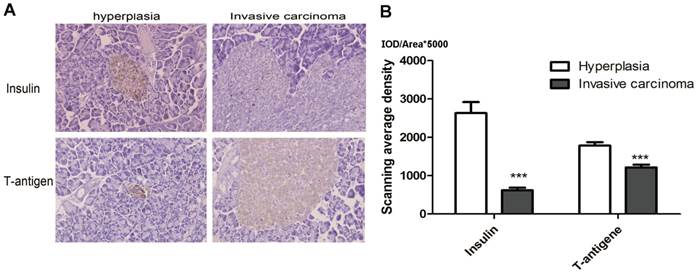
Insulin and T antigen highly express in hyperplasia islet but down-regulation in invasive carcinoma
Early reported indicated the protein expression of T antigen would decrease with the development of tumor, in our study, to further confirm that the insulin and T antigen expression are decrease with the development of primary tumor, we detected the Insulin and T antigen expression in hyperplastic islet (4 weeks) and invasive carcinoma (about 14 week) by performing IHC. The results demonstrated that Insulin expression was obviously down-regulation in invasive carcinoma than in hyperplastic islet, as shown in Fig. 5, P<0.01. The results indicated that tumor cells highly expressed insulin in hyperplasia, while the level of insulin and T antigen decrease in invasive carcinoma.
Insulin and T antigen low-expression invasive carcinoma. A. Representative image of Insulin and T-antigen IHC in Hyperplasia (left, 4 weeks) and invasive carcinoma (right, 14 weeks) (magnification: ×1000). B. The results showed insulin and T-antigen down-regulation in hyperplasia comparison with invasive carcinoma (***P<0.01). The mean gray level IOD/Area was used to analysis in 10 fields, the color stained brown was objective to analyze (HRP-DAB).
Above data clue us that the micrometastasis cell maybe derived from hyperplasia at early tumor stage remain quiescent state. Alternatively, micrometastasis return undifferentiated cell state from invasive carcinoma.
DISCUSSION
Rip1-Tag2 mice have been used as pancreatic islet β cell tumor model for studying angiogenesis, cancer development and autoimmunity etc. In many reports, with the growth of transgenic mice, islet β cell tumor begin to happen in transgenic mice [4, 22] and the pathologic structure of pancreas will alter with mice age. There is a trend toward loss islet morphological structure in advanced stage of tumor. And we also confirmed that the pancreatic islets undergo from incipient neoplasia to advanced stage of tumors. T antigen was regarded as manipulating under insulin promoter which is essential for both the initiation of the tumor transformed state [22-25].
In this transgenic mice model, Insulin is linked together T-antigen in gene location under control insulin promoter, and we confirmed that T antigen and insulin both are down-regulated in invasive carcinoma stage by IHC analysis, which is consistent with previous reports [26, 27]. Generally speaking, the micrometastasis may happen in multiple tissue in advanced stage of tumor patients, and we also observed a few of micrometastasis in spleen, and lung in transgenic mice, this results maybe is analogous to thing that we see in tumor patient. Lung and spleen structure damage demonstrates that it may produce subtle influence on organ structure and their function with the developing of tumor and micrometastasis. Micrometastasis tumor cell has potent effects on lung and spleen in advanced stage (14 weeks), which may result in chronically cancer microenvironment and structure change.
Interestingly, the other team study informed us that these metastasis cells was supposed as tumor-initiating cells or cancer stem cell can originate from proliferating undifferentiated precursor cells. In this study, micrometastasis expressed insulin but the tumor cell in invasive carcinoma barely express insulin, which potently revealed those micrometasis cancer cell remains precursor cell feature, suggesting that micrometastasis remain undifferentiated cell state in non cancer cells population and microenvironment.
In summary, the results demonstrated the micrometastasis cell may be derived from pancreatic islet expressing insulin at early stage, because in invasive carcinoma stage its expression is down-regulated, lastly affecting on the organs tissue structure. Alternatively, micrometastasis left at invasive carcinoma stage, then go back an undifferentiated cell state to secrete insulin. micrometastasis have cancer stem cell feature similar to the original pancreatic β cell. Of course, further study is still needed to explore these metastatic sporadic tumor cell.
Acknowledgements
This work is supported by the Medical Scientific Research Foundation of Guangdong Province (No. A2013312), the National Natural Science Foundation of China (No.30971606, No.81172338, No.21005022 and No.81000863).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Palmiter RD, Chen HY, Messing A, Brinster RL. SV40 enhancer and large-T antigen are instrumental in development of choroid plexus tumours in transgenic mice. Nature. 1985;316:457-60
2. Park WY, Kim JI, Shim EH, Lee WH, Kim SH, Seo JW. et al. Development of thymic carcinoma in transgenic mice expressing SV40 T antigen. Cancer letters. 1996;107:293-300
3. Butel JS, Sepulveda AR, Finegold MJ, Woo SL. SV40 large T antigen directed by regulatory elements of the human alpha-1-antitrypsin gene. A transgenic mouse system that exhibits stages in liver carcinogenesis. Intervirology. 1990;31:85-100
4. Brinster RL, Chen HY, Messing A, van Dyke T, Levine AJ, Palmiter RD. Transgenic mice harboring SV40 T-antigen genes develop characteristic brain tumors. Cell. 1984;37:367-79
5. Hanahan D. Heritable formation of pancreatic beta-cell tumours in transgenic mice expressing recombinant insulin/simian virus 40 oncogenes. Nature. 1985;315:115-22
6. Hruban RH, Adsay NV, Albores-Saavedra J, Anver MR, Biankin AV, Boivin GP. et al. Pathology of genetically engineered mouse models of pancreatic exocrine cancer: consensus report and recommendations. Cancer research. 2006;66:95-106
7. Herreros-Villanueva M, Hijona E, Cosme A, Bujanda L. Mouse models of pancreatic cancer. World J Gastroenterol. 2012;18:1286-94
8. Li Q, Zhang Y, Sheng Y, Huo R, Sun B, Teng X. et al. Large T-antigen up-regulates Kv4.3 K(+) channels through Sp1, and Kv4.3 K(+) channels contribute to cell apoptosis and necrosis through activation of calcium/calmodulin-dependent protein kinase II. The Biochemical journal. 2012;441:859-67
9. Noutsopoulos D, Vartholomatos G, Kolaitis N, Tzavaras T. SV40 large T antigen up-regulates the retrotransposition frequency of viral-like 30 elements. Journal of molecular biology. 2006;361:450-61
10. Iwasaki T, Kodama H, Matsushita M, Kuroda N, Yamasaki Y, Murakami I. et al. Merkel cell polyomavirus infection in both components of a combined Merkel cell carcinoma and basal cell carcinoma with ductal differentiation; each component had a similar but different novel Merkel cell polyomavirus large T antigen truncating mutation. Human pathology. 2013;44:442-47
11. Ahuja D, Saenz-Robles MT, Pipas JM. SV40 large T antigen targets multiple cellular pathways to elicit cellular transformation. Oncogene. 2005;24:7729-45
12. An P, Saenz Robles MT, Pipas JM. Large T antigens of polyomaviruses: amazing molecular machines. Annual review of microbiology. 2012;66:213-36
13. Saenz Robles MT, Pipas JM. T antigen transgenic mouse models. Seminars in cancer biology. 2009;19:229-35
14. Logsdon CD, Simeone DM, Binkley C, Arumugam T, Greenson JK, Giordano TJ. et al. Molecular profiling of pancreatic adenocarcinoma and chronic pancreatitis identifies multiple genes differentially regulated in pancreatic cancer. Cancer research. 2003;63:2649-57
15. Reisfeld RA. The Tumor Microenvironment: A Target for Combination therapy of Breast Cancer. Critical reviews in oncogenesis. 2013;18:115-33
16. Chang DZ, Ma Y, Ji B, Wang H, Deng D, Liu Y. et al. Mast cells in tumor microenvironment promotes the in vivo growth of pancreatic ductal adenocarcinoma. Clin Cancer Res. 2011;17:7015-23
17. Evans A, Costello E. The role of inflammatory cells in fostering pancreatic cancer cell growth and invasion. Frontiers in physiology. 2012;3:270
18. Hager JH, Hodgson JG, Fridlyand J, Hariono S, Gray JW, Hanahan D. Oncogene expression and genetic background influence the frequency of DNA copy number abnormalities in mouse pancreatic islet cell carcinomas. Cancer research. 2004;64:2406-10
19. Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627-32
20. Guo QS, Zhu MY, Wang L, Fan XJ, Lu YH, Wang ZW. et al. Combined transfection of the three transcriptional factors, PDX-1, NeuroD1, and MafA, causes differentiation of bone marrow mesenchymal stem cells into insulin-producing cells. Experimental diabetes research. 2012;2012:672013. doi:10.1155/2012/672013
21. Li J, Yang H, Chen L, Li Y, Zhu Y, Dai Y. et al. Establishment and characterization of human non-small cell lung cancer cell lines. Molecular medicine reports. 2012;5:114-7
22. Kimura G, Itagaki A. Initiation and maintenance of cell transformation by simian virus 40: a viral genetic property. Proceedings of the National Academy of Sciences of the United States of America. 1975;72:673-7
23. Tegtmeyer P. Function of simian virus 40 gene A in transforming infection. Journal of virology. 1975;15:613-8
24. Brockman WW. Transformation of BALB/c-3T3 cells by tsA mutants of simian virus 40: temperature sensitivity of the transformed phenotype and retransofrmation by wild-type virus. Journal of virology. 1978;25:860-70
25. Lane DP, Crawford LV. T antigen is bound to a host protein in SV40-transformed cells. Nature. 1979;278:261-3
26. Bardeesy N, DePinho RA. Pancreatic cancer biology and genetics. Nature reviews. 2002;2:897-909
27. Faca VM, Song KS, Wang H, Zhang Q, Krasnoselsky AL, Newcomb LF. et al. A mouse to human search for plasma proteome changes associated with pancreatic tumor development. PLoS medicine. 2008;5:e123
Author contact
Corresponding author: Li-jing Wang, Vascular Biology Research Institute, Guangdong Pharmaceutical University, Guangzhou 510006, China. Tel: 86-20-39352126; E-mail: wanglijingedu.cn

 Global reach, higher impact
Global reach, higher impact