Impact Factor ISSN: 1449-2288
Int J Biol Sci 2015; 11(3):246-255. doi:10.7150/ijbs.11138 This issue Cite
Research Paper
MicroRNA-1 Participates in Nitric Oxide-Induced Apoptotic Insults to MC3T3-E1 Cells by Targeting Heat-Shock Protein-70
1. Department of Orthopedic Surgery, Shin Kong Wu Ho-Su Memorial Hospital, Taipei, Taiwan
2. Cell Biology and Molecular Image Research Center, Taipei Medical University-Wan Fang Hospital, Taipei, Taiwan
3. Department of Internal Medicine, Taipei Medical University-Wan Fang Hospital, Taipei, Taiwan
4. Graduate Institute of Medical Sciences, College of Medicine, Taipei Medical University, Taipei, Taiwan.
5. Anesthetics and Toxicology Research Center, Taipei Medical University Hospital, Taipei, Taiwan.
Abstract
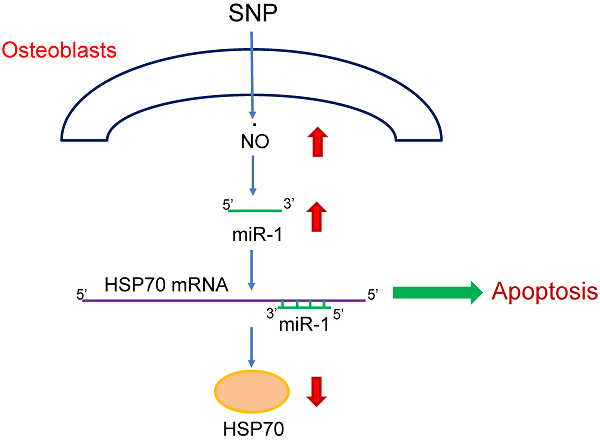
Our previous studies showed that nitric oxide (NO) could induce osteoblast apoptosis. MicroRNA-1 (miR-1), a skeletal- and cardiac muscle-specific small non-coding RNA, contributes to the regulation of multiple cell activities. In this study, we evaluated the roles of miR-1 in NO-induced insults to osteoblasts and the possible mechanisms. Exposure of mouse MC3T3-E1 cells to sodium nitroprusside (SNP) increased amounts of cellular NO and intracellular reactive oxygen species. Sequentially, SNP decreased cell survival but induced caspase-3 activation, DNA fragmentation, and cell apoptosis. In parallel, treatment with SNP induced miR-1 expression in a time-dependent manner. Application of miR-1 antisense inhibitors to osteoblasts caused significant inhibition of SNP-induced miR-1 expression. Knocking down miR-1 concurrently attenuated SNP-induced alterations in cell morphology and survival. Consecutively, SNP time-dependently inhibited heat-shock protein (HSP)-70 messenger (m)RNA and protein expressions. A bioinformatic search predicted the existence of miR-1-specific binding elements in the 3'-untranslational region of HSP-70 mRNA. Downregulation of miR-1 expression simultaneously lessened SNP-induced inhibition of HSP-70 mRNA and protein expressions. Consequently, SNP-induced modifications in the mitochondrial membrane potential, caspase-3 activation, DNA fragmentation, and apoptotic insults were significantly alleviated by miR-1 antisense inhibitors. Therefore, this study showed that miR-1 participates in NO-induced apoptotic insults through targeting HSP-70 gene expression.
Keywords: Nitric oxide, MicroRNA-1, Apoptosis, HSP-70, Mitochondria

 Global reach, higher impact
Global reach, higher impact