Impact Factor ISSN: 1449-2288
Int J Biol Sci 2025; 21(6):2606-2628. doi:10.7150/ijbs.108803 This issue Cite
Review
Unraveling the breast cancer tumor microenvironment: crucial factors influencing natural killer cell function and therapeutic strategies
1. Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai, China.
2. Shanghai Research Institute of Acupuncture and Meridian, Shanghai, China.
3. Shanghai University of Traditional Chinese Medicine, Shanghai, China.
4. Integrated Traditional Chinese and Western Medicine Breast Department, Longhua Hospital Shanghai University of Traditional Chinese Medicine, Shanghai, China.
* These authors have contributed equally to this work and share first authorship.
Received 2024-12-14; Accepted 2025-3-4; Published 2025-3-24
Abstract
Natural killer (NK) cells have emerged as a novel and effective treatment for breast cancer. Nevertheless, the breast cancer tumor microenvironment (TME) manifests multiple immunosuppressive mechanisms, impeding the proper execution of NK cell functions. This review summarizes recent research on the influence of the TME on the functionality of NK cells in breast cancer. It delves into the effects of the internal environment of the TME on NK cells and elucidates the roles of diverse stromal components, immune cells, and signaling molecules in regulating NK cell activity within the TME. It also summarizes therapeutic strategies based on small-molecule inhibitors, antibody therapies, and natural products, as well as the progress of research in preclinical and clinical trials. By enhancing our understanding of the immunosuppressive TME and formulating strategies to counteract its effects, we could fully harness the therapeutic promise of NK cells in breast cancer treatment.
Keywords: natural killer cells, breast cancer tumor microenvironment, intrinsic metabolic factors, stromal components, immunosuppressive mechanisms, therapeutic strategies
Introduction
Breast cancer is a highly prevalent female malignancy worldwide [1]. Immunotherapy has emerged as a pivotal treatment modality for breast cancer [2]. Natural killer (NK) cells play a critical role in tumor cell recognition and elimination through the secretion of cytotoxic granules, chemokines, and proinflammatory cytokines [3]. In locally advanced (T3-4, N1-2, and M0) breast cancer, NK cells exhibit reductions in their absolute number, cytotoxicity, and lytic capacity toward K562 cells [4,5]. An increase in the activity of peripheral NK (pNK) cells after preoperative neoadjuvant chemotherapy is highly associated with the disappearance of axillary lymph node metastases, suggesting that the systemic activation of pNK cells may improve the elimination of metastatic tumors in patients with breast cancer [6]. In patients with breast cancer, the count of pNK cells is associated with the objective response to chemotherapy [7], as well as with long overall survival (OS) and relapse-free survival (RFS) [8]. The function and activity of NK cells can predict the prognosis and treatment outcomes of breast cancer [9].
NK cells exhibit the unique capability to target tumor cells in an antigen-independent fashion, distinguishing them from adaptive immune cells, such as T lymphocytes [10]. Additionally, the lack of surface T cell receptors on NK cells diminishes the risks associated with cytokine release syndrome and graft-versus-host disease, further highlighting their therapeutic safety and immense potential [11,12]. In a phase I trial, autologous NK cells from human epidermal growth factor receptor 2 (HER2)-positive breast cancer patients were infused after culture and expansion, demonstrating certain antitumor activity and good tolerability [13].
However, breast cancer cells can evade recognition by NK cells through various mechanisms [14]. For example, NK cells isolated from the peripheral blood of patients with breast cancer exhibit the reduced activity of their surface activating receptors and increased levels of inhibitory receptors [15]. Previous therapeutic approaches have mostly focused on enhancing the functional alterations of NK cells themselves, such as using specific antibodies to target CD16 [16], NK group (NKG) 2D [17], and NKG2A gene knockout [18]. Apart from the inherent functional limitations of NK cells, the tumor microenvironment (TME) harbors a multitude of immunological mechanisms that impair and hinder the functionality and infiltration of NK cells, ultimately facilitating tumor escape [19]. In pyMT mice, the developmental phenotypes of NK cells in peripheral tissues, such as the spleen, differs from those in tumors, suggesting that the TME may convert activated NK cells into an immature, inactivated, or potentially tumor-promoting state [20]. The TME of breast cancer is highly heterogeneous, with adverse metabolic factors, such as hypoxia, promoting the polarization of macrophages toward the M2 phenotype, accelerating the maturation of myeloid-derived suppressor cells (MDSCs), and inducing immunosuppression [21]. Studies have demonstrated that diabetes increases the accumulation of IL-10-positive regulatory T cells (Tregs) and transforming growth factor-β (TGF-β)-positive MDSCs in the spleen and tumors of mice but decreases the percentage of NK cells producing interferon gamma (IFN-γ) and production of natural cytotoxicity triggering receptor 1 (NCR1/NKp46), PRF, granzyme B (GZMB), CD107a, and interleukin (IL)-17. The accumulation of immunosuppressive cells and exhaustion of NK cells in a hyperglycemic culture environment may be mediated by indoleamine 2,3-dioxygenase (IDO) [22]. In the TME, NK cells interact with other immune cells through inflammatory cytokines, such as IFN-γ, IL-1β, and tumor necrosis factor-alpha (TNF-α) [23]. Nonetheless, the efficacy of NK cell-based therapies for breast cancer remains suboptimal.
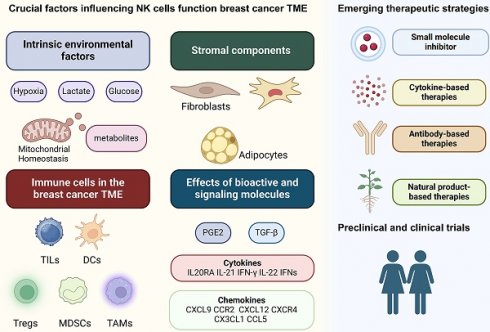
In this review, we delineate the effects of environmental and metabolic factors, stromal cells, immune cells, and cytokines on NK cells within the breast cancer TME and their subsequent effects on tumorigenesis. Given that tumor metabolism and tumor immunology have recently become attractive research areas, this review may provide insights for cross-disciplinary research in these fields. We also summarize relevant studies on NK cells in breast cancer using small-molecule inhibitors, antibody therapies, and natural products and discuss the progress of preclinical and clinical trials (Figure 1).
Graphical abstract.
Effects of intrinsic environmental factors in the TME on NK cells
Hypoxia
Hypoxia exerts a dual influence within the TME of breast cancer because it can contribute to promoting tumor proliferation and impede the cytotoxic function of NK cells simultaneously [24]. In hypoxic environments, NK cells demonstrate reduced killing capacity and diminished antibody-dependent cellular cytotoxicity (ADCC) against breast cancer, as well as prostate and lung cancer cells [25]. Single-cell sequencing analyses uncovered a link between the infiltration of NK cells and absence of hypoxia-inducible factor 1-alpha (HIF-1α) expression in murine breast cancer tumors. The depletion of HIF-1α elevates the expression of IFN-γ, activates NK cells, and enhances tumor infiltration, thereby highlighting the inhibitory effect of HIF-1α on NK cell activity [26]. HIF-1α-induced hypoxic conditions attenuate interferon signaling, leading to the decreased cytotoxic efficacy of immune cells, including NK cells, and confer resistance to the blockade of programmed death protein 1 (PD-1) through epigenetic reprogramming mediated by histone deacetylase (HDAC) 1/polycomb repressive complex 2 (PRC2) [27]. Treatment with the HDAC inhibitor entinostat can restore the expression of IFN-γ, TNF-α, and GZMB in hypoxic NK cells. Furthermore, hypoxia-induced autophagy contributes to the degradation of GZMB, reducing the sensitivity of breast cancer cells to NK-mediated cell lysis [28].
Lactate
During tumorigenesis, hyperproliferative tumor cells generate elevated lactate levels, which can reach 40 mM in the extracellular environment, presenting a notable 20-fold increase relative to the lactate levels in healthy blood or tissues [29]. Lactate, a prominent metabolic byproduct within the TME, considerably contributes to tumor immune evasion and invasiveness. Specifically, it suppresses the activation and proliferation of CD8+ T lymphocytes, NK cells, and dendritic cells (DCs), thereby undermining immune surveillance and diminishing the cytotoxic properties of therapeutic NK cells intended for adoptive therapy [30,31]. Lactate production is regulated by the enzymatic activity of lactate dehydrogenase (LDH) and functionality of the monocarboxylate transporter (MCT) [32,33]. The high expression of LDHB facilitates the reverse conversion of lactate into pyruvate by decreasing acidity microenvironment via MCT1. This improved environment promotes NK cell activation, leading to increased IFN-γ and GZMB production, ultimately augmenting tumor-specific immune responses against breast cancer cells [34]. The depletion of MCT4 initiates autophagic responses as a compensatory metabolic mechanism to reduce lactate export in tumors. This phenomenon reverses the acidic microenvironment, consequently enhancing the cytotoxic activity of NK cells against breast cancer cells [35].
Glucose
Glucose, a vital energy source in biological systems, is metabolized via multiple pathways, including glycolysis, the pentose phosphate pathway, and oxidative phosphorylation. Under normal cellular physiological conditions, adenosine (ADO) triphosphate is primarily generated through oxidative phosphorylation in aerobic environments, whereas glycolysis prevails under anaerobic conditions [36]. NK cells depend on glucose as their primary metabolic substrate, utilizing the glucose available in the TME to fulfill their energetic and functional needs [37]. The inhibition of glucose transporter proteins, specifically glucose transporter (GLUT) 1, GLUT2, and GLUT3, can diminish the glycolytic capacity of NK cells. This reduction can lead to the compromised proliferation of cells, decreased secretion of IFN-γ, and hindered serial killing efficacy of NK cells [38]. The activation of the TNF-α/TNF receptor 2 (TNFR2) signaling axis upregulates the expression of CD25 (IL-2 receptor alpha) and nutrient transporters in NK cells. This phenomenon promotes a metabolic shift toward aerobic glycolysis. Conversely, the genetic deletion of TNFR2 suppresses CD25 expression and glycolytic metabolism, thereby compromising the functionality of NK cells [39].
Mitochondrial homeostasis
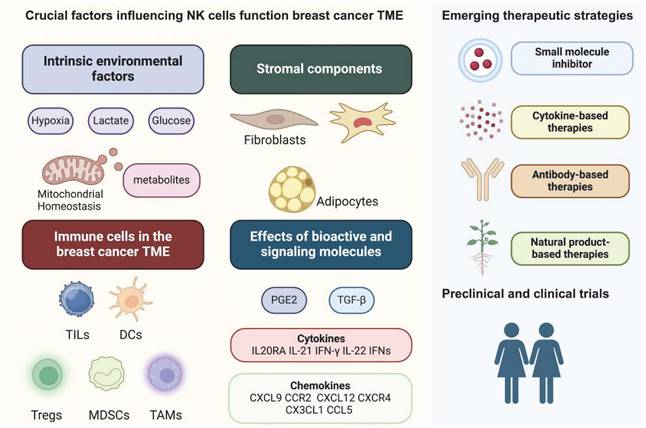
Mitochondria play a critical role in shaping the phenotypic state of NK cells, and the maintenance of their metabolic homeostasis serves as a primary modulator of NK cell activation, survival, and effector functions [40]. Substantial disturbances in mitochondrial metabolism can severely impede the antitumor efficacy of NK cells [41]. Within the TME, NK cells encounter several challenges, including hypoxia; the scarce availability of glucose; and the accumulation of tumor-derived metabolic waste products, such as lactic acid. These environmental stressors contribute to metabolic strain and hinder NK cell activation [42]. Recent investigations have uncovered that in tumor-infiltrating NK cells, disruptions in mitochondrial homeostasis manifest as the shrinkage or fragmentation of mitochondria, elongation of the mitochondrial network, and elevated production of reactive oxygen species (ROS). Mitochondrial fragmentation is correlated with diminished cytotoxic potential and compromised NK cell function, enabling tumors to evade immune surveillance by NK cells, ultimately leading to poor patient outcomes [43]. Notably, exercise interventions conducted under hypoxic conditions demonstrated protective effects on the tumoricidal capabilities of NK cells against triple-negative breast cancer (TNBC) by mitigating mitochondrial oxidative stress [44]. Consequently, targeting the regulatory factors involved in maintaining the mitochondrial metabolic homeostasis of NK cells represents a promising therapeutic approach [45]. Collectively, the above insights reveal that the adverse conditions prevalent in the TME, such as hypoxia, lactate accumulation, and limited glucose supply, affect NK cell function through multifaceted mechanisms (Figure 2).
Crucial environmental factors associated with NK cell function in the TME of breast cancer. a) Hypoxia modulates epigenetics through the HDAC1/PRC2 axis, thereby influencing the NF-κB and STAT signaling pathways. Hypoxia regulates IFN-γ secretion in NK cells. Furthermore, HIF-1α-induced autophagy promotes the degradation of GZMB. b) Glucose, as the primary energy source for NK cells, is critical for the function of these cells. The upregulation of GLUT1/2/3 and TNFα-TNFR2 signaling facilitates glucose uptake, promoting glycolysis and enhancing the antitumor activity of NK cells. c) Lactate production in the TME is tightly coupled with LDHB activity. MCT4 mediates lactate export from tumor cells, whereas MCT1 can reverse the acidic microenvironment. This modulation ultimately augments NKp46 expression and NK cell functionality. d) The hypoxic microenvironment, glycolytic metabolism of tumor cells, and excretion of lactate within breast cancer tumors considerably affect the mitochondrial homeostasis of NK cells. This disruption leads to mitochondrial shrinkage and fragmentation, compromising the antitumor efficacy of NK cells.
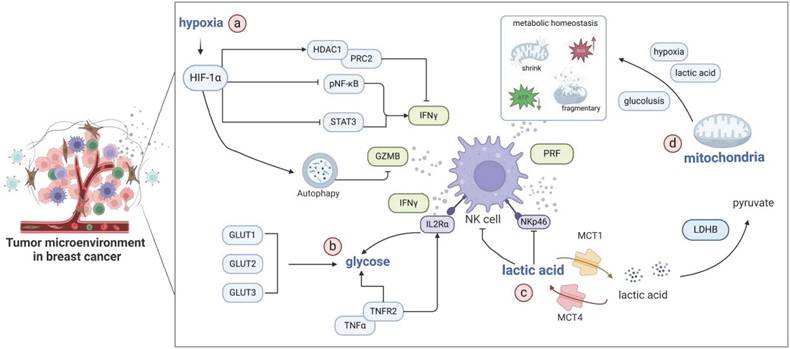
Metabolites
NK cell functionality is modulated by several factors, including metabolites, such as ADO, IDO, and uric acid, and perturbations in serine-sphingolipid metabolism (Figure 3). ADO, an immunosuppressive metabolite, engages ADO receptors on NK and T cells, thereby suppressing their activation [46]. Research has indicated that reducing intratumoral ADO levels can boost the activation of NK and CD8+ T cells, retard tumor growth, and enhance survival rates [47]. Furthermore, ADO antagonists have exhibited potential in revitalizing lymphocyte activity specifically in breast cancer [48].
Main metabolites in the TME of breast cancer that affect NK cell function. a) IDO catalyzes the degradation of Trp into L-Kyn, subsequently downregulating the expression of the NK cell activation receptors NKG2D and NKp46. Additionally, IDO upregulates HLA-G expression, thereby diminishing NK cell functionality. b) In DNA-damaged tumor cells, uric acid activates the TAK1/NF-κB/ERK signaling pathway, ultimately resulting in the upregulation of NKG2D ligands (MICA/B) and enhancement of antitumor immune responses. c) ADO exerts immunosuppressive effects by engaging ADO receptors on NK cells. d) Disruptions in serine-sphingomyelin metabolism hinder synapse formation on NK cell membranes, compromising the tumor cell recognition and elimination capabilities of NK cells.
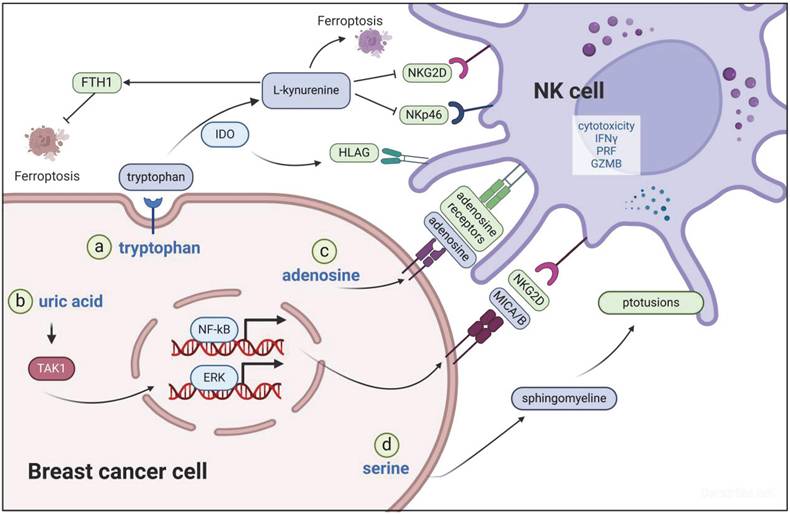
IDO, the only rate-limiting enzyme outside the liver capable of catalyzing tryptophan (Trp) metabolism, is constitutively overexpressed in tumors, enabling the continuous consumption of Trp within the TME and its conversion into L-kynurenine (Kyn) [49]. Compared with patients with low IDO expression in TNBC, those with high IDO-1 expression exhibit decreased NK cell activity [50] because IDO-1 impairs the cytotoxicity of NK cells by upregulating the expression of human leucocyte antigen (HLA)-G on TNBC cells. In TNBC mice, the silencing of IDO-1 or injection of the HLA-G antibody could rescue NK cell function, which in turn inhibits tumor growth.
In a diabetic model of 4T1 mice, an increase in IDO was found to result in the reduced cytotoxicity of NK cells, and IDO inhibitors almost completely restore the phenotype of suppressed the function of NK cells [22]. A TOP study (NCT02883699) revealed that IDO levels in patients with breast and prostate cancer can be influenced by various exercise regimens, subsequently affecting NK cells. Specifically, acute exercise has been found to elevate IDO levels, whereas chronic exercise helps regulate IDO levels to preserve NK cell function [51].
Kyn is derived from the IDO-catalyzed catabolism of the essential human amino acid Trp. It is an important immunosuppressive molecule. Compared with that in normal tissues adjacent to tumors, the Kyn content in head and neck squamous cell carcinoma (HNSCC) is remarkably elevated with a notable fold change [52]. In colorectal cancer, a high Kyn:Trp ratio is associated with an increased risk of mortality [53]. Kyn can enable disseminated breast cancer cells to resist ferroptosis by upregulating the levels of ferritin heavy chain 1 (FTH1) [54]. Interestingly, L-Kyn can, in turn, induce ferroptosis in NK cells via a nonaryl hydrocarbon receptor [55]. L-Kyn has been found to inhibit NK cell function in breast cancer, and this inhibition results in the impaired NK cell killing of target cells recognized through NKp46 and NKG2D [56]. However, the effect of L-Kyn on NK cells, especially those in breast cancer, remains largely unknown and needs further exploration.
Uric acid serves as the final product of purine catabolism and has been long been acknowledged as a danger signal for activating the immune system [57]. In DNA-damaged tumor cells, uric acid can activate the TGF-β-activated kinase (TAK1)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)/extracellular regulated protein kinase (ERK) signaling pathway, ultimately resulting in the upregulation of NKG2D ligands (MICA/B) and enhancement of antitumor immune responses [58]. Disturbances in serine-sphingolipid metabolism within the TME may impede synapse formation on NK cell membranes, thereby compromising the tumor cell recognition and elimination capabilities of NK cells [59]. Consequently, sphingomyelinases are being explored as potential targets in antitumor combination therapies aiming to restore optimal NK cell function [60].
Effects of the stromal components of the TME on NK cells
Fibroblasts
Cancer-associated fibroblasts (CAFs) constitute a prominent cell subset within the tumor stroma, exhibiting heightened activity and proliferative capacity compared with normal fibroblasts. They play a pivotal role in facilitating immune evasion [61,62]. However, the absence of specific markers has led to the classification of CAFs into diverse subpopulations [63].
The commonly recognized classical subgroups of CAFs mainly include progenitor cell-type CAFs (proCAFs), myofibroblast-type CAFs (myCAFs), matrix-producing CAFs (matCAFs), and inflammatory CAFs (iCAFs). proCAFs have high proliferative capacity and differentiation potential and play an important role in the early stages of tumors. MyCAFs are characterized by the high expression of α-SMA, which is mainly involved in extracellular matrix (ECM) remodeling and tissue fibrosis and promotes tumor invasion and metastasis [64]. MatCAFs mainly synthesize and secrete ECM proteins, which affect the proliferation and migration of tumor cells. ICAFs regulate the immune microenvironment mainly through the secretion of cytokines and chemokines and promote the progression of tumors [65]. In human and murine breast cancers, the ECM deposited by senescent CAFs directly suppresses NK cytotoxicity against tumor cells, which include a plethora of collagens and enzymes that modify collagen fibers [66]. Coculture with ECM-myCAFs and TGFβ-myCAFs leads to reduced levels of PRF and GZMB in NK cells, accompanied with an increase in the expression of the inhibitory receptor NKG2A [67].
In addition to the classical subgroups of CAFs, some subgroups of CAFs that have only been identified in recent years are gradually being discovered and valued. The membrane protein podoplanin (PDPN) is a well-known cancer-promoting molecule [68]. It is a type I transmembrane glycoprotein that contains three platelet aggregation-stimulating structural domains, which could specifically characterize a subpopulation of CAFs with proangiogenic functions [69]. In patients with node-negative and hormone receptor-positive/HER2-negative breast cancer subtypes, the positive expression of PDPN in CAFs is associated with a high Ki67 labeling index, increased tumor-infiltrating lymphocytes (TILs), and a progesterone receptor-negative status and is remarkably associated with a short disease-free survival (DFS) [70].
Notably, PDPN-positive CAFs actively suppress ADCC mediated by functional NK cells through the secretion of immunosuppressive factors, such as IDO-1 and Trp 2,3-dioxygenase (TDO-2) [71]. PDPN-positive CAFs are associated with low IL-2 activity and trastuzumab resistance in patients with HER2-positive breast cancer [72]. PMab-117-mG2a, a cancer-specific monoclonal antibody (mAb), selectively targets PDPN in tumor cells and exerts ADCC and remarkable antitumor effects in the presence of effector splenocytes [73]. The PDPN antagonist peptide CY12-RP2 binds specifically to PDPN and enhances the antitumor capacity of PBMC, including increasing the ratios of CD3+CD4+ T, CD3+CD8+ T, and CD49b+ granzyme B+ NK cells and abrogating the immunosuppressive effects of PDPN [74].
Dickkopf-1 (DKK1) is a secreted protein and regulator of the Wnt signaling pathway [75]. Its upregulation could inhibit the therapeutic effect of paclitaxel by hindering the infiltration and activity of CD8+ T cells in the TME [76]. Recent studies [77] have highlighted DKK1 as a marker of immune cell populations that may contribute to a promoting TME by inhibiting NK cell activation in breast cancer. The targeted knockout of DKK1 in CAFs has been shown to hinder tumor growth. DKN-01 is an IgG4 antibody that effectively and specifically neutralizes human and mouse DKK1, and DKN-01 treatment promotes the induction of the NK-activating cytokines IL-15 and IL-33, as well as enhances CD45 cell recruitment, which is important in reducing lung metastasis in a mouse model of 4T1 breast cancer [78].
CAR-T cell therapy targeting CAFs is another potential therapeutic strategy. Fibroblast activation protein (FAP) is a specific marker on the surfaces of CAFs, and its high expression on CAFs makes it an ideal target for CAR-T cell therapy [79]. CAR-T cells targeting FAP can deplete CAFs and promote in vivo T cell infiltration and antitumor activity in a TNBC in situ mouse model composed of patient-derived CAFs and tumor cells [80].
Overall, the immunosuppressive roles of CAFs and their modulation of NK cells play a critical role in cancer immune evasion, underscoring their potential as therapeutic targets [81]. Future research should focus on elucidating the mechanisms underlying the heterogeneity and NK cell interactions of CAFs, as well as developing strategies to target CAFs and enhance NK cell-mediated antitumor immunity.
Adipocytes
Adipocytes, the predominant cell type in breast cancer tumor tissue, serve not only as storage units but also secrete adipokines, such as leptin, adiponectin, and IL-6 [82,83]. They are categorized into white, brown, and beige types and play distinct roles in tumor biology. White adipocytes, in particular, store energy in the form of large lipid droplets and are often implicated in tumor progression [84]. Furthermore, adipocytes modulate the functionality of NK cells [85,86]. Recent studies have also revealed that leptin decreases the cytotoxicity of NK cells against breast cancer cells by upregulating PPARG coactivator 1 alpha (PGC1α), suggesting that targeting leptin or PGC1α could enhance NK-based immunotherapy against breast cancer [87]. The exposure of BT-474 cells to adipocyte-conditioned media considerably reduces their sensitivity to trastuzumab-mediated ADCC. Similarly, trastuzumab has been proven to be ineffective in inhibiting BT-474 tumor growth in the presence of lipomas [88]. In a study using female BALB/c nude mice fed a high-calorie diet (5320 kcal/kg) for six months, accelerated breast cancer progression was observed alongside a reduction in splenic NK cell count and cytotoxicity [89]. Elevated plasma levels of leptin and IL-6 were noted in a postmenopausal obesity tumor-bearing mouse model of breast cancer. Additionally, in obese women with breast cancer, the decreased expression of the NK cell activation receptor NKp46 in peripheral blood suggests compromised NK cell function [90]. Coculturing NK cells with human adipose-derived stem cells leads to the reduced degranulation and lytic abilities of NK cells [91]. In a breast cancer animal model, IL-1β suppresses lipolysis, resulting in neutral lipid accumulation in lung mesenchymal cells. These lipid-laden mesenchymal cells then transport lipids to tumor and NK cells via exosome-like vesicles, ultimately leading to NK cell dysfunction and breast cancer metastasis [92].
Mesenchymal stem cells
In the TME, CAFs can be induced to differentiate from mesenchymal stem cells (MSCs), which have a fibroblast-like morphology, the ability to secrete ECM components, and immunomodulatory capabilities [93]. MSCs have the ability for self-renewal and multidirectional differentiation [94]. Therefore, they can promote tumor progression by differentiating into CAFs or other cells, enhancing angiogenesis, or regulating immune cell function. By contrast, CAFs directly promote tumor growth and metastasis by secreting cytokines, remodeling the ECM, and forming an immunosuppressive microenvironment [95].
When exposed to the TME, they MSCs transform into tumor-associated MSCs (T-MSCs) [96]. Research has indicated that lung cancer-derived MSCs diminish the tumor-killing capacity of NK cells and activated T cells originating from peripheral blood monocytes [97]. MSCs have been observed to foster breast tumor growth [98]. Furthermore, T-MSCs have been found to hinder the production of proinflammatory cytokines and cytotoxic molecules, such as granzyme and PRF, by NK cells [99]. Nevertheless, studies have also revealed that MSCs overexpressing Sirtuins 1 (MSCs-Sirt1) notably suppress tumor growth by inhibiting cell proliferation and promoting apoptosis. This effect is mediated by MSCs-Sirt1 to upregulate chemokine (C-X-C motif) ligand (CXCL)-10 and IFN-γ, consequently attracting NK cells within the TME to restrain breast tumor growth potently [100].
The TME of breast cancer encompasses a sophisticated network of cellular components, which collectively influence the functionality of NK cells (Figure 4). In current research endeavors, researchers often direct their attention predominantly toward the immunoinhibitory effects stemming from metabolism within the TME. However, the intricate interplay among cells within the TME underscores its inherent complexity and underscores the importance of adopting a holistic perspective in understanding tumor immunology. Elucidating these intricate interactions will pave the way for the development of potent immunotherapy strategies, ultimately enhancing the effectiveness of cancer treatment.
Key stromal components within the breast cancer TME regulate NK cell functionality. a) The PDPN-positive CAF subset impairs ADCC via the release of immunosuppressive enzymes, specifically IDO-1 and TDO-2. Senescent CAFs secrete ECM components that selectively undermine the cytotoxicity of NK cells. The targeted deletion of DKK1 in CAFs restores NK cell activation and tumoricidal capacity. ECM- and TGF-β-myCAFs downregulate PERF and GZMB expression while upregulating NKG2A. Notably, DPP4 and yes-associated protein 1 drive the differentiation of CAFs toward the ECM-myCAF phenotype. b) Adipocytes suppress the lytic capacity and degranulation of NK cells by releasing immunosuppressive factors, such as leptin, IL-6, and IDO-1. c) MSCs hinder the secretion of PERF and GZMB. Remarkably, adipose-derived MSCs impart immunosuppressive effects by transferring lipids to NK cells via exosome vesicles. However, MSCs overexpressing SIRT1 can augment the antitumor capabilities of NK cells through the release of CXCL10 and IFN-γ.

Immune-activating cells in the breast cancer TME
TILs
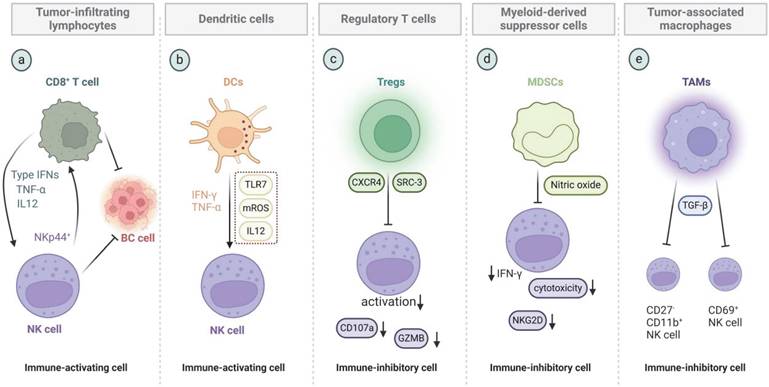
Immunoactivating and immunosuppressive cells within the TME exert distinctly different effects on NK cells (Figure 5). TILs have been established as reliable biomarkers for predicting therapeutic response and prognosis in breast cancer [101]. A systematic review and meta-analysis revealed that among 6161 patients with TNBC across 29 clinical studies, high TIL expression is associated with considerably improved DFS, OS, and pathological complete response outcomes. Stromal TILs are correlated with progression-free survival (PFS) in patients with metastatic breast cancer (MBC) [102]. CD8+ T cells, a crucial subset of TILs, exhibit tumoricidal activity. They directly kill tumor cells by recognizing antigens on the tumor cell surface and releasing cytotoxic molecules, such as PRF and GZMB [103,104]. Although CD8+ T and NK cells differ functionally, they can collaborate in certain contexts to enhance immune responses in lieu of the direct action of CD8+ T cells on NK cells [105]. CD8+ T cells also influence NK cell activity through cytokines, such as IL-12, type I IFNs, and TNF-α [106]. Anil Shanker found that the gene expression profile of NK cell-infiltrating tumors in mice with specific CD8+ T cells indicate an activated effector phenotype [107]. NKp44+ NK cells can regulate HLA-DP+CD8+ T cells in individuals expressing the HLA-DP haplotype that binds to NKp44, thereby influencing CD8+ T cell clonality [108].
Immune-activating and -inhibiting cells in the breast cancer TME that affect NK cell function. a) TILs, which are represented by CD8+ T cells, and NK cells exert their respective antitumor effects. Meanwhile, CD8+ T cells can enhance the function of NK cells through cytokines, such as type I IFNs, TNF-α, and IL12. NKp44+ NK cells can also affect the clonality of CD8+ T cells. b) DCs enhance NK cell function by releasing IFN-γ and TNF-α and regulating the TLR7/mROS/IL12 axis. c) CXCR4-positive and SRC-overexpressing Tregs inhibit the activation of NK cells and release of GZMB and CD107a. d) MDSCs inhibit the killing ability of NK cells through mechanisms, such as nitric oxide production. e) TAMs could inhibit the activation of CD69+ NK cells and TGF-β-mediated maturation of CD27-CD11b+ NK cells.
DCs
With the deepening of research on immune cells, scholars have discovered that DCs are also a key factor in converting the “cold” TME of solid tumors into a “hot” one [109]. Antigen presentation by DCs is crucial in immune surveillance and integrated antitumor immune responses [110,111], and functional defects in DCs are associated with immune suppression in cancer, leading to tumor progression [112]. NK cells and DCs can interact and activate each other, with activated DCs directly activating NK cells, thereby bridging innate and adaptive immune responses [113, 114]. Breast cancer chemotherapy drugs, such as doxorubicin (Dox), are immunogenic cell death inducers that promote DC maturation, facilitating TIL infiltration and NK cell recruitment, increasing the secretion of proinflammatory cytokines (IFN-γ, TNF-α, and IL-6), and resulting in enhanced antitumor activity [115]. CD11c+ DCs in peripheral blood can activate toll-like receptor 7 (TLR7) to trigger the production of ROS, driving the TLR7/mROS/IL-12 axis to enhance the tumor-killing ability of NK cells [116].
Immune-inhibiting cells in the breast cancer TME
Tregs
Complex interactions among various types of immune cells exist in the breast cancer TME [117,118]. CD4+FOXP3+ Tregs are an important cell type involved in immunosuppression in cancer [119]. In breast cancer, high levels of Tregs are associated with high tumor grades and low survival rates [120]. Tregs regulate NK cell activation in lymph nodes by blocking the release of GZMB and CD107a from NK cells, thereby promoting the lymphatic metastasis of breast cancer [121]. In tumor-bearing mice, the depletion of Tregs during neoadjuvant immune checkpoint blockade (ICB) not only reshapes the intratumoral immune landscape to favor an ICB response but also induces profound and sustained changes in systemic immunity, which are characterized by increased CD8+ T and NK cells and sustained T cell activation after treatment cessation [122]. Although NK cells can inhibit the dissemination of breast cancer cells in the lungs, chemokine (C-C motif) receptor (CCR)-4-positive Tregs utilize β-galactosidase-binding protein to inhibit NK cells, thereby promoting lung metastasis [123]. Steroid receptor coactivator 3 (SRC-3) is the most highly expressed transcriptional coactivator in Tregs [124]. Breast cancer mice with Treg-specific SRC-3 knockout exhibit high resistance to tumors because SRC-3 knockout Tregs infiltrate breast tumors by activating the C-C motif chemokine ligand (CCL)-19/CCL21/CCR7 signaling axis and promote the entry and function of effector T and NK cells by enhancing the IFN-γ/CXCL9 signaling axis [125]. Studies using SI-2, a specific small-molecule inhibitor of SRC-3, also found that in breast cancer, SI-2 not only reduces the number of Tregs but also effectively increases the number of CD4+ T, CD8+ T, and CD56+ NK cells, as well as the secretion of IFN-γ [126].
MDSCs
MDSCs are a heterogeneous population of immature myeloid cells that suppress innate and adaptive immunity [127]. They can promote the stemness of TNBC and interact with NK cells [128,129], playing an important role in promoting tumor immune evasion and inducing an immunosuppressive TME [130]. The coculture of MDSCs with NK cells impairs the cytotoxicity of NK cells, and this effect is primarily mediated by the reduction in NKG2D ligand expression. Andrew Stiff found that MDSCs derived from patients with cancer considerably inhibit NK cell FcR-mediated functions, including antibody-dependent cellular cytotoxicity and IFN-γ production, in a contact-independent manner. The mechanism underlying this effect may partially involve MDSCs producing nitric oxide. In vivo, the nonspecific depletion of MDSCs or inhibition of inducible nitric oxide synthase substantially enhances the efficacy of mAb treatment in breast cancer mouse models [131]. Researchers employed listeria-based immunotherapy to inhibit MDSCs, resulting in a decrease in the numbers of MDSCs in blood and primary tumors. They observed remarkable improvements in T cells and NK cell function, accompanied with a considerable reduction in tumor growth and metastasis in young and old mice [132].
Tumor-associated macrophages
Tumor-associated macrophages (TAMs) exhibit high plasticity and are involved in tumor progression, metastasis, and immune suppression [133]. Mass cytometry has shown that TAMs are particularly abundant in breast cancer relative to in adjacent tissues and lack tissue-resident macrophages and classical circulating and proinflammatory monocytes [134]. M2 macrophages derived from the peritoneum and bone marrow, as well as TAMs, remarkably inhibit NK cell activation and induce a CD27lowCD11bhigh-exhausted NK cell phenotype [135]. IL-9 stimulates macrophage proliferation and polarizes them toward a proinflammatory M1 phenotype in an IFN-γ-dependent manner, causing them to release CCL3/4 and CXCL9/10 to recruit antitumor immune cells, including T and NK cells, into the TME [136]. Metastasis-associated macrophages (MAMs) are one of the most abundant immune cell types in the metastatic tumor niche in mouse models of MBC [137,138]. Studies have found that MAMs isolated from lung metastatic tissues of MBC inhibit NK cell-induced tumor cell apoptosis in vitro through a TGF-β-dependent mechanism. The depletion of MAMs increases the percentage of activated (CD69+) and mature (CD11b+CD27-) NK cells at metastatic sites, as well as the number of NCR1+ NK cells. Furthermore, MAM depletion or treatment with a TGF-β receptor antagonist substantially enhances the therapeutic effect of NK cell infusion in inhibiting early metastatic tumor growth [139].
Effects of bioactive and signaling molecules on NK cells
Prostaglandin E2
Cytokines also possess the capability to impede the proliferation and activation of NK cells (Table 1). Malignant breast tumors typically exhibit high levels of cyclooxygenase-2 (COX-2), and prostaglandin E2 (PGE2), the primary COX-2 product in cancer, contributes to the high metastatic capacity of breast tumors [140]. The cellular effects of PGE2 are mediated through a family of G-protein-coupled receptors, such as E prostanoid (EP) 1, EP2, EP3, and EP4, which are highly expressed in mouse mammary tumors [141]. Among these receptors, EP4 appears to have a great role because EP4 agonists inhibit IFN-γ production by NK cells, whereas EP1, EP2, and EP3 agonists are ineffective in inhibiting IFN-γ [142]. The use of frondoside A, an antagonist of EP4, inhibits breast tumor metastasis in an NK-dependent manner [143]. When EP4 antagonists are used, breast cancer metastasis is inhibited; this effect is closely associated with a decrease in the expression of major histocompatibility complex class I antigens in tumor cells, as well as an increase in NK cell recognition [144].
Emerging small-molecule inhibitor-based therapeutic strategies for enhancing NK cell function in breast cancer
| Drugs | Target | Related mechanisms | Effects | Type of study | References |
|---|---|---|---|---|---|
| SL-2 | SRC-3 | Small-molecule inhibitors specific for SRC-3 | Treg number ↓ CD4 + and CD8+ T cells ↑ CD56+ NK cells ↑ IFN-γ secretion ↑ | In vivo experiments | [124] |
| RQ-15986 | PGE2 | Novel small-molecule EP4 antagonist | NK cells are protected from PGE2-mediated immunosuppression Breast cancer metastasis ↓ | In vivo experiments | [166] |
| SSB NMs | TGF-β | Codelivery of the TGF-β inhibitor and selenocysteine | NKG2DL expression on tumor cells ↑ NKG2D expression ↑ NK cell sensitivity of MDA-MB-231 cells ↑ | In vitro experiment | [167] |
| ELK3 depletion | Mid51 /CXCL16 | Transcriptional repressor of Mid51, regulates mitochondrial dynamics and ROS accumulation | Susceptibility of TNBC to NK cells ↑ NK cell cytotoxicity ↑ | In vivo and vitro experiments | [168] |
| CIP therapy | STING | Intratumoral STING agonist therapy | Antitumor T cell responses ↑ Granzyme on NK cells ↑ IFNAR-1 and CD25 on lung NK cells ↑ Tumor metastases ↑ | In vivo and vitro experiments | [169] |
| STING/RGD-(KLAKLAK) 2 | STING /KLAK /LAK | Combination of a STING agonist with antivascular RGD-(KLAKLAK) 2 | Antitumor efficacy ↑ Activated NK cells ↑ | In vivo experiment | [170] |
Note: ↓ indicates inhibition/reduction, whereas ↑ indicates increase/promotion.
TGF-β
In the TME, TGF-β not only promotes angiogenic and epithelial-mesenchymal transition (EMT) malignant phenotypes [145], it also serves as a crucial immunosuppressive factor that inhibits the tumor infiltration of immune cells, including the suppression of NK cell activity [146]. Compared with those from healthy control individuals, freshly isolated PBMCs from patients with breast cancer contain more circulating NK cells expressing intracellular IL-10 and TGF-β [147]. In patients with MBC, IFN-γ production by pNK cells is impaired and accompanied with the decreased expression of TNF-related apoptosis-inducing ligand and reduced cytotoxicity against K562 tumor cells. Yue Zhao found that blocking the TGF-β signaling pathway in TNBC cells can enhance the antitumor activity of adoptive NK cells in vitro [148]. Notably, when TGF-β is neutralized in NK cells, the functional impairments of NK cells and metabolic disturbances, such as oxidative phosphorylation, are substantially ameliorated, highlighting TGF-β as a crucial mechanism underlying NK cell dysfunction in cancer [149].
Cytokines
Cytokines communicate with the TME through endocrine, paracrine, or autocrine modes [150,151]. In human and mouse breast cancer tumors, IL20RA is highly expressed, which activates the Janus kinase 1/signal transducer and activator of transcription 3/SRY-box containing gene 2 signaling pathway, thus leading to the increased expression of PD-L1 in tumors and decreased recruitment of anticancer lymphocytes, including CD8+ T and NK cells [152].
The nonclassical histocompatibility antigen HLA-G expressed by breast cancer cells binds to the NK cell receptor KIR2DL4 and, in the presence of cytokines, such as TGF-β and IFN-γ, impairs NK cell cytotoxicity by upregulating PD-L1 on tumor cells and PD-1 on NK cells, making breast cancer cells resistant to trastuzumab [153,154]. Type I IFNs signal canonically by binding to their homologous receptor complex IFNAR1:IFNAR2, triggering a cascade that results in the expression of thousands of IFN-regulated genes controlling NK cell states. The cytotoxic function of NK cells is affected by the absence of IFNAR expression [155]. The loss of host type I IFN signaling accelerates metastasis and impairs the antitumor function of NK cells in multiple syngeneic preclinical breast cancer models lacking a functional type I interferon receptor (IFNAR 1-/- mice) [156]. In breast and lung cancer cells, a T helper cell-derived IL-22 subset drives the expression of CD155. Excess CD155 promotes the internalization of CD226 in NK cells, rendering them inert and promoting metastasis [157]. IL-21 remarkably enhances IFN-γ secretion from NK cells when cocultured with trastuzumab-coated human breast cancer cells in vitro and enhances the antitumor effect of anti-HER2 monoclonal antibodies in a mouse tumor model [158].
Chemokines
Chemokines are chemotactic cytokines that regulate the migration of immune cells and are crucial in directing the migration of immune cells to establish and subsequently provide an effective antitumor immune response [159,160]. CXCL9 plays an important role in antitumor immunity through the recruitment, proliferation, and activation of immune cells. Cao X analyzed TNBC datasets from The Cancer Genome Atlas and found that CXCL9 is expressed at higher levels in immune cells than in tumor cells and that XCL9 mRNA expression is strongly and positively correlated with immune scores, including NK cells [161]. The chemokine CXCL12 secreted by hepatic stellate cells suppresses the proliferation of NK cells through its homologous receptor CXCR4, promoting the liver metastasis of breast cancer caused by disseminated tumor cells [162].
The overexpression of CX3CL1 in HER2-positive breast cancer could increase NK cell-mediated cytotoxicity in vitro and synergizes with trastuzumab to inhibit tumor growth [163]. A retrospective cohort study on 72 patients with TNBC found that the high expression of CCL5 in tumor tissues is associated with the recruitment of CD8+ T cells, NK cells, and M1 macrophages [164]. Serine/threonine/tyrosine kinase 1 (STYK1) is a receptor protein-tyrosine kinase-like molecule that is primarily expressed in NK cells. Studies have found that knocking out STYK1 reduces CCR2 expression, inhibits the accumulation of NK cells in the TME, and promotes spontaneous breast tumor progression [165].
Emerging therapeutic strategies targeting the TME
Small-molecule inhibitor-based therapies
Emerging therapeutic strategies targeting the TME represent a frontier in cancer treatment, offering new hope for enhancing NK cell activity (Figure 6). Among these emerging strategies, therapies based on small-molecule inhibitors are rapidly gaining prominence as a vital research focus (Table 1). In a mouse model of MBC, the novel small-molecule EP4 antagonist (RQ-15986) effectively blocks the immunosuppressive effects of PGE2 on NK cells, thereby enhancing NK cell function and inhibiting tumor growth and metastasis [166]. Chang Liu developed a nanoemulsion system (SSB NMs) for the codelivery of a TGF-β inhibitor and selenocysteine. SSB NMs effectively inhibit TGF-β/TGF-β RI/Smad 2/3 signaling. This effect leads to the enhanced expression of NKG2DL on tumor cells and stimulates the surface expression of NKG2D on NK92 cells. This effect renders MDA-MB-231 cells sensitive to NK cells derived from seven patients with TNBC, resulting in a 13.8-fold increase in cancer lysis rates [167].
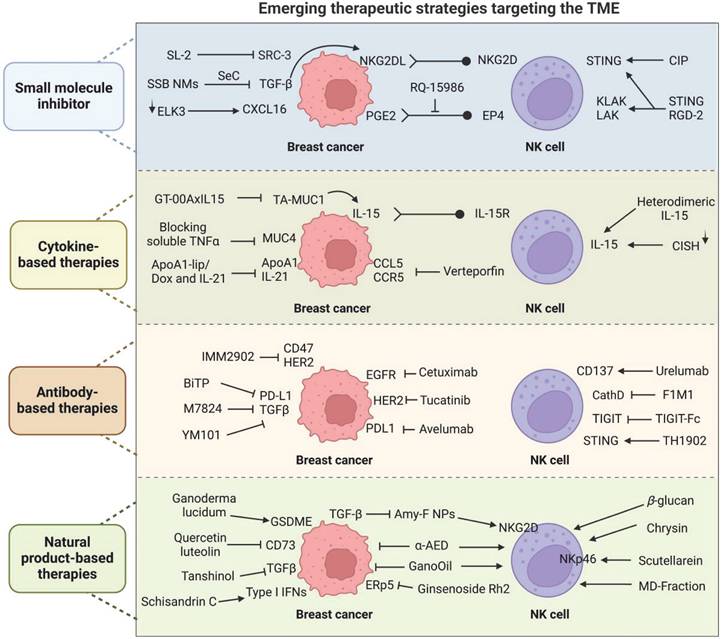
Emerging therapeutic strategies for enhancing NK cell function. We summarize four therapeutic strategies using small-molecule inhibitors, cellular therapies, antibody therapies, and natural products to improve NK cell function in breast cancer.
E-twenty-six-like protein-1/TCF12-related protein 3 (ELK3) serves as a transcriptional repressor of Mid51, thereby regulating mitochondrial dynamics and ROS accumulation. Previous studies have demonstrated that the suppression of ELK3 reinstates the susceptibility of TNBC cells to NK cells. Moreover, ELK3 depletion promotes NK cell cytotoxicity through CXCL16-mediated NK cell recruitment in TNBC [168].
The combination therapy of a stimulator of interferon gene (STING) agonist with extended half-life IL-2 and anti-PD-1 (CIP therapy) considerably enhances the cancer response in TNBC models by upregulating the expression of IFNAR-1 and CD25 on lung NK cells [169]. Justyna Czapla proposed that combining STING agonists with antiangiogenic agents can improve the control of the growth of tumors with low STING protein levels. Their team investigated a novel antitumor therapy that combines a STING agonist with the antivascular agent RGD-(KLAKLAK) 2, finding that it promotes NK cell activation and inhibits the growth of mouse breast cancer [170].
Cytokine-based therapies
Strategies for enhancing the function of NK cells in the treatment of breast cancer often rely on cytokine-based therapies. These strategies hold promise for improving the effectiveness of breast cancer treatment and may lead to the development of personalized and targeted therapies for patients (Table 2). GT-00AxIL15 can promote NK cell proliferation; tumor immune infiltration; ADCC; and lytic capacity against the breast cancer cell lines ZR-75-1, MCF-7, and T-47D by targeting the tumor-associated MUC1 epitope [171]. Vasiliki Stravokefalou discovered that heterodimeric IL-15 (hetIL-15), administered either as a monotherapy or in conjunction with chemotherapy and surgery, markedly decreases metastasis and enhances survival rates in a mouse model of TNBC. This treatment modulates the immune system by decreasing polymorphonuclear MDSCs and concurrently increasing the abundance and activation of effector cells, including CD8+ T and NK cells, thereby demonstrating the potent antimetastatic capabilities of hetIL-15 and hinting at its potential application in TNBC therapy [172].
Emerging cytokine-based therapeutic strategies for enhancing NK cell function in breast cancer
| Drugs | Target | Related mechanisms | Effects | Type of study | References |
|---|---|---|---|---|---|
| GT-00AxIL15 | Tumor-associated MUC1 | Novel IL-15-based immune-cytokine | NK cell proliferation ↑ Tumor immune infiltration ↑ ADCC ↑ | In vitro and vivo experiments | [171] |
| Heterodimeric IL-15 | IL-15 | / | CD8+ T and NK cells ↑ Polymorphonuclear MDSCs systemically ↓ | In vivo experiments | [172] |
| CISH | Il15 | Optimization of NK cell killing properties and reduction in TIGIT immune checkpoint receptor expression | NK cell killing properties ↑ TIGIT expression ↓ NK cell accumulation in the primary tumor ↑ NK cell immunity to metastasis ↑ | In vitro and vivo experiments | [174] |
| Verteporfin | CCL5 /CCR5 | Antagonist of the CCL5/CCR5 axis | Infiltration of NK and CD8+ T cells into TNBC tumors ↑ | In vivo experiments | [175, 176] |
| IL-6 receptor | IL-6 | Positive feedback interconnections of the MCT-1/IL-6/IL-6R/CXCL7/PD-L1 axis in TNBC cells | NK cell function ↑ Recurrence, metastasis, and mortality ↓ | In vivo experiments | [177] |
| ApoA1-lip/Dox and IL-21 | ApoA1/ IL-21 | / | Cytotoxicity of CD8+ T and NK cells ↑ Antitumor efficacy ↑ | In vitro and vivo experiments | [178] |
| Blocking soluble TNFα | MUC4 | Downregulates the expression of MUC4 | Macrophage polarization to the M1-like phenotype ↑ NK cell degranulation ↑ Sensitization of HER2-positive breast cancer to trastuzumab ↑ | In vivo experiments | [179] |
Note: ↓ indicates inhibition/reduction, whereas ↑ indicates increase/promotion.
Cytokine-inducible SH2-containing protein (CISH) is a member of the suppressor of cytokine signaling family of E3 ligases and plays a pivotal role in innate and adaptive immune responses by mediating the negative feedback inhibition of cytokine signal transduction [173]. The deletion of CISH enhances the killing properties of NK cells and reduces the expression of the T cell immune receptor with the Ig and ITIM domain (TIGIT) immune checkpoint receptor, thereby augmenting the suppression of primary tumors and metastases by NK cells [174].
The CCL5/CCR5 chemokine axis has a considerable effect on the growth of tumors, drug resistance, and the functional status of immune cells within the TME [175]. Through high-throughput screening, verteporfin has been identified as a potent antagonist of the CCL5/CCR5 axis, demonstrating the ability to inhibit tumor growth in TNBC through immunomodulation. Mechanistically, verteporfin modulates the secretion of chemokines, such as CXCL16 and CXCL8, by inhibiting the transcription factors HIF-1α and YAP1, thereby regulating the migration of immune cells, including CD8+ T and NK cells [176].
Targeting the IL-6 receptor has demonstrated promising outcomes in enhancing NK cell function within the lymphatic system [177]. This targeted approach has been proven to be effective in reducing recurrence, metastasis, and mortality rates following surgery for TNBC. The innovative combination of ApoA1-lip/Dox with long-acting IL-21 enhances antitumor response and reduces toxicity, showing improved efficacy and safety in TNBC treatment [178].
Bruni S found that MUC4 expression, induced by TNF-α, hinders the effectiveness of trastuzumab in treating HER2-positive breast cancer by promoting immune evasion. By blocking TNF-α with a specific inhibitor in combination with trastuzumab, the antitumor effect is restored through modulation of the immunosuppressive tumor microenvironment. This approach promotes M1-like macrophage polarization, enhances NK cell degranulation, and increases trastuzumab-dependent cellular phagocytosis [179].
Antibody-based therapies
Strategies for enhancing the function of NK cells in the treatment of breast cancer are increasingly focusing on the use of antibodies (Table 3). They involve targeting specific antigens on cancer cells with antibodies that can activate or recruit NK cells to tumor sites. IMM2902, a bispecific antibody targeting CD47 and HER2, promotes the infiltration of T and NK cells into tumors by inducing the release of CXCL9 and CXCL10. It also enhances trastuzumab-induced ADCC and antibody-dependent cellular phagocytosis (ADCP) [180]. Polina Kaidun developed bispecific fusion proteins (BFPs) targeting NKG2D ligands on TNBC cells, which effectively activate NK and T cells, leading to tumor cell lysis. These BFPs could be a promising therapeutic option for patients with TNBC [16].
Emerging antibody-based therapeutic strategies for enhancing NK cell functions in breast cancer
| Drugs | Target | Related mechanisms | Effects | Type of study | References |
|---|---|---|---|---|---|
| IMM2902 | CD47 /HER2 | A bispecific antibody against CD47/HER2 | Release of CXCL9 and CXCL10 ↑ ADCC and ADCP triggered by trastuzumab ↑ | In vivo and vitro experiments | [180] |
| BFPs consisting of the NKG2D receptor | NKG2D/CD3 /CD16 | BFPs fused to either anti-CD3 (NKG2D-CD3) or anti-CD16 (NKG2D-CD16) Fab fragments | NK and T cell activity ↑ | In vitro experiment | [16] |
| urelumab | CD137 | CD137 costimulation counteracts the TGFβ inhibition of NK cell antitumor function | NK cell infiltration in tumors ↑ | In vivo and vitro experiments | [181] |
| M7824 | PD-L1 /TGFβ | A novel bifunctional anti-PD-L1/TGFβ Trap fusion protein | CD8+ T and NK cell activation ↑ OS ↑ | In vivo experiment | [182] |
| BiTP | PD-L1 /TGFβ | A bispecific antibody blocking TGF-β and murine PD-L1 based on the Check-BODY platform | TIL, CD8+ T, and NK cell activation ↑ Total DC and mature (CD80+/CD86+) DC ↑ Antitumor immunity ↑ | In vivo experiment | [183] |
| YM101 and MSA-2 | TGF-β/PD-L1/STING | TGF-β/PD-L1 bispecific antibody/STING agonist | TIL, CD8+ T, and NK cell activation ↑ Total DC and mature (CD80+/CD86+) DC ↑ Antitumor immunity ↑ | In vivo experiment | [184] |
| YM101 and Maraviroc | TGF-β/PD-L1/CCR5 | TGF-β/PD-L1 bispecific antibody/CCR5 antagonist | TIL, CD8+ T, and NK cell activation ↑ Antitumor immunity ↑ | In vivo experiment | [185] |
| Avelumab | PD-L1 | An IgG1 anti-PD-L1 immune checkpoint inhibitor | NK cell-mediated cytotoxicity against TNBC cells ↑ Cytokine production and degranulation ↑ Cleavage activity of NK on tumor cells ↑ | In vitro experiment | [186] |
| NHS-muIL12 and Avelumab | IL2/PDL1 | Combination therapy | Antitumor efficacy ↑ Long-term protective immunity ↑ Proliferation of NK and CD8+ T cells ↑ | In vivo experiment | [187] |
| Tucatinib | HER2 | Selective anti-HER2 tyrosine kinase inhibitor | Frequency of CD8+PD-1+ and CD8+TIM3+ T cells and CD49+ NK cells ↑ Monocytes and expression of major histocompatibility complex II on DCs and macrophages ↑ MDSCs ↓ | In vivo experiment | [188] |
| TIGIT-Fc | TIGIT | Blocking TIGIT | ADCC ↑ Effector functions of CD8 T and NK cells ↑ | In vivo and vitro experiments | [191] |
| TH1902 | STING | A peptide-drug conjugate internalized through a sortilin-mediated process | PRF, GZMB, and caspase-3 staining ↑ Elevated cytotoxic T and NK cells ↑ Tumor growth ↓ Median animal survival ↓ | In vivo and vitro experiments | [192] |
| Cetuximab and IL-15 | EGFR/IL-15 | IgG1 mAb against EGFR | NK and DC activation in vitro ↑ IFN-γ and TNF-α production by NK cells cocultured with DCs ↑ | In vitro experiment | [193] |
| F1M1 | CathD | The CathD antibody F1 and its modified Fab-unglycosylated version | Recruitment of M2-TAMs ↓ Antitumor immunity of NK cells ↑ | In vitro experiment | [195] |
Note: ↓ indicates inhibition/reduction, whereas ↑ indicates increase/promotion.
The anti-CD137 agonist urelumab could prevent the TGF-β inhibition of CD16-induced NK cell proliferation and alters the TGF-β-induced differentiation of activated NK cells [181]. M7824, a new bifunctional checkpoint inhibitor, targets TGF-β and PD-L1/PD-1 pathways to reduce tumor immune evasion and promote antitumor responses in mouse models of breast and colon cancers. M7824 treatment decreases tumor burden, increases survival, and activates CD8+ T and NK cells, showing promise as a monotherapy or in combination with other immunotherapies [182]. Researchers have developed BsAb (BiTP) targeting TGF-β and PD-L1 by using the Check-BODY platform and found that BiTP remarkably increases NK cell proliferation and cytotoxicity in the 4T1 mouse tumor model group [183]. MSA-2 is an oral nonnucleotide STING agonist. MSA-2, in combination with YM101, a bispecific antibody targeting TGF-β and PD-L1, inhibits tumor growth in EMT-6 mice and increases CD3-CD49b+, Ki67+, IFN-γ+, GZMB+, and TNF-α+ NK ratios [184]. Encouragingly, the combination of YM101 with maraviroc, a CCR5 antagonist, also inhibits breast cancer growth and promotes the infiltration of tumor immune cells, suggesting that YM101 is a promising therapeutic approach [185].
Avelumab, a human IgG1 mAb against PD-L1, remarkably enhances NK cell-mediated cytotoxicity against TNBC cells in vitro and triggers cytokine production and degranulation in NK cells [186]. The combination of NHS-muIL12 and avelumab enhances anti-TNBC tumor efficacy and induces the generation of tumor-specific immune memory, augmenting the proliferation of cytotoxic NK and CD8+ T cells, potentially representing a promising approach for the treatment of patients with solid tumors [187]. In the treatment of HER2-positive breast cancer, tucatinib, an effective selective HER2 tyrosine kinase inhibitor, increases the frequency of CD8+PD-1+ and CD8+TIM3+ T cells and CD49+ NK cells and synergizes with PD-1/PD-L1 inhibitors [188].
TIGIT, an inhibitory receptor expressed on lymphocytes, is expressed on T and NK cells and binds with high affinity to the inhibitory ligand CD155, downregulating T and NK cell functions [189, 190]. Xian Shen fused the extracellular domain of TIGIT with the Ig Fc domain (TIGIT-Fc) and found that it could coordinately reverse NK cell exhaustion and accelerate T cell activation to control tumors [191]. Sudocetaxel zendusortide (TH1902), a peptide-drug conjugate internalized through a sortilin-mediated process, increases the activity of cytotoxic T and NK cells, as evidenced by PRF, GZMB, and caspase-3 staining after TH1902 treatment. Combination therapy with TH1902 and anti-PD-L1 results in tumor growth inhibition and increased median animal survival [192].
After cetuximab treatment, activated NK cells in TNBC promote the uptake of tumor material and maturation of DCs, as well as their ability to produce IL-12. IL-15 can enhance the efficacy of cetuximab in the treatment of TNBC by promoting the activation of NK cells and DCs, suggesting potential for their combined therapy [193]. Cathepsin D (CathD), an adverse prognostic marker overexpressed by breast cancer cells and secreted excessively in the TME, exhibits protumorigenic activity [194]. The team led by Timothée David found that the anti-CathD antibody F1 and its improved Fab-deglycosylated version F1M1 can prevent the recruitment of immunosuppressive M2-polarized TAMs, activate NK cells in the TME, and promote innate antitumor immunity [195].
Natural product-based therapies
Notably, natural products have shown great potential in enhancing the function of NK cells (Table 4). In breast cancer, the androgenic steroid compound androstene-3β,17α-diol could inhibit lung metastasis and increase the percentage of NK and plasma cells; this effect is accompanied with the decreased expression of vascular endothelial growth factor (VEGF) in the TME [196]. In a mouse model of breast cancer lung metastasis, Ganoderma lucidum spore oil boosts NK cell activity in the spleen, increases CD8+ T cell and IL-6 levels in the blood, and inhibits the lung metastasis of circulating breast cancer cells [197]. Quercetin and luteolin have been identified as targeting CD73, which leads to the inhibition of the YAP/Wnt signaling pathway.
Emerging natural product-based therapeutic strategies for enhancing NK cell function in breast cancer
| Drugs | Target | Effects and related mechanisms | Type of study | References |
|---|---|---|---|---|
| Androstene-3β,17α-diol | / | Lung metastasis ↓ Percentage of NK cells in mouse tumors ↑ | In vivo experiment | [196] |
| GanoOil | GanoOil | CD8+ T cells in the blood of mice ↑ NK cell activity in the spleen ↑ | In vivo experiment | [197] |
| Quercetin and luteolin | CD73 | Antitumor immune function of NK and CD8+ T cells ↑ | In vivo and vitro experiments | [198] |
| Schisandrin C | Type I IFNs | Antitumor immunity ↑ Tumor growth ↓ Sensitivity to chemotherapy ↑ | In vivo experiment | [199] |
| Amy-F NPs | TGF-β/INF-γ/IL-2/IL-6/VEGF | CD4 expression ↓ NKG2D expression ↑ | In vitro experiment | [200] |
| G. lucidum extract | Caspase 3/GSDME | Antitumor immune responses ↑ Subsets of NK and CD8+ T cells in the peripheral immune system ↑ | In vivo experiment | [201] |
| Scutellarein | / | Proliferative capacity of NK cells ↑ NKp46 expression ↑ | In vitro experiment | [202] |
| Tanshinol | TGF-β1 | NKG2D and DAP10 expression levels ↑ NK cell activation ↑ Granule release from NK cells ↑ | In vivo experiment | [203] |
| Chrysin | / | Cytotoxicity of NK-92 and T cells toward MCF-7 and MDA-MB-231 cells ↑ Granzyme-B, IL-2, and IFN-γ production ↑ | In vitro experiment | [204] |
| Ginsenoside Rh2 | ERp5 | PRF, GZMB, and IFN-γ release ↑ Breast cancer growth and metastasis ↓ | In vivo and vitro experiments | [205] |
| β-glucan | MD-Fraction | ADCC, complement-dependent cellular cytotoxicity, and complement-dependent cytotoxicity ↑ Anti-HER2+ breast cancer tumor effects ↑ | In vivo experiment | [206] |
| XK-81 | / | CD8+ T and NK cell numbers ↑ | In vivo experiment | [207] |
Note: ↓ indicates inhibition/reduction, whereas ↑ indicates increase/promotion.
This inhibition ultimately results in the suppression of the enrichment of mesenchymal- and epithelial-like cancer stem cells. Furthermore, these compounds have been found to enhance the antitumor immune response mediated by NK and CD8+ T cells, thus inhibiting the proliferation of TNBC [198]. In TNBC, schisandrin C reduces tumor growth by enhancing the response of type I IFNs in a cGAS/STING pathway-dependent manner, which guides the infiltration of T and NK cells into the tumor site [199].
Amygdalin, a naturally occurring compound that is also known as vitamin B17, enhances the targeting efficiency of cancer treatment due to its preferential binding to folate receptor alpha, which is overexpressed on cancer cells. Mostafa A. Askar developed amygdalin-folic acid nanoparticles (Amy-F NPs) for the targeted therapy of breast cancer. Their results showed that Amy-F NPs could downregulate CD4 expression, synergize with the CD8-mediated suppression of CD80, and activate NKG2D expression in human breast cancer cells [200]. G. lucidum extract remarkably enhances antitumor immune responses by boosting the subsets of NK and CD8+ T cells in the peripheral immune system and TME, highlighting its potent immunomodulatory effects in breast cancer treatment [201]. The TCM formula Xianling Lianxia, which is based on the therapeutic principle of “strengthening the body and eliminating pathogens,” enhances trastuzumab-mediated ADCC against HER2-positive breast cancer. Among the components of this formula, scutellarein can increase the proliferative capacity of NK cells and expression of NKp46 [202].
TGF-β1 facilitates the lung metastasis of breast cancer while inhibiting NK cell cytotoxicity. However, tanshinol reverses these effects, enhancing mouse survival and optimizing NK cell function by restoring the NKG2D/NKG2DL axis and PI3K/ERK1/2-PLCγ2 signaling pathway, leading to increased tumor cell immune attacks [203]. Chrysin, a natural flavonoid, enhances the activation and cytotoxicity of NK-92 and T cells against breast cancer cells, particularly MCF-7, by increasing cytokine production and GZMB levels [204]. Ginsenoside Rh2 substantially inhibits breast cancer growth and metastasis by enhancing NK cell cytotoxicity through direct binding to ERp5, thus modulating the NKG2D/MICA signaling axis, and holds potential as a therapeutic agent for breast cancer [205]. Researchers have explored the synergistic effect of β-glucan (named MD-Fraction) from Grifola frondosa with trastuzumab on HER2-positive breast cancer. MD-Fraction combined with trastuzumab could enhance antitumor efficacy by augmenting ADCC, complement-dependent cellular cytotoxicity (CDCC), and complement-dependent cytotoxicity (CDC), suggesting a promising strategy for improving the benefits of trastuzumab in HER2-positive breast cancer [206]. XK-81, a novel bromophenol compound derived from Leathesia nana, could increase CD8+ T and NK cell numbers and regulate the M1:M2 macrophage ratio, demonstrating potential antitumor advantage in 4T1 tumor tissues [207].
Collectively, these studies highlight the potential of targeting the TME to enhance NK cell function in breast cancer immunotherapy. However, further preclinical and clinical studies must be conducted to evaluate fully the safety, efficacy, and potential side effects of the intervention or treatment in question.
Preclinical and clinical trials
The safety and efficacy of NK cell therapy have been thoroughly demonstrated [208,209]. With the ongoing efforts to improve therapeutic responses and develop viable novel methods for the clinical-scale generation of NK cells, emerging clinical trials are being designed to evaluate these new modalities and expand their indications (Table 5) [210,211]. Allogeneic NK cells have been investigated in patients with recurrent ovarian and breast cancers following lymphodepleting chemotherapy, with some patients also receiving radiation therapy. Findings demonstrated transient donor chimerism but limited NK cell expansion, underscoring the need for improved strategies to enhance NK cell persistence [212]. A phase I/II study treated 10 patients with cancer with an autologous DC vaccine pulsed with tumor lysate and keyhole limpet hemocyanin+ IL-2. One patient with renal cell carcinoma (RCC) had stable disease (SD), and six patients showed antigen-specific immune responses and remarkable NK activation [213].
Preclinical and clinical trials
| Drugs | Target | Diseases | Cases | Effect | Trial design | Trial No. | References |
|---|---|---|---|---|---|---|---|
| Allogeneic NK cell therapy | / | Recurrent ovarian and breast cancers | 20 | One patient who was not evaluable had successful in vivo NK cell expansion. | I/II | / | [212] |
| DC vaccine and IL-2 | / | RCC or breast cancer | 10 | A clinical response of stabilization was observed in one patient with RCC. NK activity was remarkably induced after vaccination in six patients. | I/II | / | [213] |
| LAG525/spartalizumab | PD-1 /LAG-3 | Advanced/metastatic solid tumors | 255 | The ORR was 10.7 (13/121) in the combination group, with no patients achieving CR or PR in the ieramilimab monotherapy group. The responding population had high levels of gene expression of immune cells, including NK cells. | I/II | NCT02460224 | [214] |
| CpG ODN | TLR9 | HER2-positive MBC | 6 | Three patients (50%) had SD at 12 weeks as their best response. NK cell populations did not change considerably throughout the course of treatment. | Single-arm, open-label phase II clinical trial | NCT00824733 | [215] |
| Monalizumab | NKG2A | HER2-positive MBC | 15 | Although the novel combination was well-tolerated, no clinical responses were observed. | II | NCT04307329 | [216] |
| High intensity exercise | / | Postmenopausal women with overweight status at high risk of breast cancer | 33 | No significant differences for the NK cell function variable were observed within or between groups. | Randomized controlled trial | NCT02923401 | [217] |
Clinical studies aiming to enhance NK cell function through indirect means in addition to direct NK cell therapy to improve the efficacy of tumor immunity also exist. The NCT02460224 clinical trial evaluated the efficacy and safety of monotherapy with the lymphocyte activation gene-3 inhibitor ieramilimab (LAG525) or in combination with the antiprogrammed cell death-1 antibody spartalizumab in patients with advanced solid tumors, including breast cancer. The overall response rate (ORR) in the combination therapy group was found to be 10.7% (13/121), whereas no patient in the ieramilimab monotherapy group was observed to achieve complete response (CR) or partial response (PR). High levels of immune gene expression, including those related to NK cells, were observed in the responding population [214].
Dionisia Quiroga conducted a single-arm, open-label phase II clinical trial on the combination of a toll-like receptor 9 agonist (CpG ODN) with trastuzumab for the treatment of patients with advanced/metastatic HER2-positive breast cancer. Three patients discontinued treatment due to tolerability issues. Among the remaining patients, three (50%) were found to exhibit SD at 12 weeks, but no notable change in NK cell counts were observed during treatment [215]. The MIMOSA trial is a phase II study that investigated the novel combination of monalizumab, an NKG2A checkpoint inhibitor, with trastuzumab in heavily pretreated patients with HER2-positive MBC. Although the treatment was found to be well-tolerated without dose-limiting toxicities, no objective responses were observed. Therefore, the MIMOSA trial did not meet its primary endpoint, indicating that the novel combination of monalizumab and trastuzumab does not induce objective responses in the above patient population [216]. A randomized controlled trial compared the effects of high-intensity interval training and moderate-intensity continuous training on resting NK cell function and circulating myokines in obese women with breast cancer. Although it discovered no notable differences between groups, its results are suggestive of a great effect on NK cells in patients with low cardiorespiratory fitness [217].
Conclusion and future perspectives
NK cells play a critical role in combating breast cancer, and restoring their functionality holds utmost importance in cancer immunotherapy [218]. This article underscores the necessity of acquiring a thorough understanding of the multifaceted factors operating within the TME of breast cancer. It emphasizes the need to comprehend the complex interactions occurring within the TME given that they are pivotal in shaping the overall tumor immune response. By organizing the extensive research conducted by various scholars, we aim to facilitate deep investigations into the regulatory mechanisms that govern NK cell-mediated tumor elimination within the breast cancer TME.
The cytotoxic potency of NK cells relies on the integration of multiple activating and inhibitory receptors [219]. Studies have revealed that knocking out the killer cell lectin-like receptor subfamily C1 (KLRC1) in NK cells boosts their cytotoxic activity against breast cancer cells expressing HLA-E [220]. This research highlights the importance of the KLRC1/HLA-E axis in immunotherapy and suggests that targeting KLRC1 is a potential strategy to enhance the antitumor activity of NK cells.
Moreover, nanotherapies and CAR-NK and adoptive NK cell therapies exist [221,222]. Eun-Jeong Won synthesized artificial nanomaterials that could target TNBC by loading plasmid DNA encoding hypermethylated in cancer 1 (HIC1) into lipid-based nanoparticles (LNPs) and fusing NK cell membrane proteins to the surfaces of the LNPs. Their results demonstrated that NK-LNPs can induce antitumor immune responses and inhibit the metastasis of TNBC [223]. Bolong Tao designed CAR-NK cell-derived exosome-camouflaged nanobombs for the enhanced treatment of HER2-positive breast cancer with brain metastases [224]. The downregulation of intercellular cell adhesion molecule-1 (ICAM-1) on breast cancer cells is a key escape mechanism for trastuzumab-triggered ADCC, and the ability of CAR-NK cells to overcome ICAM-1 reduction-induced cancer cell resistance highlights the potential of CAR-NK cells in cancer immunotherapy [225]. Adoptive cell therapy is a novel immunotherapy approach that involves the extraction, modification, and expansion of immune cells, which are then infused back into the patient [226]. In a phase I trial, autologous NK cell infusion in addition to basic anti-HER2 targeting was found to be well tolerated in patients with refractory HER2-positive solid tumors, with six of 19 subjects stable for ≥6 months and one patient experiencing PR [13].
The ultimate goal behind fostering these research efforts is to drive the advancement of immunotherapy strategies that are increasingly precise and potent. We strive to overcome the immunosuppressive challenges inherent in breast cancer treatment. Through rigorous exploration and experimentation, we aim to develop targeted immunotherapies capable of effectively countering the immunosuppressive effects observed in patients with breast cancer, thereby improving their OS and quality of life.
Abbreviations
ACT: Adoptive Cell Therapy; AHR: Aryl hydrocarbon receptor; ATP: Adenosine Triphosphate; CAFs: Cancer-Associated Fibroblasts; CCL: C-C Motif Chemokine Ligand; CCR: Chemokine (C-C motif) Receptor; CISH: Cytokine-inducible SH2-containing protein; COX-2: Cyclooxygenase-2; CRS: Cytokine Release Syndrome; CX3CL1: C-X3-C Motif Chemokine Ligand 1; CXCL: Chemokine (C-X-C motif) Ligand; CXCL: C-X-C Motif Chemokine Ligand; CXCR4: C-X-C Motif Chemokine Receptor 4; DCs: Dendritic Cells; DFS: Disease-free Survival; DKK1: Dickkopf-1; DPP4: Dipeptidyl Peptidase 4; ECM: Extracellular Matrix; EGFR: Epidermal Growth Factor Receptor; EMT: Epithelial-mesenchymal Transition; ENT: Entinostat; EP: E prostanoid; ERK: Extracellular Regulated Protein Kinases; FcR: Fc Receptor; FTH1: Ferritin Heavy Chain 1; GLUT1/2/3: Glucose Transporter 1/2/3; GVHD: Graft-Versus-Host Disease; GZMB: Granzyme B; HER2: Human Epidermal Growth Factor Receptor 2; HIC1: Hypermethylated in Cancer 1; HIF-1α: Hypoxia-inducible Factor 1 Alpha; HLA: Human Leucocyte Antigen; HNSCC: Head and Neck Squamous Cell Carcinoma; ICAM-1: Intercellular Cell Adhesion Molecule-1; ICD: immunogenic cell death; IDO: Indoleamine 2,3-Dioxygenase; IFNAR1/2: Interferon-α Receptor 1/2; IFN-γ: Interferon Gamma; IgG1: Immunoglobulin G1; IL: Interleukin; iNOS: Inducible Nitric Oxide Synthase; LNPs: Lipid-based Nnanoparticles; JAK1: Janus Kinase 1; KIR2DL4: Killer Cell Immunoglobulin-like Receptor 2DL4; Kyn: L-kynurenine; LDH: Lactate Dehydrogenase; M1/M2: Macrophage 1/2; mAb: Monoclonal Antibody; MAMs: Metastasis-associated Macrophages; matCAFs: Matrix-producing CAFs; MBC: Metastatic Breast Cancer; MCT: Monocarboxylate Transporter; MDSCs: Myeloid-Derived Suppressor Cells; MHC: Major Histocompatibility Complex; MICA/B: MHC Class I Chain-Related Genes A/B; MSCs: Mesenchymal Stem Cells; myCAFs: myofibroblast-type CAFs; NCR1: Natural Cytotoxicity Triggering Receptor 1; NF-κB: Nuclear Factor Kappa-light-chain-enhancer of Activated B Cells; NK: Natural Killer; NKG2D: NK Group 2D; NKp46: Natural Killer Cell Protein 46; OS: Overall Survival; PBMCs: Peripheral Blood Mononuclear Cells; pCR: Pathological Complete Response; PD-1: Programmed Death Protein 1; PD-L1: Programmed Death-Ligand 1; PDPN: Podoplanin; PFS: progression Free Survival; PGC1α: PPARG Coactivator 1 Alpha; PGE2: Prostaglandin E2; PMN-MDSCs: Polymorphonuclear Myeloid-Derived Suppressor Cells; pNK: Peripheral Blood Natural Killer; PPARG: Peroxisome Proliferator-Activated Receptor Gamma; PRF: Perforin; proCAFs: Progenitor cell-type CAFs; ROS: Reactive Oxygen Species; RPTK: Receptor Protein-Tyrosine Kinase; SOCS: Suppressor of Cytokine Signaling; SOX2: SRY-box Containing Gene 2; SRC-3: Steroid Receptor Coactivator 3; STAT: Signal Transducer and Activator of Transcription; sTILs: Stromal Tumor-infiltrating Lymphocytes; STING: Stimulator of Interferon Genes; STYK1: Serine/Threonine/Tyrosine Kinase 1; TAK1: TGF-β-Activated Kinase 1; TAMs: Tumor-Associated Macrophages; TCGA: The Cancer Genome Atlas; TCRs: T Cell Receptors; TDO-2: Tryptophan 2,3-Dioxygenase 2; TGF-β: Transforming Growth Factor Beta; TIGIT: T cell immune receptor with Ig and ITIM domains; TILs: Tumor-infiltrating lymphocytes; TLR7: Toll like receptor 7; TME: Tumor Microenvironment; T-MSCs: Tumor-associated MSCs; TNBC: Triple-negative Breast Cancer; TNFR2: TNF Receptor 2; TNF-α: Tumor Necrosis Factor-alpha; TRAIL: TNF-related apoptosis-inducing ligand; Tregs: Regulatory T Cells; Trp: Tryptophan; YAP1: Yes-associated Protein 1.
Acknowledgements
The Figures were created with BioRender.com.
Funding
This work was supported by the High-level Chinese Medicine Key Discipline Construction Project of National Administration of Traditional Chinese Medicine (Grant No.: zyyzdxk-2023068), National Natural Science Foundation of China funded project (Grant No.: 82405389), the Postdoctoral Fellowship Program of CPSF (Grant No.: GZC20231706), and the Shanghai Post-doctoral Excellence Program (Grant No.: 2023547).
Availability of data and materials
All data relevant to this review are included in the article. Data and materials are available upon reasonable request.
Author contributions
Conceptualization: FF.L., CF.G., Y.H. Writing of manuscript: FF.L., Y.Q., HX.X. Reviewing of manuscript: All authors. Study supervision: HG.W., S.L.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Sung H, Ferlay J, Siegel R.L. et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249
2. Liu T, Ellisen LW. Exploring the “chemo” in chemoimmunotherapy for triple-negative breast cancer. Cancer Cell. 2025;43:332-334
3. Chang Y, Jin G, Luo W. et al. Engineered human pluripotent stem cell-derived natural killer cells with PD-L1 responsive immunological memory for enhanced immunotherapeutic efficacy. Bioact Mater. 2023;27:168-180
4. Verma C, Kaewkangsadan V, Eremin J.M. et al. Natural killer (NK) cell profiles in blood and tumor in women with large and locally advanced breast cancer (LLABC) and their contribution to a pathological complete response (PCR) in the tumor following neoadjuvant chemotherapy (NAC): differential restoration of blood profiles by NAC and surgery. J Transl Med. 2015;13:180
5. Yi J.I, Schneider J, Lim S.T. et al. Interferon-Gamma Secretion Is Significantly Decreased in Stage III Breast Cancer Patients. Int J Mol Sci. 2024;25:4561
6. Kim R, Kawai A, Wakisaka M. et al. A potential role for peripheral natural killer cell activity induced by preoperative chemotherapy in breast cancer patients. Cancer Immunol Immunother. 2019;68:577-585
7. Muraro E, Comaro E, Talamini R. et al. Improved Natural Killer cell activity and retained anti-tumor CD8(+) T cell responses contribute to the induction of a pathological complete response in HER2-positive breast cancer patients undergoing neoadjuvant chemotherapy. J Transl Med. 2015;13:204
8. Zhang C, Wu F, Lu X. et al. Peripheral NK cell count predicts response and prognosis in breast cancer patients underwent neoadjuvant chemotherapy. Front Immunol. 2024;15:1437193
9. Liu Z, Ding M, Qiu P. et al. Natural killer cell-related prognostic risk model predicts prognosis and treatment outcomes in triple-negative breast cancer. Front Immunol. 2023;14:1200282
10. Ayuso J.M, Farooqui M, Virumbrales-Muñoz M. et al. Author Correction: Microphysiological model reveals the promise of memory-like natural killer cell immunotherapy for HIV± cancer. Nat Commun. 2023;14:7292
11. Niu Z, Wu J, Zhao Q. et al. CAR-based immunotherapy for breast cancer: peculiarities, ongoing investigations, and future strategies. Front Immunol. 2024;15:1385571
12. He C, Wang D, Shukla S.K. et al. Vitamin B6 Competition in the Tumor Microenvironment Hampers Antitumor Functions of NK Cells. Cancer Discov. 2024;14:176-193
13. Lee S. C, Shimasaki N, Lim J.S.J, et al. Phase I Trial of Expanded, Activated Autologous NK-cell Infusions with Trastuzumab in Patients with HER2-positive Cancers. Clin Cancer Res. 2020;26:4494-4502
14. Mamessier E, Bourgin C, Olive D. et al. When breast cancer cells start to fend the educational process of NK cells off. Oncoimmunology. 2013;2:e26688
15. Mamessier E, Sylvain A, Thibult M. L, et al. Human breast cancer cells enhance self tolerance by promoting evasion from NK cell antitumor immunity. J Clin Invest. 2011;121:3609-3622
16. Kaidun P, Holzmayer S.J, Greiner S.M. et al. Targeting NKG2DL with Bispecific NKG2D-CD16 and NKG2D-CD3 Fusion Proteins on Triple-Negative Breast Cancer. Int J Mol Sci. 2023;24:13156
17. Herault A, Mak J, de la Cruz-Chuh J. et al. NKG2D-bispecific enhances NK and CD8+ T cell antitumor immunity. Cancer Immunol Immunother. 2024;73:209
18. Gong Y, Germeraad W.T.V, Zhang X. et al. NKG2A genetic deletion promotes human primary NK cell anti-tumor responses better than an anti-NKG2A monoclonal antibody. Mol Ther. 2024;32:2711-2727
19. Xue D, Lu S, Zhang H. et al. Induced pluripotent stem cell-derived engineered T cells, natural killer cells, macrophages, and dendritic cells in immunotherapy. Trends Biotechnol. 2023;41:907-922
20. Krneta T, Gillgrass A, Chew M. et al. The breast tumor microenvironment alters the phenotype and function of natural killer cells. Cell Mol Immunol. 2016;13:628-639
21. Duechler M, Peczek L, Zuk K. et al. The heterogeneous immune microenvironment in breast cancer is affected by hypoxia-related genes. Immunobiology. 2014;219:158-165
22. Gajovic N, Jurisevic M, Pantic J. et al. Attenuation of NK cells facilitates mammary tumor growth in streptozotocin-induced diabetes in mice. Endocr Relat Cancer. 2018;25:493-507
23. Faget J, Biota C, Bachelot T. et al. Early detection of tumor cells by innate immune cells leads to T(reg) recruitment through CCL22 production by tumor cells. Cancer Res. 2011;71:6143-6152
24. Zhang H, Yang L, Wang T. et al. NK cell-based tumor immunotherapy. Bioact Mater. 2024;31:63-86
25. Solocinski K, Padget M.R, Fabian K.P. et al. Overcoming hypoxia-induced functional suppression of NK cells. J Immunother Cancer. 2020;8:e000246
26. Ni J, Wang X, Stojanovic A. et al. Single-Cell RNA Sequencing of Tumor-Infiltrating NK Cells Reveals that Inhibition of Transcription Factor HIF-1α Unleashes NK Cell Activity. Immunity. 2020;52:1075-1087.e8
27. Ma S, Zhao Y, Lee W.C. et al. Hypoxia induces HIF1α-dependent epigenetic vulnerability in triple negative breast cancer to confer immune effector dysfunction and resistance to anti-PD-1 immunotherapy. Nat Commun. 2022;13:4118
28. Viry E, Baginska J, Berchem G. et al. Autophagic degradation of GZMB/granzyme B: a new mechanism of hypoxic tumor cell escape from natural killer cell-mediated lysis. Autophagy. 2014;10:173-175
29. Brown T.P, Ganapathy V. Lactate/GPR81 signaling and proton motive force in cancer: Role in angiogenesis, immune escape, nutrition, and Warburg phenomenon. Pharmacol Ther. 2020;206:107451
30. Elia I, Rowe J.H, Johnson S. et al. Tumor cells dictate anti-tumor immune responses by altering pyruvate utilization and succinate signaling in CD8+ T cells. Cell Metab. 2022;34:1137-1150.e6
31. Harmon C, Robinson M.W, Hand F. et al. Lactate-Mediated Acidification of Tumor Microenvironment Induces Apoptosis of Liver-Resident NK Cells in Colorectal Liver Metastasis. Cancer Immunol Res. 2019;7:335-346
32. Brand A, Singer K, Koehl G.E. et al. LDHA-Associated Lactic Acid Production Blunts Tumor Immunosurveillance by T and NK Cells. Cell Metab. 2016;24:657-671
33. Jedlička M, Feglarová T, Janstová L. et al. Lactate from the tumor microenvironment - A key obstacle in NK cell-based immunotherapies. Front Immunol. 2022;13:932055
34. Luo Z, Huang X, Xu X. et al. Decreased LDHB expression in breast tumor cells causes NK cell activation and promotes tumor progression. Cancer Biol Med. 2024;21:513-540
35. Long Y, Gao Z, Hu X. et al. Downregulation of MCT4 for lactate exchange promotes the cytotoxicity of NK cells in breast carcinoma. Cancer Med. 2018;7:4690-4700
36. Borde S, Matosevic S. Metabolic adaptation of NK cell activity and behavior in tumors: challenges and therapeutic opportunities. Trends Pharmacol Sci. 2023;44:832-848
37. Gardiner C.M. NK cell metabolism. J Leukoc Biol. 2019;105:1235-1242
38. Picard L.K, Niemann J.A, Littwitz-Salomon E. et al. Restriction of Glycolysis Increases Serial Killing Capacity of Natural Killer Cells. Int J Mol Sci. 2024;25:2917
39. Khan A.U.H, Ali A.K, Marr B. et al. The TNFα/TNFR2 axis mediates natural killer cell proliferation by promoting aerobic glycolysis. Cell Mol Immunol. 2023;20:1140-1155
40. Park J.D, Kim K.S, Choi S.H. et al. ELK3 modulates the antitumor efficacy of natural killer cells against triple negative breast cancer by regulating mitochondrial dynamics. J Immunother Cancer. 2022;10:e004825
41. Guo X, Tan S, Wang T. et al. NAD+ salvage governs mitochondrial metabolism, invigorating natural killer cell antitumor immunity. Hepatology. 2023;78:468-485
42. Zheng X, Qian Y, Fu B. et al. Mitochondrial fragmentation limits NK cell-based tumor immunosurveillance. Nat Immunol. 2019;20:1656-1667
43. Reynolds JC, Lai RW, Woodhead JST. et al. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nat Commun. 2021;12:470
44. Cho E, Stampley J, Wall R. et al. Acute Exercise Increases NK Cell Mitochondrial Respiration and Cytotoxicity against Triple-Negative Breast Cancer Cells under Hypoxic Conditions. Med Sci Sports Exerc. 2023;55:2132-2142
45. Westhaver LP, Nersesian S, Nelson A. et al. Mitochondrial damage-associated molecular patterns trigger arginase-dependent lymphocyte immunoregulation. Cell Rep. 2022;39:110847
46. Zhang C, Wang K, Wang H. Adenosine in cancer immunotherapy: Taking off on a new plane. Biochim Biophys Acta Rev Cancer. 2023;1878:189005
47. Gong X, Zheng C, Cai Y. et al. Adenosine-modulating synthetic high-density lipoprotein for chemoimmunotherapy of triple-negative breast cancer. J Control Release. 2024;367:637-648
48. Tay AHM, Prieto-Díaz R, Neo S. et al. A2B adenosine receptor antagonists rescue lymphocyte activity in adenosine-producing patient-derived cancer models. J Immunother Cancer. 2022;10:e004592
49. Su C, Zhang P, Liu J. et al. Erianin inhibits indoleamine 2, 3-dioxygenase -induced tumor angiogenesis. Biomed Pharmacother. 2017;88:521-528
50. Jing R, Bai S, Zhang P. et al. IDO-1 impairs antitumor immunity of natural killer cells in triple-negative breast cancer via up-regulation of HLA-G. Breast Cancer. 2024;31:135-147.43
51. Pal A, Schneider J, Schlüter K. et al. Different endurance exercises modulate NK cell cytotoxic and inhibiting receptors. Eur J Appl Physiol. 2021;121:3379-3387
52. Zhang X.Y, Shi J.B, Jin S.F. et al. Metabolic landscape of head and neck squamous cell carcinoma informs a novel kynurenine/Siglec-15 axis in immune escape. Cancer Commun (Lond). 2024;44:670-694
53. Damerell V, Klaassen-Dekker N, Brezina S. et al. Circulating tryptophan-kynurenine pathway metabolites are associated with all-cause mortality among patients with stage I-III colorectal cancer. Int J Cancer. 2025;156:552-565
54. Liu Y, Chen S, Wan X. et al. Tryptophan 2,3-dioxygenase-positive matrix fibroblasts fuel breast cancer lung metastasis via kynurenine-mediated ferroptosis resistance of metastatic cells and T cell dysfunction. Cancer Commun (Lond). 2024;44:1261-1286
55. Cui J.X, Xu X.H, He T. et al. L-kynurenine induces NK cell loss in gastric cancer microenvironment via promoting ferroptosis. J Exp Clin Cancer Res. 2023;42:52
56. Della Chiesa M, Carlomagno S, Frumento G. et al. The tryptophan catabolite L-kynurenine inhibits the surface expression of NKp46- and NKG2D-activating receptors and regulates NK-cell function. Blood. 2006;108:4118-4125
57. Rock K.L, Kataoka H, Lai J.J. Uric acid as a danger signal in gout and its comorbidities. Nat Rev Rheumatol. 2013;9:13-23
58. Wang J, Liu K, Xiao T. et al. Uric acid accumulation in DNA-damaged tumor cells induces NKG2D ligand expression and antitumor immunity by activating TGF-β-activated kinase 1. Oncoimmunology. 2022;11:2016159
59. Ma H, Wang X, Zheng X. et al. Sphingomyelin is a prospective metabolic immune checkpoint for natural killer cells. Clin Transl Med. 2023;13:e1395
60. Zheng X, Hou Z, Qian Y. et al. Tumors evade immune cytotoxicity by altering the surface topology of NK cells. Nat Immunol. 2023;24:802-813
61. Biffi G. and Tuveson D.A. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol Rev. 2021;101:147-176
62. Costa A, Kieffer Y, Scholer-Dahirel A. et al. Fibroblast Heterogeneity and Immunosuppressive Environment in Human Breast Cancer. Cancer Cell. 2018;33:463-479.e10
63. Sidaway P. Breast cancer: Fibroblast subtypes alter the microenvironment. Nat Rev Clin Oncol. 2018;15:264-265
64. Zhan Y, Sun D, Gao J. et al. Single-cell transcriptomics reveals intratumor heterogeneity and the potential roles of cancer stem cells and myCAFs in colorectal cancer liver metastasis and recurrence. Cancer Lett. 2025;612:217452
65. Houthuijzen JM, de Bruijn R, van der Burg E. et al. CD26-negative and CD26-positive tissue-resident fibroblasts contribute to functionally distinct CAF subpopulations in breast cancer. Nat Commun. 2023;14:183
66. Ye J, Baer J.M, Faget D.V. et al. Senescent CAFs Mediate Immunosuppression and Drive Breast Cancer Progression. Cancer Discov. 2024;14:1302-1323
67. Croizer H, Mhaidly R, Kieffer Y. et al. Deciphering the spatial landscape and plasticity of immunosuppressive fibroblasts in breast cancer. Nat Commun. 2024;15:2806
68. Zhao Z, Sun H, Liu Y. et al. PDPN+ cancer-associated fibroblasts enhance gastric cancer angiogenesis via AKT/NF-κB activation and the CCL2-ACKR1 axis. MedComm (2020). 2025;6:e70037
69. Yu D, Xu H, Zhou J, Fang K, Zhao Z, Xu K. PDPN/CCL2/STAT3 feedback loop alter CAF heterogeneity to promote angiogenesis in colorectal cancer. Angiogenesis. 2024;27:809-825
70. Tanaka Y, Ohno T, Kadonaga T. et al. Podoplanin expression in cancer-associated fibroblasts predicts unfavorable prognosis in node-negative breast cancer patients with hormone receptor-positive/HER2 - negative subtype. Breast Cancer. 2021;28:822-828
71. Du R, Zhang X, Lu X. et al. PDPN positive CAFs contribute to HER2 positive breast cancer resistance to trastuzumab by inhibiting antibody-dependent NK cell-mediated cytotoxicity. Drug Resist Updat. 2023;68:100947
72. Rivas EI, Linares J, Zwick M. et al. Targeted immunotherapy against distinct cancer-associated fibroblasts overcomes treatment resistance in refractory HER2+ breast tumors. Nat Commun. 2022;13:5310
73. Tanaka T, Suzuki H, Ohishi T. et al. A Cancer-Specific Anti-Podoplanin Monoclonal Antibody, PMab-117-mG2a Exerts Antitumor Activities in Human Tumor Xenograft Models. Cells. 2024;13:1833
74. Feng C, Yu A, Wang Z. et al. A novel PDPN antagonist peptide CY12-RP2 inhibits melanoma growth via Wnt/β-catenin and modulates the immune cells. J Exp Clin Cancer Res. 2024;43:9
75. Kasoha M, Bohle RM, Seibold A. et al. Dickkopf-1 (Dkk1) protein expression in breast cancer with special reference to bone metastases. Clin Exp Metastasis. 2018;35:763-775
76. Shi HX, Tao HT, He JJ. et al. Targeting DKK1 enhances the antitumor activity of paclitaxel and alleviates chemotherapy-induced peripheral neuropathy in breast cancer. Mol Cancer. 2024;23:152
77. Faccio R, Lee S, Ricci B. et al. Cancer-associated fibroblast-derived Dickkopf-1 suppresses NK cell cytotoxicity in breast cancer. Preprint. Res Sq. 2024 rs.3: rs-4202878
78. Haas MS, Kagey MH, Heath H. et al. mDKN-01, a Novel Anti-DKK1 mAb, Enhances Innate Immune Responses in the Tumor Microenvironment. Mol Cancer Res. 2021;19:717-725
79. Xiao Z, Todd L, Huang L. et al. Desmoplastic stroma restricts T cell extravasation and mediates immune exclusion and immunosuppression in solid tumors. Nat Commun. 2023;14:5110
80. Das S, Valton J, Duchateau P. et al. Stromal depletion by TALEN-edited universal hypoimmunogenic FAP-CAR T cells enables infiltration and anti-tumor cytotoxicity of tumor antigen-targeted CAR-T immunotherapy. Front Immunol. 2023;14:1172681
81. Guo Q, Minnier J, Burchard J. et al. Physiologically activated mammary fibroblasts promote postpartum mammary cancer. JCI Insight. 2017;2:e89206
82. Waki H, Tontonoz P. Endocrine functions of adipose tissue. Annu Rev Pathol. 2007;2:31-56
83. Elaraby E, Malek A.I, Abdullah H.W. et al. Natural Killer Cell Dysfunction in Obese Patients with Breast Cancer: A Review of a Triad and Its Implications. J Immunol Res. 2021;2021:9972927
84. Liu S.Q, Chen D.Y, Li B. et al. Single-cell analysis of white adipose tissue reveals the tumor-promoting adipocyte subtypes. J Transl Med. 2023;21:470
85. Zhang Y, Zhao Z, Huang L.A. et al. Molecular mechanisms of snoRNA-IL-15 crosstalk in adipocyte lipolysis and NK cell rejuvenation. Cell Metab. 2023;35:1457-1473.e13
86. Schoenfeld J.D, Vardhana S.A. Not so neutral lipids: Metabolic regulation of the pre-metastatic niche. Cell Metab. 2022;34:1899-1900
87. Bouguerra H, Amal G, Clavel S. et al. Leptin decreases BC cell susceptibility to NK lysis via PGC1A pathway. Endocr Connect. 2020;9:578-586
88. Duong M.N, Cleret A, Matera E.L. et al. Adipose cells promote resistance of breast cancer cells to trastuzumab-mediated antibody-dependent cellular cytotoxicity. Breast Cancer Res. 2015;17:57
89. Lamas B, Nachat-Kappes R, Goncalves-Mendes N. et al. Dietary fat without body weight gain increases in vivo MCF-7 human breast cancer cell growth and decreases natural killer cell cytotoxicity. Mol Carcinog. 2015;54:58-71
90. Spielmann J, Mattheis L, Jung J.-S. et al. Effects of obesity on NK cells in a mouse model of postmenopausal breast cancer. Sci Rep. 2020;10:20606
91. DelaRosa O, Sánchez-Correa B, Morgado S. et al. Human adipose-derived stem cells impair natural killer cell function and exhibit low susceptibility to natural killer-mediated lysis. Stem Cells Dev. 2012;21:1333-1343
92. Gong Z, Li Q, Shi J. et al. Lipid-laden lung mesenchymal cells foster breast cancer metastasis via metabolic reprogramming of tumor cells and natural killer cells. Cell Metab. 2022;34:1960-1976.e9
93. Guo T, Xu J. Cancer-associated fibroblasts: a versatile mediator in tumor progression, metastasis, and targeted therapy. Cancer Metastasis Rev. 2024;43:1095-1116
94. Zarubova J, Hasani-Sadrabadi MM, Norris SCP. et al. Cell-Taxi: Mesenchymal Cells Carry and Transport Clusters of Cancer Cells. Small. 2022;18:e2203515
95. Zhao Y, Shen M, Wu L. et al. Stromal cells in the tumor microenvironment: accomplices of tumor progression? Cell death disease. 2023;14:587
96. Chauhan A, Agarwal S, Masih M. et al. The Multifunction Role of Tumor-Associated Mesenchymal Stem Cells and Their Interaction with Immune Cells in Breast Cancer. Immunol Invest. 2023;52:856-878
97. Gao X, Ren H, Zhang Z. et al. Human lung cancer-derived mesenchymal stem cells promote tumor growth and immunosuppression. Biol Direct. 2024;19:39
98. Hazrati A, Malekpour K, Mirsanei Z. et al. Cancer-associated mesenchymal stem/stromal cells: role in progression and potential targets for therapeutic approaches. Front Immunol. 2023;14:1280601
99. Abbasi B, Shamsasenjan K, Ahmadi M. et al. Mesenchymal stem cells and natural killer cells interaction mechanisms and potential clinical applications. Stem Cell Res Ther. 2022;13:97
100. Yu Y, Liu Y, Zong C. et al. Mesenchymal stem cells with Sirt1 overexpression suppress breast tumor growth via chemokine-dependent natural killer cells recruitment. Sci Rep. 2016;6:35998
101. Hu L, Gu Y, Xu W. et al. Association of clinicopathologic and sonographic features with stromal tumor-infiltrating lymphocytes in triple-negative breast cancer. BMC Cancer. 2024;24:997
102. Stover D.G, Salgado R, Savenkov O. et al. Association between tumor-infiltrating lymphocytes and survival in patients with metastatic breast cancer receiving first-line chemotherapy: analysis of CALGB 40502. NPJ Breast Cancer. 2024;10:75
103. Zhang X, Kim S, Hundal J. et al. Breast Cancer Neoantigens Can Induce CD8+ T-Cell Responses and Antitumor Immunity. Cancer Immunol Res. 2017;5:516-523
104. Kaplanov I, Carmi Y, Kornetsky R. et al. Blocking IL-1β reverses the immunosuppression in mouse breast cancer and synergizes with anti-PD-1 for tumor abrogation. Proc Natl Acad Sci U S A. 2019;116:1361-1369
105. He Y, Hong C, Huang S. et al. STING Protein-Based In Situ Vaccine Synergizes CD4+ T, CD8+ T, and NK Cells for Tumor Eradication. Adv Healthc Mater. 2023;12:e2300688
106. Uzhachenko R.V, Shanker A. CD8+ T Lymphocyte and NK Cell Network: Circuitry in the Cytotoxic Domain of Immunity. Front Immunol. 2019;10:1906
107. Shanker A, Verdeil G, Buferne M. et al. CD8 T cell help for innate antitumor immunity. J Immunol. 2007;179:6651-6662
108. Padoan B, Casar C, Krause J. et al. NKp44/HLA-DP-dependent regulation of CD8 effector T cells by NK cells. Cell Rep. 2024;43:114089
109. Shang L, Jiang X, Zhao X. et al. Mitochondrial DNA-boosted dendritic cell-based nanovaccination triggers antitumor immunity in lung and pancreatic cancers. Cell Rep Med. 2024;5:101648
110. Nguyen N.T, Le X.T, Lee W.T. et al. STING-activating dendritic cell-targeted nanovaccines that evoke potent antigen cross-presentation for cancer immunotherapy. Bioact Mater. 2024;42:345-365
111. Li Y, Shen X, Ding H. et al. Dendritic nanomedicine enhances chemo-immunotherapy by disturbing metabolism of cancer-associated fibroblasts for deep penetration and activating function of immune cells. Acta Pharm Sin B. 2023;14:3680-3696
112. Hegde S, Krisnawan V.E, Herzog B.H. et al. Dendritic Cell Paucity Leads to Dysfunctional Immune Surveillance in Pancreatic Cancer. Cancer Cell. 2020;37:289-307.e9
113. Marcenaro E, Ferranti B, Moretta A. NK-DC interaction: on the usefulness of auto-aggression. Autoimmun Rev. 2005;4:520-525
114. Della Chiesa M, Sivori S, Castriconi R. et al. Pathogen-induced private conversations between natural killer and dendritic cells. Trends Microbiol. 2005;13:128-136
115. Gao W, Wang Y, Wang P. et al. Biosynthetic MnSe nanobomb with low Mn content activates the cGAS-STING pathway and induces immunogenic cell death to enhance antitumor immunity. Acta Biomater. 2024;184:383-396
116. Paladhi A, Daripa S, Nath A. et al. TLR7-Induced Mitochondrial Reactive Oxygen Species Production in Monocyte-derived Dendritic Cells Drives IL-12-Dependent NK Cell Activation and Enhances Antitumor Immunity. J Immunol. 2024;213:1255-1263
117. Liu R, Yang X, Quan Y. et al. Pivotal models and biomarkers related to the prognosis of breast cancer based on the immune cell interaction network. Sci Rep. 2022;12:13673
118. Nagalla S, Chou J.W, Willingham M.C. et al. Interactions between immunity, proliferation and molecular subtype in breast cancer prognosis. Genome Biol. 2013;14:R34
119. Li H, Fang R, Ma R. et al. Amphiregulin promotes activated regulatory T cell-suppressive function via the AREG/EGFR pathway in laryngeal squamous cell carcinoma. Head Face Med. 2024;20:62
120. Plitas G, Konopacki C, Wu K. et al. Regulatory T Cells Exhibit Distinct Features in Human Breast Cancer. Immunity. 2016;45:1122-1134
121. Kos K, Aslam M.A, van de Ven R. et al. Tumor-educated Tregs drive organ-specific metastasis in breast cancer by impairing NK cells in the lymph node niche. Cell Rep. 2022;38:110447
122. Blomberg O.S, Kos K, Spagnuolo L. et al. Neoadjuvant immune checkpoint blockade triggers persistent and systemic Treg activation which blunts therapeutic efficacy against metastatic spread of breast tumors. Oncoimmunology. 2023;12:2201147
123. Olkhanud P.B, Baatar D, Bodogai M. et al. Breast cancer lung metastasis requires expression of chemokine receptor CCR4 and regulatory T cells. Cancer Res. 2009;69:5996-6004
124. Dasgupta S, O'Malley B.W. Transcriptional coregulators: emerging roles of SRC family of coactivators in disease pathology. J Mol Endocrinol. 2014;53:R47-R59
125. Han S.J, Jain P, Gilad Y. et al. Steroid receptor coactivator 3 is a key modulator of regulatory T cell-mediated tumor evasion. Proc Natl Acad Sci U S A. 2023;120:e2221707120
126. Han S.J, Sung N, Wang J. et al. and Lonard D.M. Steroid receptor coactivator-3 inhibition generates breast cancer antitumor immune microenvironment. Breast Cancer Res. 2022;24:73
127. Markowitz J, Wesolowski R, Papenfuss T. et al. Myeloid-derived suppressor cells in breast cancer. Breast Cancer Res Treat. 2013;140:13-21
128. Ngule C, Shi R, Ren X. et al. NAC1 promotes stemness and regulates myeloid-derived cell status in triple-negative breast cancer. Mol Cancer. 2024;23:188
129. Fabian K.P, Padget M.R, Donahue R.N. et al. PD-L1 targeting high-affinity NK (t-haNK) cells induce direct antitumor effects and target suppressive MDSC populations. J Immunother Cancer. 2020;8:e000450
130. Zhang X, Liang Q, Cao Y. et al. Dual depletion of myeloid-derived suppressor cells and tumor cells with self-assembled gemcitabine-celecoxib nano-twin drug for cancer chemoimmunotherapy. J Nanobiotechnology. 2024;22:319
131. Stiff A, Trikha P, Mundy-Bosse B. et al. Nitric Oxide Production by Myeloid-Derived Suppressor Cells Plays a Role in Impairing Fc Receptor-Mediated Natural Killer Cell Function. Clin Cancer Res. 2018;24:1891-1904
132. Chandra D, Jahangir A, Quispe-Tintaya W. et al. Myeloid-derived suppressor cells have a central role in attenuated Listeria monocytogenes-based immunotherapy against metastatic breast cancer in young and old mice. Br J Cancer. 2013;108:2281-2290
133. Chen Y, Song Y, Du W. et al. Tumor-associated macrophages: an accomplice in solid tumor progression. J Biomed Sci. 2019;26:78
134. Wagner J, Rapsomaniki M.A, Chevrier S. et al. A Single-Cell Atlas of the Tumor and Immune Ecosystem of Human Breast Cancer. Cell. 2019;177:1330-1345.e18
135. Krneta T, Gillgrass A, Poznanski S. et al. M2-polarized and tumor-associated macrophages alter NK cell phenotype and function in a contact-dependent manner. J Leukoc Biol. 2017;101:285-295
136. Do-Thi V.A, Park S.M, Park S.M. et al. IL9 Polarizes Macrophages to M1 and Induces the Infiltration of Antitumor Immune Cells via MIP-1 and CXCR3 Chemokines. Cancer Res Commun. 2023;3:80-96
137. Qian B, Deng Y, Im J.H. et al. A distinct macrophage population mediates metastatic breast cancer cell extravasation, establishment and growth. PLoS One. 2009;4:e6562
138. Chen Q, Zhang X.H.F, Massagué J. Macrophage binding to receptor VCAM-1 transmits survival signals in breast cancer cells that invade the lungs. Cancer Cell. 2011;20:538-549
139. Brownlie D, Doughty-Shenton D, Yh Soong D. et al. Metastasis-associated macrophages constrain antitumor capability of natural killer cells in the metastatic site at least partially by membrane bound transforming growth factor β. J Immunother Cancer. 2021;9:e001740
140. Bronger H, Kraeft S, Schwarz-Boeger U. et al. Modulation of CXCR3 ligand secretion by prostaglandin E2 and cyclooxygenase inhibitors in human breast cancer. Breast Cancer Res. 2012;14:R30
141. Punyawatthananukool S, Matsuura R, Wongchang T. et al. Prostaglandin E2-EP2/EP4 signaling induces immunosuppression in human cancer by impairing bioenergetics and ribosome biogenesis in immune cells. Nat Commun. 2024;15:9464
142. Holt D, Ma X, Kundu N. et al. Prostaglandin E(2) (PGE (2)) suppresses natural killer cell function primarily through the PGE(2) receptor EP4. Cancer Immunol Immunother. 2011;60:1577-1586
143. Holt D.M, Ma X, Kundu N. et al. Modulation of host natural killer cell functions in breast cancer via prostaglandin E2 receptors EP2 and EP4. J Immunother. 2012;35:179-188
144. Kundu N, Ma X, Holt D. et al. Antagonism of the prostaglandin E receptor EP4 inhibits metastasis and enhances NK function. Breast Cancer Res Treat. 2009;117:235-242
145. Heath J, Mirabelli C, Annis MG. et al. The Neurodevelopmental Protein POGZ Suppresses Metastasis in Triple-Negative Breast Cancer by Attenuating TGFβ Signaling. Cancer Res. 2024;84:3743-3760
146. Wong J.K.M, McCulloch T.R, Alim L. et al. TGF-β signalling limits effector function capacity of NK cell anti-tumor immunity in human bladder cancer. EBioMedicine. 2024;104:105176
147. Ostapchuk Y.O, Cetin E.A, Perfilyeva Y.V. et al. Peripheral blood NK cells expressing HLA-G, IL-10 and TGF-β in healthy donors and breast cancer patients. Cell Immunol. 2015;298:37-46
148. Slattery K, Woods E, Zaiatz-Bittencourt V. et al. TGFβ drives NK cell metabolic dysfunction in human metastatic breast cancer. J Immunother Cancer. 2021;9:e002044corr1
149. Zhao Y, Hu J, Li R. et al. Enhanced NK cell adoptive antitumor effects against breast cancer in vitro via blockade of the transforming growth factor-β signaling pathway. Onco Targets Ther. 2015;8:1553-1559
150. Habanjar O, Bingula R, Decombat C. et al. Crosstalk of Inflammatory Cytokines within the Breast Tumor Microenvironment. Int J Mol Sci. 2023;24:4002
151. Santana-Hernández S, Suarez-Olmos J, Servitja S. et al. NK cell-triggered CCL5/IFN-γ-CXCL9/10 axis underlies the clinical efficacy of neoadjuvant anti-HER2 antibodies in breast cancer. J Exp Clin Cancer Res. 2024;43:10
152. Gao W, Wen H, Liang L. et al. IL20RA signaling enhances stemness and promotes the formation of an immunosuppressive microenvironment in breast cancer. Theranostics. 2021;11:2564-2580
153. Zheng G, Guo Z, Li W. et al. Interaction between HLA-G and NK cell receptor KIR2DL4 orchestrates HER2-positive breast cancer resistance to trastuzumab. Signal Transduct Target Ther. 2021;6:236
154. Zheng G, Jia L, Yang A.-G. et al. Roles of HLA-G/KIR2DL4 in Breast Cancer Immune Microenvironment. Front Immunol. 2022;13:791975
155. Zanker D.J, Owen K.L, Baschuk N. et al. Loss of type I IFN responsiveness impairs natural killer cell antitumor activity in breast cancer. Cancer Immunol Immunother. 2021;70:2125-2138
156. Rautela J, Baschuk N, Slaney C.Y. et al. Loss of Host Type-I IFN Signaling Accelerates Metastasis and Impairs NK-cell Antitumor Function in Multiple Models of Breast Cancer. Cancer Immunol Res. 2025;3:1207-1217
157. Briukhovetska D, Suarez-Gosalvez J, Voigt C. et al. T cell-derived interleukin-22 drives the expression of CD155 by cancer cells to suppress NK cell function and promote metastasis. Immunity. 2023;56:143-161.e11
158. Roda J.M, Parihar R, Lehman A. et al. Interleukin-21 enhances NK cell activation in response to antibody-coated targets. J Immunol. 2006;177:120-129
159. Mollica Poeta V, Massara M. et al. Chemokines and Chemokine Receptors: New Targets for Cancer Immunotherapy. Front Immunol. 2019;10:379
160. Ozga A.J, Chow M.T, Luster A.D. et al. Chemokines and the immune response to cancer. Immunity. 2021;54:859-874
161. Cao X, Song Y, Wu H. et al. C-X-C Motif Chemokine Ligand 9 Correlates with Favorable Prognosis in Triple-Negative Breast Cancer by Promoting Immune Cell Infiltration. Mol Cancer Ther. 2023;22:1493-1502
162. Correia A.L, Guimaraes J.C, Auf der Maur P. et al. Hepatic stellate cells suppress NK cell-sustained breast cancer dormancy. Nature. 2021;594:566-571
163. Dreyer T.F, Kuhn S, Stange C. et al. The Chemokine CX3CL1 Improves Trastuzumab Efficacy in HER2 Low-Expressing Cancer In Vitro and In Vivo. Cancer Immunol Res. 2021;9(7):779-789
164. Araujo J.M, Gomez A.C, Aguilar A. et al. Effect of CCL5 expression in the recruitment of immune cells in triple negative breast cancer. Sci Rep. 2018;8:4899
165. He J, He Y, Biao R. et al. STYK1 mediates NK cell anti-tumor response through regulating CCR2 and trafficking. J Transl Med. 2024;22:943
166. Ma X, Holt D, Kundu N. et al. A prostaglandin E (PGE) receptor EP4 antagonist protects natural killer cells from PGE2-mediated immunosuppression and inhibits breast cancer metastasis. Oncoimmunology. 2013;2:e22647
167. Liu C, Lai H, Chen T. et al. Boosting Natural Killer Cell-Based Cancer Immunotherapy with Selenocystine/Transforming Growth Factor-Beta Inhibitor-Encapsulated Nanoemulsion. ACS Nano. 2020;14:11067-11082
168. Jung H.-Y, Lee D.-K, Lee M. et al. ELK3-CXCL16 axis determines natural killer cell cytotoxicity via the chemotactic activity of CXCL16 in triple negative breast cancer. Oncoimmunology. 2023;12:2190671
169. Milling L.E, Garafola D, Agarwal Y. et al. Neoadjuvant STING Activation, Extended Half-life IL2, and Checkpoint Blockade Promote Metastasis Clearance via Sustained NK-cell Activation. Cancer Immunol Res. 2022;10:26-39
170. Czapla J, Drzyzga A, Ciepła J. et al. Combination of STING agonist with anti-vascular RGD-(KLAKLAK)2 peptide as a novel anti-tumor therapy. Cancer Immunol Immunother. 2024;73:148
171. Gellert J, Jäkel A, Danielczyk A. et al. GT-00AxIL15, a Novel Tumor-Targeted IL-15-Based Immunocytokine for the Treatment of TA-MUC1-Positive Solid Tumors: Preclinical In Vitro and In Vivo Pharmacodynamics and Biodistribution Studies. Int J Mol Sci. 2024;25:1406
172. Stravokefalou V, Stellas D, Karaliota S. et al. Heterodimeric IL-15 (hetIL-15) reduces circulating tumor cells and metastasis formation improving chemotherapy and surgery in 4T1 mouse model of TNBC. Front Immunol. 2022;13:1014802
173. Guittard G, Dios-Esponera A, Palmer D.C. et al. The Cish SH2 domain is essential for PLC-γ1 regulation in TCR stimulated CD8+ T cells. Sci Rep. 2018;8:5336
174. Bernard P.-L, Delconte R, Pastor S. et al. Targeting CISH enhances natural cytotoxicity receptor signaling and reduces NK cell exhaustion to improve solid tumor immunity. J Immunother Cancer. 2022;10:e004244
175. Schlechter B.L, Stebbing J. CCR5 and CCL5 in metastatic colorectal cancer. J Immunother Cancer. 2024;12:e008722
176. Chen L, Xu G, Song X. et al. A novel antagonist of the CCL5/CCR5 axis suppresses the tumor growth and metastasis of triple-negative breast cancer by CCR5-YAP1 regulation. Cancer Lett. 2024;583:216635
177. Haq A.T.A, Yang P.P, Jin C. et al. Immunotherapeutic IL-6R and targeting the MCT-1/IL-6/CXCL7/PD-L1 circuit prevent relapse and metastasis of triple-negative breast cancer. Theranostics. 2024;14:2167-2189
178. An D, He P, Liu H. et al. Enhanced chemoimmunotherapy of breast cancer in mice by apolipoprotein A1-modified doxorubicin liposomes combined with interleukin-21. J Drug Target. 2023;31:1098-1110
179. Bruni S, Mauro F.L, Proietti C.J. et al. Blocking soluble TNFα sensitizes HER2-positive breast cancer to trastuzumab through MUC4 downregulation and subverts immunosuppression. J Immunother Cancer. 2023;11:e005325
180. Zhang B, Shi J, Shi X. et al. Development and evaluation of a human CD47/HER2 bispecific antibody for Trastuzumab-resistant breast cancer immunotherapy. Drug Resist Updat. 2024;74:101068
181. Cabo M, Santana-Hernández S, Costa-Garcia M. et al. CD137 Costimulation Counteracts TGFβ Inhibition of NK-cell Antitumor Function. Cancer Immunol Res. 2021;9:1476-1490
182. Knudson KM, Hicks KC, Luo X. et al. M7824, a novel bifunctional anti-PD-L1/TGFβ Trap fusion protein, promotes anti-tumor efficacy as monotherapy and in combination with vaccine. Oncoimmunology. 2019;8:e1584435
183. Yi M, Wu Y, Niu M. et al. Anti-TGF-β/PD-L1 bispecific antibody promotes T cell infiltration and exhibits enhanced antitumor activity in triple-negative breast cancer. J Immunother Cancer. 2022;10:e005543
184. Yi M, Niu M, Wu Y. et al. Combination of oral STING agonist MSA-2 and anti-TGF-β/PD-L1 bispecific antibody YM101: a novel immune cocktail therapy for non-inflamed tumors. J Hematol Oncol. 2022;15:142
185. Yi M, Li T, Niu M. et al. Blockade of CCR5+ T Cell Accumulation in the Tumor Microenvironment Optimizes Anti-TGF-β/PD-L1 Bispecific Antibody. Adv Sci (Weinh). 2024;11:e2408598
186. Juliá E.P, Amante A, Pampena M.B. et al. Avelumab, an IgG1 anti-PD-L1 Immune Checkpoint Inhibitor, Triggers NK Cell-Mediated Cytotoxicity and Cytokine Production Against Triple Negative Breast Cancer Cells. Front Immunol. 2018;9:2140
187. Xu C, Zhang Y, Rolfe P.A. et al. Combination Therapy with NHS-muIL12 and Avelumab (anti-PD-L1) Enhances Antitumor Efficacy in Preclinical Cancer Models. Clin Cancer Res. 2017;23:5869-5880
188. Li R, Sant S, Brown E. et al. Tucatinib promotes immune activation and synergizes with programmed cell death-1 and programmed cell death-ligand 1 inhibition in HER2-positive breast cancer. J Natl Cancer Inst. 2023;115:805-814
189. Maxwell M, Yan D, Rivest B. et al. INTASYL self-delivering RNAi decreases TIGIT expression, enhancing NK cell cytotoxicity: a potential application to increase the efficacy of NK adoptive cell therapy against cancer. Cancer Immunol Immunother. 2024;73:239
190. Harjunpää H, Guillerey C. TIGIT as an emerging immune checkpoint. Clin Exp Immunol. 2020;200:108-119
191. Shen X, Fu W, Wei Y. et al. TIGIT-Fc Promotes Antitumor Immunity. Cancer Immunol Res. 2021;9:1088-1097
192. Demeule M, Currie J.C, Charfi C. et al. Sudocetaxel Zendusortide (TH1902) triggers the cGAS/STING pathway and potentiates anti-PD-L1 immune-mediated tumor cell killing. Front Immunol. 2024;15:1355945
193. Juliá E.P, Mordoh J, Levy E.M. Cetuximab and IL-15 Promote NK and Dendritic Cell Activation In Vitro in Triple Negative Breast Cancer. Cells. 2020;9:1573
194. Fukuda M.E, Iwadate Y, Machida T. et al. Cathepsin D is a potential serum marker for poor prognosis in glioma patients. Cancer Res. 2005;65:5190-5194
195. David T, Mallavialle A, Faget J. et al. Anti-cathepsin D immunotherapy triggers both innate and adaptive anti-tumor immunity in breast cancer. Br J Pharmacol. 2023
196. Ruiz Manzano R.A, Nava-Castro K.E, Palacios-Arreola M.I. et al. Intratumoral Treatment with 5-Androstene-3β, 17α-Diol Reduces Tumor Size and Lung Metastasis in a Triple-Negative Experimental Model of Breast Cancer. Int J Mol Sci. 2022;23:11944
197. Lian S, Li W, Zhong C. et al. Ganoderma lucidum spore oil synergistically enhances the function of cyclophosphamide in the prevention of breast cancer metastasis. J Chin Med Assoc. 2024;87:305-313
198. Mediratta K, El-Sahli S, Marotel M. et al. Targeting CD73 with flavonoids inhibits cancer stem cells and increases lymphocyte infiltration in a triple-negative breast cancer mouse model. Front Immunol. 2024;15:1366197
199. Yang H, Zhan X, Zhao J. et al. Schisandrin C enhances type I IFN response activation to reduce tumor growth and sensitize chemotherapy through antitumor immunity. Front Pharmacol. 2024;15:1369563
200. Askar M.A, El-Sayyad G.S, Guida M.S. et al. Amygdalin-folic acid-nanoparticles inhibit the proliferation of breast cancer and enhance the effect of radiotherapy through the modulation of tumor-promoting factors/ immunosuppressive modulators in vitro. BMC Complement Med Ther. 2023;23:162
201. Zhong C, Li Y, Li W. et al. Ganoderma lucidum extract promotes tumor cell pyroptosis and inhibits metastasis in breast cancer. Food Chem Toxicol. 2023;174:113654
202. Li F, Shi Y, Ma M. et al. Xianling Lianxia formula improves the efficacy of trastuzumab by enhancing NK cell-mediated ADCC in HER2-positive BC. J Pharm Anal. 2024;14:100977
203. Yang C, Qian C, Zhang T. et al. The killing effect of Tanshinol on breast cancer cells: insight into the reversion of TGF-β1-mediated suppression of NK cell functions. Front Biosci (Landmark Ed). 2021;26:1106-1118
204. Durmus E, Ozman Z, Ceyran I.H. et al. Chrysin Enhances Anti-Cancer Activity of Jurkat T Cell and NK-92 Cells Against Human Breast Cancer Cell Lines. Chem Biodivers. 2024;21:e202400806
205. Yang C, Qian C, Zheng W. et al. Ginsenoside Rh2 enhances immune surveillance of natural killer (NK) cells via inhibition of ERp5 in breast cancer. Phytomedicine. 2024;123:155180
206. Masuda Y, Yamashita S, Nakayama Y. et al. Maitake Beta-Glucan Enhances the Therapeutic Effect of Trastuzumab via Antibody-Dependent Cellular Cytotoxicity and Complement-Dependent Cytotoxicity. Biol Pharm Bull. 2024;47:840-847
207. Sun R, Zhang M, Li B. et al. A Novel Bromophenol Compound from Leathesia nana Inhibits Breast Cancer in a Direct Tumor Killing and Immunotherapy Manner. Molecules. 2023;28:5349
208. Myers J.A, Miller J.S. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021;18:85-100
209. Burns L.J, Weisdorf D.J, DeFor T.E. et al. IL-2-based immunotherapy after autologous transplantation for lymphoma and breast cancer induces immune activation and cytokine release: a phase I/II trial. Bone Marrow Transplant. 2003;32:177-186
210. Becker P.S.A, Suck G, Nowakowska P. et al. Selection and expansion of natural killer cells for NK cell-based immunotherapy. Cancer Immunol Immunother. 2016;65:477-484
211. Goldenson B.H, Hor P, Kaufman D.S. iPSC-Derived Natural Killer Cell Therapies - Expansion and Targeting. Front Immunol. 2022;13:841107
212. Geller M.A, Cooley S, Judson P.L. et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy. 2011;13:98-107
213. Baek S, Kim C.S, Kim S.B. et al. Combination therapy of renal cell carcinoma or breast cancer patients with dendritic cell vaccine and IL-2: results from a phase I/II trial. J Transl Med. 2011;9:178
214. Schöffski P, Tan D.S.W, Martín M. et al. Phase I/II study of the LAG-3 inhibitor ieramilimab (LAG525) ± anti-PD-1 spartalizumab (PDR001) in patients with advanced malignancies. J Immunother Cancer. 2022;10:e003776
215. Quiroga D, Wesolowski R, Zelinskas S. et al. An Open-Label Study of Subcutaneous CpG Oligodeoxynucleotide (PF03512676) in Combination with Trastuzumab in Patients with Metastatic HER2+ Breast Cancer. Cancer Control. 2024;31:10732748241250189
216. Geurts V.C.M, Voorwerk L, Balduzzi S. et al. Unleashing NK- and CD8 T cells by combining monalizumab and trastuzumab for metastatic HER2-positive breast cancer: Results of the MIMOSA trial. Breast. 2023;70:76-81
217. Coletta A.M, Agha N.H, Baker F.L. et al. The impact of high-intensity interval exercise training on NK-cell function and circulating myokines for breast cancer prevention among women at high risk for breast cancer. Breast Cancer Res Treat. 2021;187:407-416
218. Kandala S, Ramos M, Voith von Voithenberg L. et al. Chronic chromosome instability induced by Plk1 results in immune suppression in breast cancer. Cell Rep. 2023;42:113266
219. Muraro E, De Zorzi M, Miolo G. et al. KIR-HLA Functional Repertoire Influences Trastuzumab Efficiency in Patients With HER2-Positive Breast Cancer. Front Immunol. 2021;12:791958
220. Mac Donald A, Guipouy D, Lemieux W. et al. KLRC1 knockout overcomes HLA-E-mediated inhibition and improves NK cell antitumor activity against solid tumors. Front Immunol. 2023;14:1231916
221. Li T, Niu M, Zhang W. et al. CAR-NK cells for cancer immunotherapy: recent advances and future directions. Front Immunol. 2024;15:1361194
222. Raftery M.J, Franzén A.S, Radecke C. et al. Next Generation CD44v6-Specific CAR-NK Cells Effective against Triple Negative Breast Cancer. Int J Mol Sci. 2023;24:9038
223. Won E.-J, Lee M, Lee E.-K. et al. Lipid-Based Nanoparticles Fused with Natural Killer Cell Plasma Membrane Proteins for Triple-Negative Breast Cancer Therapy. Pharmaceutics. 2024;16:1142
224. Tao B, Du R, Zhang X. et al. Engineering CAR-NK cell derived exosome disguised nano-bombs for enhanced HER2 positive breast cancer brain metastasis therapy. J Control Release. 2023;363:692-706
225. Eitler J, Rackwitz W, Wotschel N. et al. CAR-mediated targeting of NK cells overcomes tumor immune escape caused by ICAM-1 downregulation. J Immunother Cancer. 2024;12:e008155
226. Li X, Zhu Y, Yi J. et al. Adoptive cell immunotherapy for breast cancer: harnessing the power of immune cells. J Leukoc Biol. 2024;115:866-881
Author contact
Corresponding authors: Huangan Wu, Medical Laboratory, Yueyang Hospital of Integrated Traditional Chinese and Western Medicine; 110 Ganhe Road, Yueyang Hospital, Hongkou District, Shanghai 200437, P.R. China; Email: wuhuangancom; and Sheng Liu, Longhua Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine; 725 Wanping South Road, Xuhui District, Shanghai 200030, P.R. China. Email address: lhliushengcom.

 Global reach, higher impact
Global reach, higher impact