Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2019; 15(3):680-687. doi:10.7150/ijbs.30464 This issue Cite
Review
Reactive Oxygen Species Related Noncoding RNAs as Regulators of Cardiovascular Diseases
1. Qingdao University, Deng Zhou Road 38, Qingdao 266021, China
2. Biochemistry Department No.2 Middle School Qingdao Shandong P.R. China 266000
*The authors contributed equally to this work.
Abstract
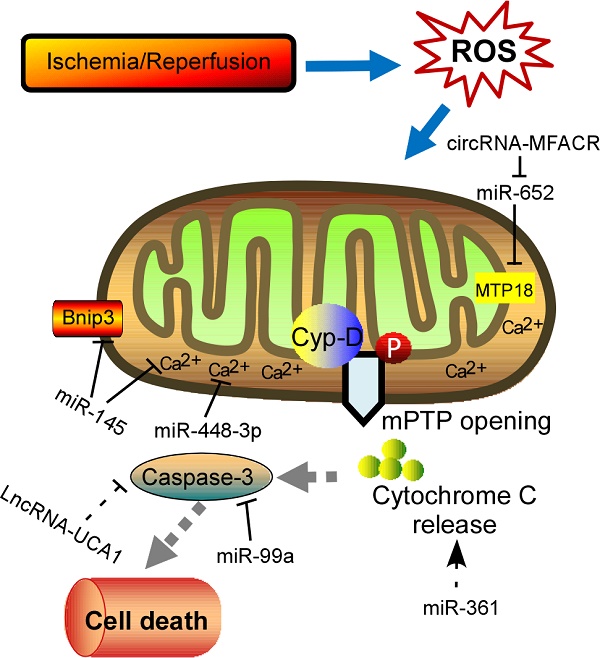
Reactive oxygen species (ROS) are a class of reactive molecules that have been implicated in a variety of cardiovascular diseases, accompanied by disorder of multiple signaling events. As cardiomyocytes maintain abundant of mitochondria, which supply the major source of endogenous ROS, oxidative damage to mitochondria often drives apoptotic cell death and initiates cardiac pathology. In recent years, non-coding RNAs (ncRNAs) have received much attention to uncover their roles in regulating gene expression during those pathological events in the heart, such as myocardial infarction, cardiac hypertrophy, and heart failure. Emerging evidences have highlighted that different ROS levels in response to diverse cardiac stresses result in differential expression of ncRNAs, subsequently altering the expression of pathogenetic genes. However, the knowledge about the ncRNA-linked ROS regulatory mechanisms in cardiac pathologies is still largely unexplored. In this review, we summarize the connections that exist among ROS, ncRNAs, and cardiac diseases to understand the interactions among the molecular entities underlying cardiac pathological events in the hopes of guiding novel therapies for heart diseases in the future.
Keywords: ROS, ncRNAs, heart diseases, mitochondria, cardiomyocyte death

 Global reach, higher impact
Global reach, higher impact