Impact Factor ISSN: 1449-2288
Int J Biol Sci 2020; 16(9):1604-1615. doi:10.7150/ijbs.38930 This issue Cite
Research Paper
The lncRNA Rhno1/miR-6979-5p/BMP2 Axis Modulates Osteoblast Differentiation
1. Department of Orthopaedics, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
2. Division of Plastic Surgery, Brigham and Women's Hospital, Harvard Medical School, Boston, 02215, USA.
*These authors contributed equally to this work.
Abstract
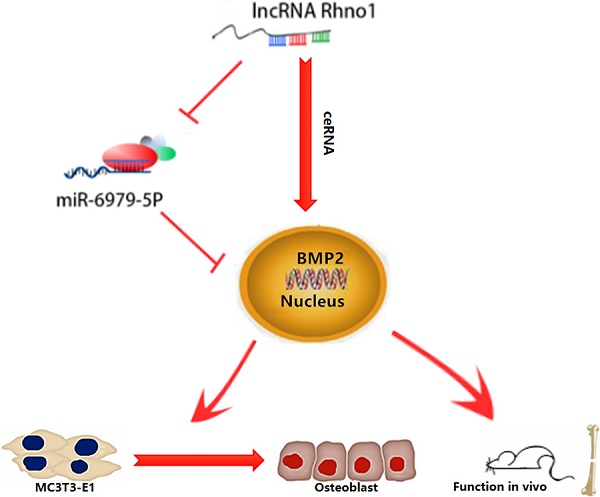
The roles of long non-coding RNAs (lncRNAs) and micro RNAs (miRNAs) as regulators of mRNA expression in various diseases have recently been reported. Osteoblast differentiation is the vital process which mediates bone formation and fracture healing. In present study, we found microRNA-6979-5p (miR-6979-5p) to be the most differentially expressed miRNA between normal bone and calluses of mice, and overexpression of miR-6979-5p was negatively associated with osteoblast differentiation. Through luciferase assays, we found evidence that bone morphogenetic protein 2 (BMP2) is a miR-6979-5p target gene that positively regulates osteoblast differentiation. We further identified the lncRNA Rhno1 as a competing endogenous RNA (ceRNA) of miR-6979-5p, and we verified that it was able to influence osteoblast differentiation both in vitro and in vivo. In summary, our data indicates that the lncRNA Rhno1/miR-6979-5p/BMP2 axis is a significant regulatory mechanism controlling osteoblast differentiation, and it may thus offer a novel therapeutic strategy for fracture healing.
Keywords: LncRNA, miRNA, mRNA, Fracture, BMP2

 Global reach, higher impact
Global reach, higher impact