ISSN: 1449-2288
Int J Biol Sci 2023; 19(13):4181-4203. doi:10.7150/ijbs.85158 This issue Cite
Review
IκB kinase β (IKKβ): Structure, transduction mechanism, biological function, and discovery of its inhibitors
1. School of Chinese Materia Medica, State Key Laboratory of Component-Based Chinese Medicine, Tianjin State Key Laboratory of Modern Chinese Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China.
2. College of Pharmacy, Second Affiliated Hospital, Dalian Medical University, Dalian 116044, China.
3. School of Pharmaceutical Sciences, Health Science Center, Shenzhen University, Shenzhen 518061, China.
4. Faculty of Pharmaceutical Sciences, Toho University, Chiba 274-8510, Japan.
*These authors contributed equally to this work.
Abstract
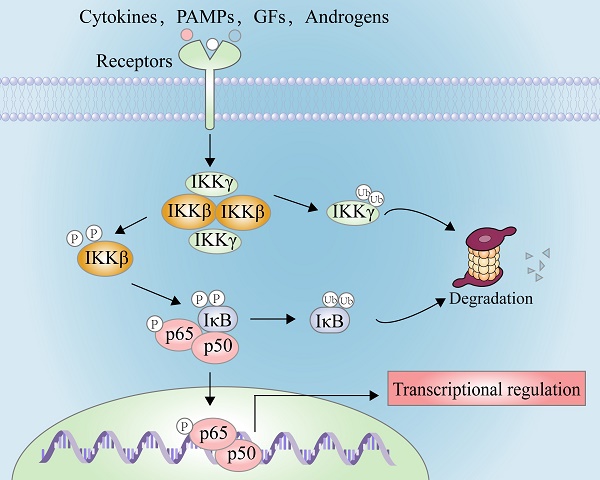
The effective approach to discover innovative drugs will ask natural products for answers because of their complex and changeable structures and multiple biological activities. Inhibitory kappa B kinase beta (IKKβ), known as IKK2, is a key regulatory kinase responsible for the activation of NF-κB through its phosphorylation at Ser177 and Ser181 to promote the phosphorylation of inhibitors of kappa B (IκBs), triggering their ubiquitination and degradation to active the nuclear factor kappa-B (NF-κB) cascade. Chemical inhibition of IKKβ or its genetic knockout has become an effective method to block NF-κB-mediated proliferation and migration of tumor cells and inflammatory response. In this review, we summarized the structural feature and transduction mechanism of IKKβ and the discovery of inhibitors from natural resources (e.g. sesquiterpenoids, diterpenoids, triterpenoids, flavonoids, and alkaloids) and chemical synthesis (e.g. pyrimidines, pyridines, pyrazines, quinoxalines, thiophenes, and thiazolidines). In addition, the biosynthetic pathway of novel natural IKKβ inhibitors and their biological potentials were discussed. This review will provide inspiration for the structural modification of IKKβ inhibitors based on the skeleton of natural products or chemical synthesis and further phytochemistry investigations.
Keywords: IKKβ, Inhibitor, Natural products, chemical synthesis, Bioactivity

 Global reach, higher impact
Global reach, higher impact