10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2016; 12(4):397-408. doi:10.7150/ijbs.13475 This issue Cite
Research Paper
MiR-487a Promotes TGF-β1-induced EMT, the Migration and Invasion of Breast Cancer Cells by Directly Targeting MAGI2
1. Department of Pharmacology, School of Pharmacy, China Medical University, Shenyang, Liaoning Province, China;
2. Department of Surgical Oncology, The First Affiliated Hospital of China Medical University, Shenyang, Liaoning Province, China.
*These authors contributed equally to this work.
Abstract
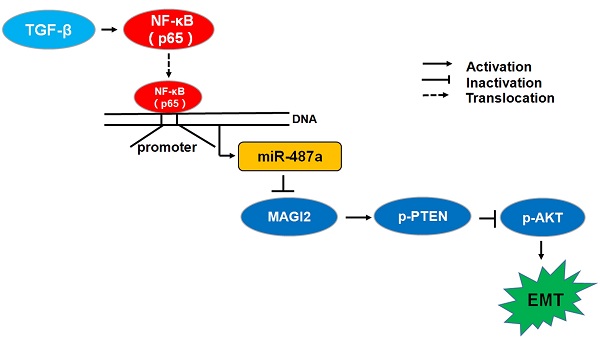
Tumor metastasis is a complex and multistep process and its exact molecular mechanisms remain unclear. We attempted to find novel microRNAs (miRNAs) contributing to the migration and invasion of breast cancer cells. In this study, we found that the expression of miR-487a was higher in MDA-MB-231breast cancer cells with high metastasis ability than MCF-7 breast cancer cells with low metastasis ability and the treatment with transforming growth factor β1 (TGF-β1) significantly increased the expression of miR-487a in MCF-7 and MDA-MB-231 breast cancer cells. Subsequently, we found that the transfection of miR-487a inhibitor significantly decreased the expression of vimentin, a mesenchymal marker, while increased the expression of E-cadherin, an epithelial marker, in both MCF-7 cells and MDA-MB-231 cells. Also, the inactivation of miR-487a inhibited the migration and invasion of breast cancer cells. Furthermore, our findings demonstrated that miR-487a directly targeted the MAGI2 involved in the stability of PTEN. The down-regulation of miR-487a increased the expression of p-PTEN and PTEN, and reduced the expression of p-AKT in both cell lines. In addition, the results showed that NF-kappaB (p65) significantly increased the miR-487a promoter activity and expression, and TGF-β1 induced the increased miR-487a promoter activity via p65 in MCF-7 cells and MDA-MB-231 cells. Moreover, we further confirmed the expression of miR-487a was positively correlated with the lymph nodes metastasis and negatively correlated with the expression of MAGI2 in human breast cancer tissues. Overall, our results suggested that miR-487a could promote the TGF-β1-induced EMT, the migration and invasion of breast cancer cells by directly targeting MAGI2.
Keywords: miR-487a, MAGI2, EMT, TGF-β1, p65, Breast cancer.

 Global reach, higher impact
Global reach, higher impact