ISSN: 1449-2288
Int J Biol Sci 2016; 12(5):505-517. doi:10.7150/ijbs.14212 This issue Cite
Research Paper
Transcriptional Reactivation of OTX2, RX1 and SIX3 during Reprogramming Contributes to the Generation of RPE Cells from Human iPSCs
1. Stem Cell Research Center, Department of Cell Biology, School of Basic Medical Sciences, Peking University, Haidian District, Beijing, 100191, China
2. Department of Histology and Embryology, Institute of Chinese Medicine, Hunan University of Chinese Medicine, Science Garden District of Hanpu, Changsha, Hunan, 410208, China
3. Peking University Eye Center, Peking University Third Hospital, Beijing, 100191, China
4. Lund Stem Cell Center, University Hospital, Lund University, Lund, 22242, Sweden
5. Beijing Cellonis Biotechnologies Co.Ltd, Zhongguancun Bio-Medicine Park, Beijing, 100191, China
6. Department of Biology, College of Chemistry and Life Sciences, Zhejiang Normal University, Jinhua, Zhejiang, 321004, China.
* These authors contributed equally to this work.
Abstract
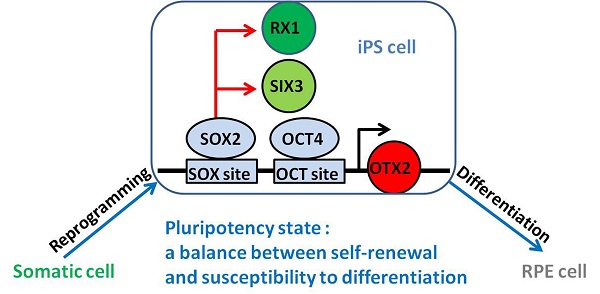
Directed differentiation of human induced pluripotent stem cells (iPSCs) into retinal pigmented epithelium (RPE) holds great promise in cell replacement therapy for patients suffering from degenerative eye diseases, including age-related macular degeneration (AMD). In this study, we generated iPSCs from human dermal fibroblasts (HDFs) by electroporation with episomal plasmid vectors encoding OCT4, SOX2, KLF4, L-MYC together with p53 suppression. Intriguingly, cell reprogramming resulted in a metastable transcriptional activation and selective demethylation of neural and retinal specification-associated genes, such as OTX2, RX1 and SIX3. In contrast, RPE progenitor genes were transcriptionally silent in HDFs and descendant iPSCs. Overexpression of OCT4 and SOX2 directly stimulated the expression of OTX2, RX1 and SIX3 in HDFs and iPSCs. Luciferase and chromatin immunoprecipitation (ChIP) assays further identified an OCT4- and two SOX2-binding sites located in the proximal promoter of OTX2. Histone acetylation and methylation on the local promoter also participated in the reactivation of OTX2. The transcriptional conversion of RX1 and SIX3 genes partially attributed to DNA demethylation. Subsequently, iPSCs were induced into the RPE cells displaying the characteristics of polygonal shapes and pigments, and expressing typical RPE cell markers. Taken together, our results establish readily efficient and safe protocols to produce iPSCs and iPSC-derived RPE cells, and underline that the reactivation of anterior neural transcription factor OTX2, eye field transcription factor RX1 and SIX3 in iPSCs is a feature of pluripotency acquisition and predetermines the potential of RPE differentiation.
Keywords: induced pluripotent stem cells, retinal pigment epithelium, reprogramming, epigenetic modification

 Global reach, higher impact
Global reach, higher impact