Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2023; 19(5):1509-1527. doi:10.7150/ijbs.78669 This issue Cite
Research Paper
LTF Induces Radioresistance by Promoting Autophagy and Forms an AMPK/SP2/NEAT1/miR-214-5p Feedback Loop in Lung Squamous Cell Carcinoma
1. Department of Radiation Oncology, Fudan University Shanghai Cancer Center, Shanghai 200032, China.
2. Department of Oncology, Shanghai Medical College, Fudan University, Shanghai 200032, China.
3. Institute of Thoracic Oncology, Fudan University, Shanghai 200032, China.
4. Institute of Radiation Medicine, Shanghai Medical College, Fudan University, Shanghai 200032, China.
5. Department of Radiation Oncology, The First Affiliated Hospital of Nanjing Medical College, Nanjing 210029, China.
6. Department of Thoracic Surgery, Zhongshan Hospital, Fudan University, Shanghai 200032, China.
7. Department of Thoracic Surgery, the Second Affiliated Hospital of Soochow University, Suzhou 215000, China.
*These authors contributed equally as first authors.
Abstract
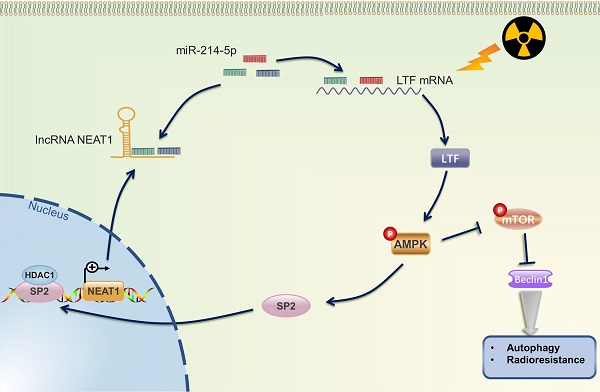
Radiotherapy is the most predominant treatment strategy for lung squamous cell carcinoma (LUSC) patients, but radioresistance is the major obstacle to therapy effectiveness. The mechanisms and regulators of LUSC radioresistance remain unclear. Here, lactotransferrin (LTF) is found to be significantly upregulated in radioresistant LUSC cell lines (H226R and H1703R) and clinical samples and promotes radioresistance of LUSC both in vitro and in vivo. Comprehensive enrichment analyses suggested that LTF potentially modulates autophagy in LUSC. Interestingly, the level of autophagy was raised in the radioresistant cells, and suppression of autophagy sensitized LUSC to irradiation. Functional experiments showed that LTF deficiency inhibits cellular autophagy through the AMPK pathway, ultimately leading to radiosensitization. Mechanistically, LTF can directly interact with AMPK to facilitate its phosphorylation and activate autophagy signaling. Moreover, NEAT1 functions as a ceRNA that targets miR-214-5p resulting in an increased LTF expression. Intriguingly, SP2, a transcription factor regulated by AMPK, induced NEAT1 expression by directly binding to its promoter region and thus forming a LTF/AMPK/SP2/NEAT1/miR-214-5p feedback loop. Our work reveals for the first time that LTF induces radioresistance by promoting autophagy and enhancing its self-expression via forming a positive feedback loop, suggesting that LTF is an appealing radiosensitization target for treating LUSC.
Keywords: lung cancer, radioresistance, LTF, autophagy

 Global reach, higher impact
Global reach, higher impact