Impact Factor ISSN: 1449-2288
Int J Biol Sci 2022; 18(9):3621-3635. doi:10.7150/ijbs.70972 This issue Cite
Research Paper
NUF2 Drives Clear Cell Renal Cell Carcinoma by Activating HMGA2 Transcription through KDM2A-mediated H3K36me2 Demethylation
1. Department of Minimally Invasive Intervention, Peking University Shenzhen Hospital, Shenzhen 518000, Guangdong, China
2. Guangdong Provincial Key Laboratory of Systems Biology and Synthetic Biology for Urogenital Tumors, Shenzhen Key Laboratory of Genitourinary Tumor, Department of Urology, The First Affiliated Hospital of Shenzhen University, Shenzhen Second People's Hospital (Shenzhen Institute of Translational Medicine), Shenzhen, 518000, Guangdong, China
3. Institute of Precision Medicine, Peking University Shenzhen Hospital, Shenzhen 518000, Guangdong, China
4. Department of Community Surveillance, The First Affiliated Hospital of Shantou University Medical College, Shantou 515041, Guangdong, China
* These authors contributed equally to this work.
Abstract
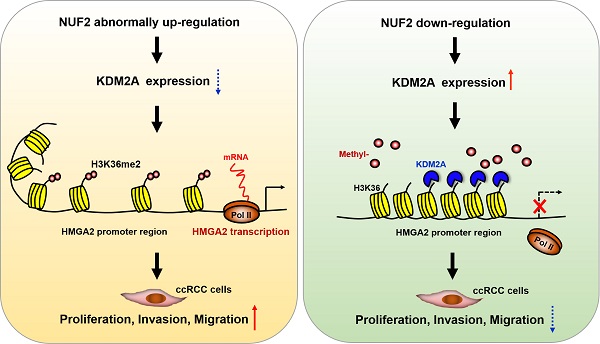
The poor sensitivity of clear cell renal cell carcinoma (ccRCC) to conventional chemotherapy and radiotherapy makes its treatment challenging. The Ndc80 kinetochore complex component (NUF2) is involved in the development and progression of several cancers. However, its role in ccRCC remains unclear. In this study, we investigated the biological functions and underlying mechanism of NUF2 in ccRCC. We found that NUF2 expression was increased in ccRCC and associated with poor prognosis. Altering NUF2 level affected cell proliferation, migration, and invasion. Moreover, NUF2 acted as a potential oncogene to promote the progression of ccRCC through epigenetic activation of high-mobility group AT-hook 2 (HMGA2) transcription by suppressing lysine demethylase 2A expression and affecting its occupancy on the HMGA2 promoter region to regulate histone H3 lysine 36 di-methylation modification. In addition, Kaplan-Meier and multivariate analysis revealed that patients whose NUF2 and HMGA2 were both elevated showed the shortest survival; and the number of upregulated markers acted as an independent predictor to evaluate survival probability. Thus, our results demonstrate that NUF2 promotes ccRCC progression, at least partly by epigenetically regulating HMGA2 transcription, and that the NUF2-HMGA2 axis could be an ideal therapeutic target and a promising prognostic indicator for ccRCC.
Keywords: Clear cell renal cell carcinoma, NUF2, HMGA2, KDM2A, H3K36me2

 Global reach, higher impact
Global reach, higher impact