10
Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2023; 19(7):2198-2219. doi:10.7150/ijbs.79651 This issue Cite
Review
Crosstalk between N6-methyladenosine (m6A) modification and noncoding RNA in tumor microenvironment
1. Department of Pathology, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
2. Departments of Ultrasound Imaging, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
3. Department of Pathology, School of Basic Medicine, Central South University, Changsha 410031, Hunan, China.
4. Key Laboratory of Hunan Province in Neurodegenerative Disorders, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
5. National Clinical Research Center for Geriatric Disorders, Xiangya Hospital, Central South University, Changsha 410008, Hunan, China.
* These authors jointly supervised this work.
Abstract
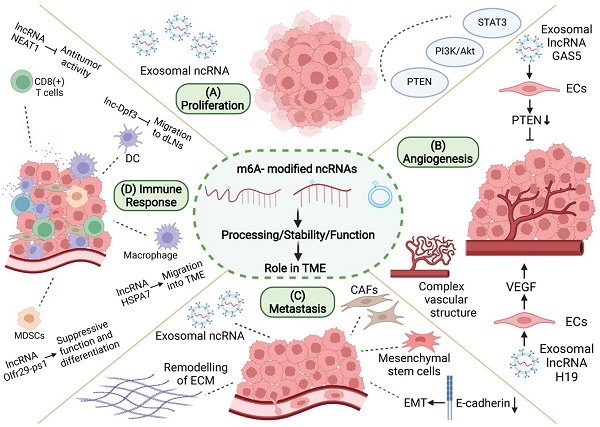
N6-methyladenosine (m6A) is the most abundant RNA modification in eukaryotes, and it participates in the regulation of pathophysiological processes in various diseases, including malignant tumors, by regulating the expression and function of both coding and non-coding RNAs (ncRNAs). More and more studies demonstrated that m6A modification regulates the production, stability, and degradation of ncRNAs and that ncRNAs also regulate the expression of m6A-related proteins. Tumor microenvironment (TME) refers to the internal and external environment of tumor cells, which is composed of numerous tumor stromal cells, immune cells, immune factors, and inflammatory factors that are closely related to tumors occurrence and development. Recent studies have suggested that crosstalk between m6A modifications and ncRNAs plays an important role in the biological regulation of TME. In this review, we summarized and analyzed the effects of m6A modification-associated ncRNAs on TME from various perspectives, including tumor proliferation, angiogenesis, invasion and metastasis, and immune escape. Herein, we showed that m6A-related ncRNAs can not only be expected to become detection markers of tumor tissue samples, but can also be wrapped into exosomes and secreted into body fluids, thus exhibiting potential as markers for liquid biopsy. This review provides a deeper understanding of the relationship between m6A-related ncRNAs and TME, which is of great significance to the development of a new strategy for precise tumor therapy.
Keywords: m6A, ncRNA, exosome, tumor metastasis, tumor microenvironment, biomarker, targeted therapy

 Global reach, higher impact
Global reach, higher impact