Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2021; 17(4):1088-1100. doi:10.7150/ijbs.56800 This issue Cite
Research Paper
Genistein-3′-sodium sulfonate Attenuates Neuroinflammation in Stroke Rats by Down-Regulating Microglial M1 Polarization through α7nAChR-NF-κB Signaling Pathway
1. Key Laboratory of Prevention and treatment of cardiovascular and cerebrovascular diseases of Ministry of Education, Gannan Medical University, Ganzhou 341000, China.
2. Department of Physiology, School of Basic Medical Sciences, Gannan Medical University, Ganzhou 341000, China.
3. First Affiliated Hospital of Gannan Medical University, Ganzhou 341000, China.
4. Department of Pathobiology, JiangXi College of Traditional Chinese Medicine, Fuzhou, 344000, China.
Abstract
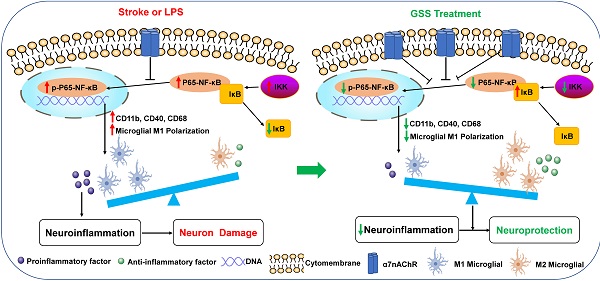
Microglial M1 depolarization mediated prolonged inflammation contributing to brain injury in ischemic stroke. Our previous study revealed that Genistein-3′-sodium sulfonate (GSS) exerted neuroprotective effects in ischemic stroke. This study aimed to explore whether GSS protected against brain injury in ischemic stroke by regulating microglial M1 depolarization and its underlying mechanisms. We established transient middle cerebral artery occlusion and reperfusion (tMCAO) model in rats and used lipopolysaccharide (LPS)-stimulated BV2 microglial cells as in vitro model. Our results showed that GSS treatment significantly reduced the brain infarcted volume and improved the neurological function in tMCAO rats. Meanwhile, GSS treatment also dramatically reduced microglia M1 depolarization and IL-1β level, reversed α7nAChR expression, and inhibited the activation of NF-κB signaling in the ischemic penumbra brain regions. These effects of GSS were further verified in LPS-induced M1 depolarization of BV2 cells. Furthermore, pretreatment of α7nAChR inhibitor (α-BTX) significantly restrained the neuroprotective effect of GSS treatment in tMCAO rats. α-BTX also blunted the regulating effects of GSS on neuroinflammation, M1 depolarization and NF-κB signaling activation. This study demonstrates that GSS protects against brain injury in ischemic stroke by reducing microglia M1 depolarization to suppress neuroinflammation in peri-infarcted brain regions through upregulating α7nAChR and thereby inhibition of NF-κB signaling. Our findings uncover a potential molecular mechanism for GSS treatment in ischemic stroke.
Keywords: Microglia polarization, Neuroinflammation, Ischemic stroke, α7nAChR-NF-κB signaling, Genistein-3′-sodium sulfonate.

 Global reach, higher impact
Global reach, higher impact