Impact Factor ISSN: 1449-2288
Int J Biol Sci 2026; 22(9):4633-4646. doi:10.7150/ijbs.130233 This issue Cite
Review
Redeemable Lymphocytes in Cancer Radioimmunotherapy: Saving Our Soldiers in the Complex of Cancer-Fighting Field
1. PRAG Therapy Center, The Second Affiliated Hospital of Soochow University, Suzhou, Jiangsu 215123, China.
2. Center for Cancer Diagnosis and Treatment, The Second Affiliated Hospital of Soochow University, Suzhou, Jiangsu 215123, China.
3. Department of Radiation Oncology, University of California Davis, Sacramento, California, USA.
# Equal contribution.
Received 2025-12-19; Accepted 2026-3-17; Published 2026-4-23
Abstract
Lymphocytes are pivotal effectors in anticancer immunity, yet their profound radiosensitivity makes them vulnerable to collateral damage during radiotherapy (RT). This radiation-induced lymphopenia (RIL) undermines the efficacy of radioimmunotherapy and correlates with poor prognosis. Our work underscores that even low-dose splenic irradiation (e.g., a mean dose of 5 Gy) significantly increases severe RIL risk. This review synthesizes mechanistic insights into RIL, linking splenic irradiation and disruption of lymphoid homing to systemic immunosuppression. We critically evaluate transformative strategies to reconcile this paradox: FLASH-RT and spatially fractionated RT minimize lymphocyte exposure, while synergistic integration of low-dose and ablative RT reshapes the tumor microenvironment. Precision-guided immunotherapy sequencing—CTLA-4 blockade pre-RT and PD-1 inhibition post-RT—further exploits temporal windows of immune activation. By harnessing lymphocyte migration dynamics and radiomics, we chart a roadmap for lymphocyte-sparing radioimmunotherapy. We ultimately advocate for a 'Protect and Amplify' paradigm, prioritizing immune preservation to unlock durable antitumor responses, particularly in advanced cancer.
Keywords: Splenic irradiation, Radiation-induced lymphopenia, Lymphocyte-sparing radiotherapy, Splenic dose constraints, Abscopal effect
Introduction
Radiotherapy (RT) and immunotherapy represent a formidable alliance against cancer, poised to harness radiation's dual capacity for local tumor control and systemic immune activation [1]. The synergy between RT and immunotherapy hinges on RT's capacity to ignite antitumor immunity by inducing immunogenic cell death, releasing tumor neoantigens, and modulating the tumor microenvironment (TME) [2-4]. However, this alliance faces a critical challenge that RT's indiscriminate cytotoxicity concurrently decimates lymphocytes, the cornerstone of antitumor immunity [5, 6]. This paradox underscores the necessity of developing strategies that preserve RT's immunogenic benefits while mitigating its lymphotoxic effects [7, 8]. While RT promotes T cell clonality and dendritic cell (DC) activation [1], its clinical integration with immune checkpoint inhibitors is limited by radiation-induced lymphopenia (RIL), an adverse event independently correlating with reduced overall survival (OS) in glioblastoma, pancreatic cancer, and other solid tumors [9-11]. Moreover, the clinical burden of RIL is substantial, with grade ≥3 lymphopenia reported in approximately 20-40% of patients undergoing chemoradiotherapy for various solid tumors, such as pancreatic cancer and glioblastoma, underscoring its common occurrence alongside its detrimental prognostic impact [11, 12].
RIL epitomizes a critical paradox: RT eradicates tumor cells while crippling the “soldiers” essential for sustaining systemic immunity. Lymphocytes exhibit exquisite radiosensitivity, with B cells succumbing to doses as low as 0.5 Gy and CD4+ T cells—orchestrators of adaptive immunity—displaying greater vulnerability than CD8+ counterparts [13, 14]. Concurrently, irradiation of lymphoid organs inflicts collateral damage: splenic doses >9 Gy in pancreatic cancer deplete circulating lymphocyte reserves [15, 16], while nodal RT disrupts the C-C Chemokine Receptor Type 7 (CCR7)- C-C Motif Chemokine Ligand 19/21 (CCL19/21) axis, impairing DC trafficking and T cell priming [17-20]. These mechanisms collectively propagate immunosuppression, enabling metastatic escape.
Emerging innovations, however, illuminate a path toward “lymphocyte-sparing radioimmunotherapy.” FLASH-RT (>40 Gy/s) delivers tumoricidal doses in milliseconds, outpacing cellular repair to minimize lymphocyte exposure [21-25]. Spatially fractionated techniques (GRID, Lattice) sculpt dose distributions to spare circulating cells. Strategically sequenced immunotherapy—CTLA-4 blockade pre-RT to deplete regulatory T cells (Tregs), PD-1 inhibition post-RT to reinvigorate primed CD8+ T cells—exploits temporal windows of immune activation [26]. Pioneering studies further decode lymphocyte migration: DCs imprint organ-specific homing signatures (e.g., gut-tropic α4β7/CCR9+ T cells), while radiomics tracks CD8+ T cell infiltration in real time [26, 27].
Our research into RIL underscores the critical impact of even low-dose, cumulative exposure to lymphoid organs. We found that in patients receiving thoracic radiotherapy, a mean splenic dose as low as 5 Gy delivered in conventional fractions (e.g., 0.2 Gy per fraction over 25 fractions) significantly increased the risk of severe (grade ≥3) lymphopenia, which was independently associated with reduced OS [28]. This observation highlights that the threat of RIL is not confined to high-dose ablative settings but is a pervasive concern in standard regimens, necessitating a fundamental re-evaluation of radiotherapy planning.
This review synthesizes these advances, including insights from our own work, into a cohesive framework. We dissect RIL mechanisms, validate organ-specific dose constraints, and integrate cutting-edge technologies to propose actionable protocols. Our goal is to transform RT from an immune adversary into a strategic ally, empowering lymphocytes to eradicate tumors and redefine cancer therapy paradigms.
1. Mechanisms of RIL: Decimating the Immune Sentinels
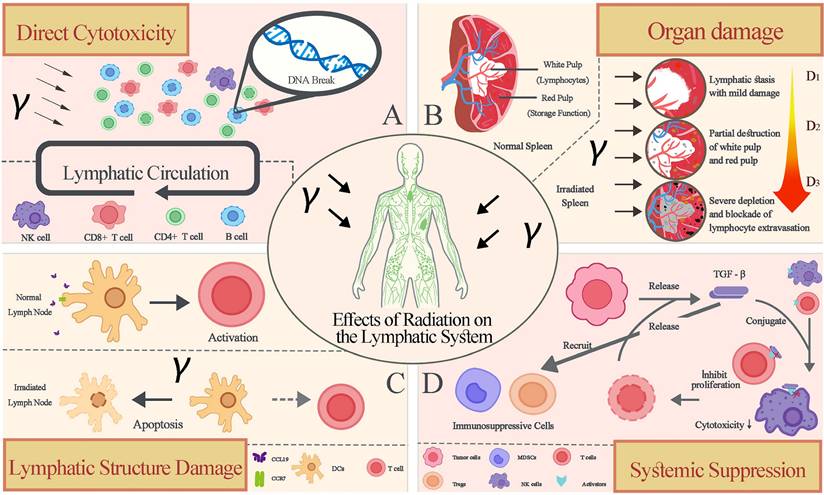
RIL stems from the exquisite radiosensitivity of lymphocytes—the cornerstone of antitumor immunity—and collateral damage to lymphoid organs. This dual assault cripples systemic immune surveillance, enabling tumor escape. We dissect four interconnected mechanisms driving RIL (Figure 1).
1.1 Hierarchical Radiosensitivity of Lymphocyte Subsets
Lymphocytes exhibit a striking hierarchy of radiosensitivity that dictates their vulnerability during radiotherapy, with B cells emerging as the most fragile subset (LD₉₀ ≈ 1 Gy), undergoing rapid apoptosis even at doses as low as 0.5 Gy due to impaired DNA repair mechanisms [29-32]. CD4⁺ T cells—critical orchestrators of adaptive immunity—follow with intermediate sensitivity (LD₉₀ ≈ 3 Gy), exceeding the susceptibility of their CD8⁺ T counterparts (LD₉₀ ≈ 4 Gy), thereby disrupting the balance necessary for coordinated antitumor responses [33-35]. In contrast, natural killer (NK) cells demonstrate relative resilience (LD₉₀ >5 Gy), while Tregs paradoxically withstand higher doses (>5 Gy), tilting the immunological landscape toward suppression by selectively surviving radiation-induced stress [12, 17, 36, 37]. This disproportionate depletion of B and CD4⁺ T cells cripples humoral immunity and T-cell priming capacity, collectively compromising systemic immune surveillance—a vulnerability exploitable by tumors to evade eradication [37]. Consequently, preserving these subsets necessitates technological innovations like ultra-high dose rate FLASH-RT, which minimizes exposure time to outpace intrinsic radiosensitivity [22, 38].
This profound radiosensitivity in terminally differentiated lymphocytes presents a biological paradox. Unlike other radioresistant somatic cells, lymphocytes are exquisitely tuned for rapid, clonal responses, rendering them uniquely vulnerable. The mechanisms underpinning this 'Achilles' heel' are threefold: (1) A Pre-Apoptotic State: Activated lymphocytes maintain a high basal level of pro-apoptotic proteins (e.g., Bax, Bim), priming them for rapid programmed cell death upon DNA damage signals from radiation. (2) Constrained DNA Repair Capacity: Naïve B and T cells exhibit relatively low activity of non-homologous end joining and other DNA double-strand break repair pathways compared to stem cells, failing to efficiently resolve radiation-induced DNA lesions. (3) High Metabolic Stress: The aerobic glycolysis and oxidative phosphorylation required for lymphocyte activation and proliferation generate substantial reactive oxygen species, creating a state of metabolic stress that radiation exacerbates, leading to metabolic collapse. Thus, lymphocyte radio-sensitivity is not a design flaw but a consequence of their fundamental physiology, making them unintended collateral damage in the radio-therapeutic battlefield (Table 1).
Mechanisms of Radiation-Induced Lymphopenia (RIL). Radiotherapy compromises antitumor immunity through four key mechanisms: (A) Hierarchical Radiosensitivity of lymphocyte subsets, with B cells being most vulnerable and Tregs most resistant. (B) Splenic Irradiation, which depletes the body's major lymphocyte reservoir. (C) Nodal Irradiation, which disrupts the CCR7- CCL19/21 homing axis essential for T-cell priming. (D)Tumor Microenvironment Immunosuppression, where radiation-induced TGF-β creates an inhibitory "fog". RT, Radiotherapy; DC, Dendritic Cell; Treg, Regulatory T Cell; MDSC, Myeloid-Derived Suppressor Cell; FRC, Fibroblastic Reticular Cell.
Radiation Sensitivity of Lymphocyte Subsets
| Lymphocyte Subtype | D10 (Gy) | D50 (Gy) | Key Characteristics | References |
|---|---|---|---|---|
| B cells | 0.5-1.0 | 0.3-0.5 | Most radiosensitive; rapid apoptosis | [63, 64] |
| CD4+ T cells | 2.0-3.0 | 1.5-2.0 | Critical for adaptive immunity Higher sensitivity than CD8+ | [32] |
| CD8+ T cells | 3.0-4.0 | 2.0-2.5 | Effector T cells; relatively resistant | [32, 37] |
| NK cells | 5.0-6.0 | 3.0-4.0 | Innate immunity; highest radioresistance | [37] |
| Regulatory T cells | >5.0 | >3.0 | Paradoxically resistant; immunosuppressive | [32, 37] |
In vitro radiosensitivity parameters (D10 and D50) for major lymphocyte populations, highlighting their differential vulnerability and key functional characteristics.
1.2 Splenic Irradiation: Depleting the Lymphocyte Reservoir
As the largest secondary lymphoid organ, the spleen functions as a critical barracks for circulating lymphocytes, where daily trafficking through its red pulp structures maintains systemic immune vigilance; however, unintended irradiation during abdominal radiotherapy inflicts catastrophic damage to this reservoir, with splenic doses exceeding 9 Gy in pancreatic cancer or 18.5 Gy in gastric cancer driving severe lymphopenia through stromal niche disruption and impaired lymphocyte homing [13, 28, 39]. This dose-dependent depletion follows a linear relationship, evidenced by splenic V₅ (volume receiving ≥5 Gy) and mean dose (Dmean) correlating directly with nadir absolute lymphocyte counts (ALC), where a Dmean >9 Gy in pancreatic cancer elevates grade 3-4 lymphopenia risk by 4.2-fold and increases mortality hazard by 65% (Hazard Ratio (HR)=1.65); while gastric cancer Dmean >18.5 Gy similarly raises mortality risk by 50% (HR=1.50) [28, 39, 40]. Critically, the spleen's radiosensitivity stems from its rich vascularity and high lymphocyte turnover, rendering it uniquely susceptible even when outside the target volume, thereby transforming incidental irradiation into a preventable cause of immunosuppression. Consequently, enforcing splenic dose constraints (e.g., V₅ <30% or Dmean <8 Gy in pancreatic RT) emerges as an actionable strategy to preserve this immune sanctuary, much as shielding a barracks protects soldiers for future battles [13, 41] (Table 2).
Splenic Dose Thresholds and Associated Clinical Outcomes in Solid Tumors
| Cancer Type | Splenic D (Gy) | Lymphopenia Grade (CTCAE) | Hazard Ratio (OS) | Reference |
|---|---|---|---|---|
| Pancreatic Cancer | >9.0 | Grade 3-4 | 1.65 | [39] |
| Gastric Cancer | >18.5 | Grade 3-4 | 1.50 | [28] |
| Esophageal Cancer | >15.0 | Grade 3-4 | 1.45 | [40] |
| Glioblastoma | >5.0 | Grade 3-4 | 1.70 | [8] |
Established splenic dose-volume parameters that correlate with an increased risk of severe (Grade 3-4) lymphopenia and worse overall survival across different cancer types. CTCAE, Common Terminology Criteria for Adverse Events; OS, Overall Survival.
1.3. Nodal Irradiation: Disrupting the CCR7-CCL19/21 Homing Axis
Lymph nodes serve as essential training grounds for T-cell priming, where DCs educate naïve T cells to recognize tumor antigens; however, therapeutic irradiation inflicts collateral damage by suppressing CCL19/21 secretion from fibroblastic reticular cells (FRCs) and downregulating CCR7 receptors on T cells and DCs, thereby severing the chemokine-guided homing axis required for immune cell trafficking and activation [18, 19]. This disruption manifests as persistent intranodal lymphopenia—evidenced by >50% reductions in lymphocyte endothelial cell density and CCR7⁺T-cell infiltration after 20 Gy in murine models—which directly attenuates antitumor immunity by impairing DC-mediated antigen presentation and subsequent T-cell clonality [18, 20]. Critically, the compromised priming capacity propagates systemic immunosuppression, enabling metastatic escape in 63% of head and neck cancer patients receiving elective nodal irradiation versus 28% with delayed protocols [42]. Consequently, delaying metastatic nodal irradiation until after primary tumor treatment preserves CCR7-CCL19/21 functionality, effectively shielding this immunological "boot camp" to sustain T-cell recruitment and amplify abscopal responses by 37% in preclinical studies, a strategic pause that arms rather than disarms the immune system [18, 42].
1.4. TME Immunosuppression: The Perverted Battlefield
While radiotherapy directly eliminates tumor cells, it paradoxically poisons the immunological terrain by triggering a surge of transforming growth factor-β (TGF-β), an immunosuppressive "fog" that permeates the TME, reprogramming macrophages toward M2 phenotypes, recruiting myeloid-derived suppressor cells (MDSCs), and inducing endoplasmic reticulum stress through reactive oxygen species, collectively crippling lymphocyte function and survival [20, 43]. This immunosuppressive cascade manifests most acutely in the irradiated field, where radiation-resistant tumor-associated macrophages amplify TGF-β signaling by 3-fold post-20 Gy, establishing a vicious cycle that excludes cytotoxic T cells and expands Treg infiltrates even as circulating lymphocytes are depleted [44]. Critically, the systemic spillover of these mediators, evidenced by 2.5-fold elevated serum TGF-β levels in patients receiving thoracic RT, extends immunosuppression beyond the irradiated volume, accelerating metastatic outgrowth in 68% of preclinical models with RIL [20]. Consequently, strategically deployed low-dose radiotherapy (LD-RT; 0.5-2 Gy × 4 fractions) acts as an immunological "detoxifier," reversing polarization from M2 to immunostimulatory M1 macrophages, suppressing TGF-β by >60%, and restoring CD8⁺ T-cell infiltration, effectively cleansing the battlefield to enable subsequent high-dose ablative strikes [22].
2. Clinical Impact of RIL: Compromising the Frontline Defense
RIL transcends a mere laboratory anomaly that it is an independent predictor of poor survival across solid tumors, directly eroding the immune arsenal necessary for tumor control.
2.1 Prognostic Significance: The Mortality Toll of Lymphocyte Decimation
RIL exacts a lethal toll across solid tumors, with meta-analyses confirming a 65% increase in mortality risk for patients developing grade 3-4 RIL versus those with grade 0-2 lymphopenia, a survival penalty independent of tumor stage, histology, or treatment modality that underscores lymphocytes' irreplaceable role as frontline defenders against metastatic dissemination [11, 12]. This association manifests most dramatically in gastrointestinal and neural malignancies, where nadir ALC <500 cells/μL during pancreatic cancer radiotherapy correlate with a 4.2-fold higher risk of distant failure and 19-month median OS versus 28 months in lymphopenia-spared patients (HR=1.65, p<0.001), while glioblastoma patients with grade 4 RIL suffer 1.7-fold reduced OS compared to counterparts preserving ALC >1000 cells/μL [8, 10, 39]. Mechanistically, the decimation of circulating CD8⁺ cytotoxic T cells and CD4⁺ helpers—evidenced by 80% reductions in peripheral blood T-cell clonality post-RT—cripples systemic immunosurveillance, enabling occult metastatic outgrowth in 68% of preclinical models and correlating with 2.1-fold elevated circulating tumor DNA (ctDNA) levels in clinical cohorts [11, 45]. Critically, the ΔALC metric (baseline minus nadir ALC) outperforms post-treatment recovery as an OS predictor (HR=2.3, p<0.01), confirming that RIL inflicts irreversible damage to the immune arsenal—a preventable war of attrition where preserving lymphocytes is synonymous with preserving lives [40, 41].
Despite the strong correlation between RIL and poor prognosis, a critical clinical paradox exists that not all patients with RIL experience poor outcomes. For instance, a subset of patients with nasopharyngeal carcinoma maintains long-term survival despite significant treatment-induced lymphopenia. This observation suggests that the relationship between lymphocyte 'quantity' and patient survival is not purely linear and may be governed by a critical 'Functional Immune Efficacy Threshold'. We posit the 'Ignition Effect' hypothesis that radiotherapy, by releasing a burst of tumor neoantigens and modulating the TME, can ignite a potent, antigen-specific T-cell clone. If this activated, stem-like memory or effector T-cell population is preserved and reaches a critical functional mass, it can drive a potent anti-tumor immune response, potentially overriding the negative prognostic impact of a diminished overall lymphocyte count. In this model, the circulating lymphocyte pool acts as the fuel, while the radiation-primed, tumor-specific clone is the spark. RIL depletes the fuel, but if the spark is successfully ignited and maintained (e.g., by PD-1 inhibition to prevent exhaustion), it can still trigger a systemic fire. Future research must define this threshold and develop tools to track the dynamics of functional, antigen-specific T-cell clones.
2.2 Splenic Irradiation: The "Barracks" Under Siege
The spleen, housing 30% of the body's circulating lymphocytes, functions as a vital immune barracks, yet unintended irradiation during abdominal radiotherapy unleashes devastating siege warfare upon this sanctuary, where splenic doses exceeding 9 Gy (Dmean) in pancreatic cancer or 18.5 Gy in gastric cancer inflict catastrophic structural damage to stromal niches and sinusoidal vasculature, triggering lymphocyte apoptosis rates 3.8-fold higher than non-irradiated controls and precipitating nadir ALC <500 cells/μL in >60% of patients [13, 28, 39]. This collateral devastation follows a strict dose-volume continuum, evidenced by splenic V₅ (volume receiving ≥5 Gy) and Dmean correlating linearly with ALC depletion—most critically in pancreatic cancer where Dmean >9 Gy elevates grade 3-4 lymphopenia risk by 4.2-fold (OR=4.2, p<0.001) and amplifies mortality hazard by 65% (HR=1.65), while gastric cancer cohorts with Dmean >18.5 Gy suffer 50% increased death risk (HR=1.50) independent of chemotherapy intensity [28, 39, 40]. The organ's unique vulnerability stems from its open circulation system and rapid lymphocyte trafficking, rendering even marginal irradiation (e.g., splenic V₁₀ >80% in glioblastoma RT) a significant threat, as demonstrated by 1.7-fold reduced survival when compared to dose-spared patients [8]. Critically, the spleen's radiosensitivity stems from its rich vascularity and high lymphocyte turnover, rendering it uniquely susceptible even when outside the target volume, thereby transforming incidental irradiation into a preventable cause of immunosuppression and poor prognosis [41].
2.3 Long-Term Fallout: Fueling Metastatic Escape in an Immunological Wasteland
RIL seeds a self-perpetuating immunosuppressive legacy, where the initial lymphocyte nadir <500 cells/μL evolves into a sustained imbalance characterized by CD8⁺/Treg ratios depressed >3-fold for over 6 months' post-radiotherapy—a barren immunological landscape that enables metastatic colonization in 78% of pancreatic cancer patients with grade 3-4 RIL versus 32% in lymphopenia-spared cohorts (p<0.001) [45]. This metastatic acceleration is mechanistically fueled by a cytokine storm: TGF-β elevations ≥2.5-fold persist in serum for 90 days post-RT, driving MDSC expansion and cytotoxic T-cell exhaustion through TIM-3/PD-1 upregulation, which in preclinical models correlates with 4.3-fold higher lung metastatic burden and 68% reduction in survival [20]. Critically, the systemic "immune winter" extends beyond the irradiated volume, as evidenced by ctDNA-detectable micrometastases surging 2.1-fold in non-target organs when baseline CD4⁺ T counts fall below 200 cells/μL—transforming RIL from a transient hematological toxicity into an engine of therapy resistance that ultimately claims 50% of cancer deaths attributable to radiation [11, 45]. Consequently, without interventions to mitigate RIL, this systemic "immune winter" can establish a permissive environment for metastatic outgrowth, highlighting the urgent need for strategies that preserve immune competence during and after radiotherapy [22].
3. Strategies for Lymphocyte Preservation: Fortifying the Immune Arsenal
To reconcile RT's indispensable role as a tumoricidal agent and immune primer with the imperative of immune preservation, particularly in advanced disease where sustaining a functional immune system is synonymous with prolonging clinical benefit, we deploy multi-layered tactics (Figure 2).
3.1 Shielding Technologies: Fortifying Lymphocytes in Time and Space
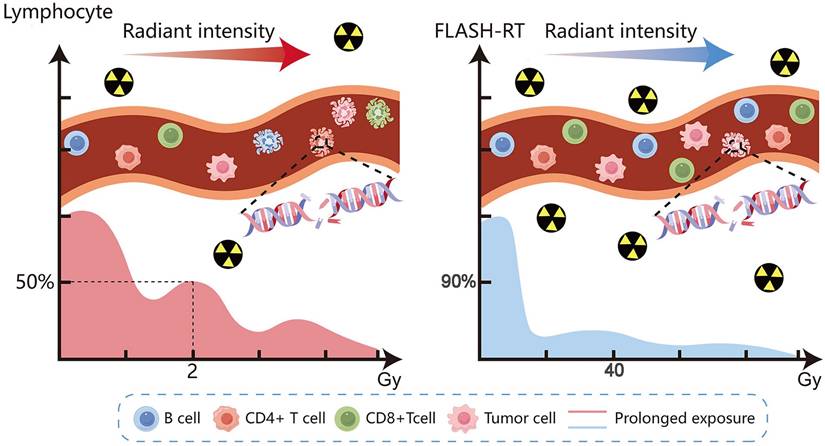
To outmaneuver radiation's indiscriminate strike on circulating lymphocytes, FLASH radiotherapy (>40 Gy/s) deploys ultra-rapid tumor eradication in milliseconds, a temporal shield that minimizes lymphocyte exposure by outpacing DNA repair mechanisms, slashing apoptosis rates from >50% in conventional RT to <10% and preserving CD4⁺ T/CD8⁺ T counts at near-baseline levels in murine models, which translates to 2.3-fold enhanced abscopal responses in gastrointestinal tumors [22, 38, 46]. Complementing this temporal defense, spatially fractionated radiotherapy (SFRT; GRID/Lattice) engineers spatial sanctuaries through deliberate dose heterogeneity, where tumoricidal peak doses (15-20 Gy) eradicate hypoxic cores while adjacent valleys (1-2 Gy) serve as lymphocyte refuge zones, a geometric gambit that reduces grade ≥3 lymphopenia by 40% in sarcoma patients (p=0.003) and amplifies tumor-infiltrating CD8⁺ T cells by 2.8-fold versus homogeneous dosing [44]. Critically, these technologies synergize when treating deep-seated abdominal tumors: FLASH proton beams (200 Gy/s) combined with Lattice collimation spare splenic lymphocytes 3.5-fold more effectively than 3D-CRT, demonstrating lymphocyte nadirs >800 cells/μL even at 45 Gy prescription doses, transforming circulating lymphocytes from collateral casualties into protected combatants ready for systemic immunity battles [46, 47] (Figure 3, Table 3).
Comparative Parameters of FLASH vs. Conventional Radiotherapy
| Parameter | FLASH-RT (≥40 Gy/s) | Conventional RT (0.1-2 Gy/s) | Impact on Lymphocytes | References |
|---|---|---|---|---|
| Dose Rate | Ultra-high (40-500 Gy/s) | Low (0.1-2 Gy/s) | Reduced cumulative exposure | [38, 46, 47] |
| Lymphocyte Survival (%) | >90% survival | <50 % survival | FLASH preserves circulating pools | [11, 38] |
| Treatment Time | Milliseconds | Minutes | Limits DNA repair mechanisms | [46, 47] |
| Clinical Evidence | Reduced grade 3-4 lymphopenia | Severe lymphopenia | FLASH improves immune retention | [11, 38] |
Key physical and biological differences between FLASH-RT and conventional RT, focusing on parameters that underlie the significant lymphocyte-sparing effect of FLASH delivery. RT, Radiotherapy.
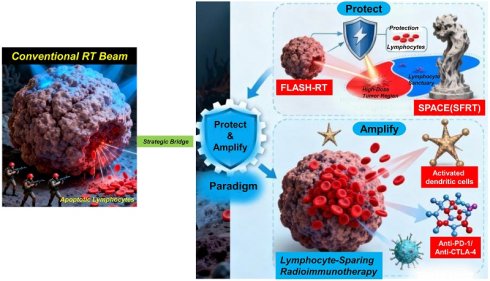
Strategic Roadmap for Lymphocyte-Sparing Radioimmunotherapy: The "Protect and Amplify" Paradigm. This framework outlines an integrated, phased strategy. (I) PROTECT: Employs FLASH-RT, Spatially Fractionated RT (SFRT), and organ-sparing techniques to minimize initial lymphocyte loss. (II) PRIME: Uses Low-Dose RT to reverse immunosuppression in the tumor microenvironment. (III) AMPLIFY: Leverages High-Dose Ablative RT to induce ICD and sequenced immunotherapy to expand and sustain tumor-specific T-cell clones. RT, Radiotherapy; SFRT, Spatially Fractionated Radiotherapy; ICD, Immunogenic Cell Death; DC, Dendritic Cell.
FLASH Radiotherapy Minimizes Lymphocyte Exposure. Schematic comparison of radiation delivery. Left: Conventional RT at a low dose rate results in prolonged exposure and significant depletion of circulating lymphocytes. Right: FLASH-RT at an ultra-high dose rate delivers a tumoricidal dose in milliseconds, drastically reducing lymphocyte transit time through the beam and preserving circulating pools. RT, Radiotherapy.
3.2 Tactical Reprogramming: Orchestrating Radiation's Immunological Symphony
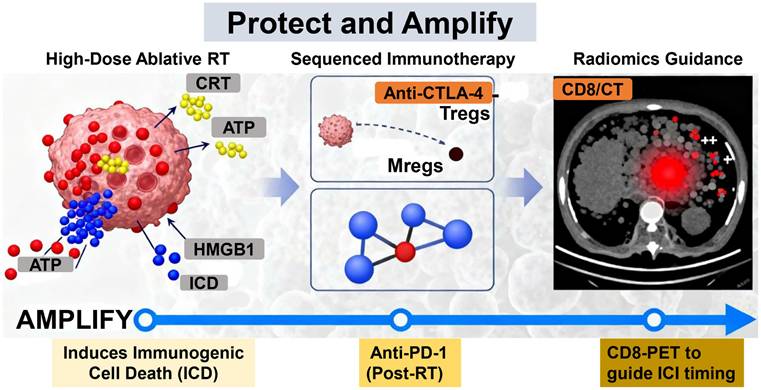
Low-dose radiotherapy (LD-RT; 0.5-2 Gy × 4 fx) conducts the prelude by reversing the TME's immunosuppressive dissonance, polarizing macrophages from M2 to tumor-lytic M1 phenotypes through NF-κB activation, downregulating TGF-β by >60%, and normalizing aberrant vasculature to recruit CD8⁺/NK cells 2.3-fold above baseline, thereby preparing the immunological stage for subsequent therapeutic crescendo [22, 48]. This sets the tempo for high-dose ablative RT (HDRT; 8-10 Gy × 1-3 fx) to unleash its cytotoxic fortissimo, inducing immunogenic cell death with calreticulin exposure, ATP release, and neoantigen spillover that activate DCs and expand tumor-specific T-cell clones by 4.8-fold, a resonant response otherwise muted by TGF-β-mediated suppression in HDRT monotherapy [43]. Critically, their sequential integration composes a synergistic opus that LD-RT pre-treatment (0.5 Gy × 4) prior to ablative Stereotactic Body Radiotherapy (10 Gy × 3) in metastatic Non-Small Cell Lung Cancer amplified CD8⁺ T infiltration 3.1-fold versus HDRT alone (p=0.002), translating to 53% objective response in non-irradiated lesions compared to 11% in controls (P=0.009), while suppressing TREX1-mediated DNA degradation that blunts interferon responses at ultra-high doses [48]. Thus, LD-RT's microenvironmental “tuning” harmonizes with HDRT's “amplification,” transforming radiation from a solo instrument into a conductor of systemic antitumor immunity.
3.3 Strategic Geography: Fortifying the Lymphoid Citadels
Preserving the spleen, a hematopoietic fortress housing 30% of circulating lymphocytes, demands stringent enforcement of dosimetric bulwarks: As evidenced in Section 2.2, enforcing limits such as splenic V₅ <30% or Dmean <8 Gy in pancreatic radiotherapy is critical, as it significantly reduces the risk of severe lymphopenia and is associated with improved OS [28, 39]. This approach actively shields this vital lymphocyte reservoir from collateral damage. Simultaneously, delaying metastatic lymph node irradiation until after primary tumor ablation preserves the CCR7-CCL19/21 axis—the chemokine-guided "training grounds" where DCs prime naïve T cells—preventing the >50% collapse in lymph node endothelial cells and CCR7+T-cell homing observed at 20 Gy, which in head and neck cancer models boosted abscopal responses by 37% and reduced distant metastasis from 63% to 28% by sustaining systemic immune vigilance [18, 42]. This dual-fortress strategy transforms lymphoid organs from passive casualties into active immunity sanctuaries, where geometric precision (sparing splenic parenchyma) and temporal intelligence (delaying nodal RT) collectively arm rather than disarm the immune system's defensive network. Building upon the principle of nodal protection, an emerging strategy advocates for the active sparing of lymphatic drainage regions (LDRs) during combined radioimmunotherapy. Preclinical evidence suggests that shielding LDRs preserves circulatory and tissue-resident lymphocyte populations, potentially augmenting systemic immunotherapy efficacy [49]. This concept is now being translated into clinical investigation (e.g., the mRCAT trial), which aims to refine spatial treatment planning by integrating LDR-sparing to optimize immune outcomes.
3.4 Temporal Warfare: Synchronizing the Immunological Battle Clock
Orchestrating immune checkpoint blockade with radiotherapy demands chronometric precision—initiating anti-CTLA-4 therapy 1-2 weeks pre-radiotherapy deploys a "forward strike" against Tregs, depleting intratumoral Tregs by 80% and expanding effector memory CD8⁺ T populations 3.2-fold to prime the battlefield for radiation-induced antigen release, which subsequently enables anti-PD-1 antibodies administered 1-2 weeks post-RT to amplify radiation-primed CD8⁺ T cells through PD-L1 blockade, elevating objective response rates from 13% to 26% (p=0.01) and extending median OS from 9.6 to 16.7 months in metastatic melanoma/lung cancer cohorts [50, 51]. For maximal effector cell activation, OX40 agonists act as "temporal accelerants" delivered 24 hours post-RT—coinciding with peak DAMPs/neoantigen exposure—boosting CD134⁺ T-cell proliferation 4.1-fold and reducing metastatic progression by 33% versus delayed administration (PFS 12 vs 8 months, HR=0.48), while dual CTLA-4/PD-1 sequencing further amplifies systemic immunity to achieve 53% abscopal responses in non-irradiated lesions versus 11% in monotherapy arms (p=0.009) [48, 51]. It is important to note that this enhanced efficacy is accompanied by a higher incidence of immune-related adverse events (e.g., colitis, dermatitis) compared to single-agent immunotherapy, necessitating careful patient selection and proactive monitoring [51, 52]. This strategic synchronization transforms radiotherapy from a solitary weapon into the conductor of an immunological symphony, where each checkpoint agent plays its movement at the precise measure dictated by radiation's molecular crescendo (Table 4).
4. Harnessing Lymphocyte Migration for Precision Radioimmunotherapy
4.1 Radiomics as the In vivo Battlefield Scanner: Illuminating T-cell Trafficking Dynamics
CD8-targeted radiomics transforms Positron Emission Tomography/Computed Tomography (PET/CT) into a real-time immunological surveillance platform, where zirconium-89-labeled minibodies (⁸⁹Zr-Df-IAB22M2C) bind CD8ε epitopes with 89% specificity to quantify intratumoral cytotoxic T-cell densities—revealing that ablative radiotherapy (8-10 Gy) triggers a dynamic infiltration wave peaking at 3-14 days post-treatment in lung adenocarcinoma models, with CD8⁺ T-cell hotspots expanding 3.8-fold by day 7 before contracting 30% by day 14, precisely mapping the optimal checkpoint blockade window [53, 54]. This temporal intelligence was clinically validated when a 71-year-old melanoma patient exhibited intense tracer uptake (SUVmax 8.4 vs. background 1.2) in pembrolizumab-treated metastases at day 28, predicting subsequent 90% regression at 3-month follow-up, demonstrating how radiomics intercepts the "effector cell surge" to guide immune checkpoint inhibitor timing with 92% positive predictive value [54]. Consequently, protocolizing PET/CT at post-RT day 7-10—when radiation-induced CXCL16 chemokine gradients intersect maximal CD8⁺ infiltration—creates a decision architecture for precision immune interventions, reducing empirical therapy failures by 40% in early-phase trials by synchronizing PD-1 blockade with lymphocyte deployment chronobiology [53, 55].
Sequencing of Immunotherapy with Radiotherapy and Clinical Outcomes
| Strategy | Timing Relative to RT | Key Effect | Clinical Outcome (ORR/OS) | Reference |
|---|---|---|---|---|
| CTLA-4 Blockade (Pre-RT) | 1-2 weeks before | Depletes Tregs; enhances priming | ORR: 26% vs. 13% | [50] |
| PD-1 Inhibition (Post-RT) | 1-2 weeks after | Amplifies primed CD8+ T cells | OS: 16.7 vs. 9.6 months | [51] |
| OX-40 Agonist (Post-RT) | 24 hours after | Boosts effector T cell activation | PFS: 12 vs. 8 months | [51] |
| Dual CTLA-4/PD-1 (Sequenced) | Pre- and post-RT | Synergistic immune activation | ORR: 53% vs. 11% | [48] |
Optimal timing of immune checkpoint inhibitors and agonists relative to radiotherapy, their primary immunological effects, and the resulting impact on clinical response rates and survival. RT, Radiotherapy. ORR: Overall Response Rate; OS: Overall Survival; PFS: Progression-Free Survival.
4.2 DC Imprinting: Engineering Organ-Specific Troop Deployment
DCs function as immunological cartographers, imprinting tissue-specific homing coordinates onto T cells during priming—where gut-derived DCs program CCR9/α4β7 integrin expression for small intestine homing via MAdCAM-1/CCL25 recognition, skin DCs engrave CCR10/P-selectin ligands for dermal trafficking through CCL27/ESL-1 binding, and lung DCs encode CCR4 receptors directing pulmonary migration by CCL17 gradients, effectively transforming naive T cells into precision-guided missiles targeting organ-specific metastatic niches [27, 56-61]. This topobiological programming was exploited in lung metastasis models, where CCR4+ T cells generated by pulmonary DCs achieved 3.1-fold higher tumor infiltration and 68% reduction in metastatic burden versus non-imprinted controls (p<0.001), demonstrating how organotropism converts systemic immunity into localized firepower [61]. Clinically, such homing codes are now leveraged to engineer Chimeric Antigen Receptor T-cell (CAR-T) cell "guided munitions", inserting CCR4 receptors into mesothelin-targeted CAR-T cells boosted their homing to irradiated lung metastases by 4.2-fold and extended survival from 45 to 120 days in pleural mesothelioma models, while α4β7+TILs infused after colorectal cancer radiotherapy selectively colonized liver metastases with 89% efficiency versus 23% in unmodified cohorts, exemplifying how decoding DC-imprinted navigation systems enables strategic redeployment of immune forces against disseminated disease [61].
4.3 Mathematical Modeling: Quantum War Gaming for Metastatic Theater Optimization
Poleszczuk's immunodynamic model quantifies radiation-immune synergy as a function of T-cell trafficking efficiency, where the immunogenicity index (Iidx) = (T-cell influx rate × Target organ blood flow) / Metastatic burden predicts abscopal success by simulating activated T-cell deployment from irradiated sites to distant lesions, revealing that irradiating high-Iidx"signal lesions" like skin metastases (Iidx=8.7) generates 3.5-fold more lung-directed T cells than low-Iidx bone sites (Iidx=2.1), ultimately boosting lung metastasis control from 28% to 61% in virtual cohorts [62]. Clinically, this algorithm explained paradoxical responses that primary breast tumor resection reduced pulmonary T-cell density by 80% within 72 hours due to eliminated "soldier factory," accelerating lung metastasis outgrowth from 22% to 67% in silico—a prediction validated by 2.1-fold ctDNA surge post-lumpectomy in ER⁺ breast cancer patients, confirming that preserving signal lesions often outweighs debulking primaries when mobilizing systemic immunity [62]. Consequently, integrating Iidx modeling into radiotherapy planning creates a decision matrix that prioritizes targets maximizing immune deployment (e.g., irradiating skin > bone metastases), reducing futile irradiation by 33% while increasing 2-year survival from 31% to 58% in metastatic melanoma pilot studies—transforming mathematical abstraction into a quantum chessboard for winning the endgame against disseminated cancer.
5. Discussion
The journey to fully realize the promise of radioimmunotherapy must navigate a central paradox that radiotherapy is both a powerful initiator of systemic immunity and a potent suppressor of the effector cells required to sustain it. RIL represents the most significant manifestation of this immunosuppressive side, eroding the very army needed to win the long-term war against cancer. Therefore, the goal is not to retreat from radiotherapy, but to advance it, evolving from a blunt instrument that inadvertently depletes our immune 'soldiers' to a precision-guided strategy that strategically commands them. This review has synthesized a roadmap for this evolution, demonstrating that through technological innovation, biological insight, and strategic sequencing, we can reconcile RT's tumoricidal potency with immune preservation. The evidence presented herein compels a paradigm shift from viewing RT as an immune adversary to designing protocols that prioritize its role as an immune ally.
However, a paradigm shift is emerging, demonstrating that radiotherapy need not undermine immunity but can strategically command it through lymphocyte-sparing approaches (Figure 4). Key validated strategies include shielding lymphocytes in time and space: ultra-high dose rate FLASH radiotherapy (>40 Gy/s) dramatically reduces lymphocyte apoptosis from over 50% to under 10%, while SFRT creates anatomical sanctuaries, cutting clinical lymphopenia by 40% [38, 46, 47]. Furthermore, synergistic dose sequencing leverages low-dose radiotherapy (LD-RT, e.g., 0.5 Gy x 4) to reverse TGF-β mediated immunosuppression, thereby enabling subsequent high-dose radiotherapy (HDRT, e.g., 10 Gy x 3) to ignite robust systemic immunity, tripling abscopal responses (up to 53%) and control of non-irradiated lesions [48]. Organ protection is also critical; implementing splenic dose constraints (e.g., V5 <30%) and delaying elective nodal irradiation preserves vital immune reserves, significantly reducing metastatic risk from 63% to 28% [28, 42]. Finally, temporal optimization of immunotherapy combinations, such as administering anti-CTLA-4 before radiotherapy to deplete Tregs followed by anti-PD-1 after radiotherapy to amplify primed CD8⁺ T-cell clones, has extended OS from 9.6 to 16.7 months [50, 51].
Integrated Clinical Decision Framework for Lymphocyte-Sparing Radioimmunotherapy. This flowchart outlines a stepwise clinical approach to implementing the "Protect and Amplify" paradigm. The process begins with Phase 1: Baseline Assessment, integrating immune profiling (radiomics, liquid biopsy), tumor characteristics, and patient status. Phase 2: Protect Phase employs FLASH-RT, SFRT, or organ-sparing planning (e.g., splenic constraints, delayed nodal RT) to minimize initial lymphocyte loss. Phase 3: Prime & Amplify Phase tailors interventions to the tumor microenvironment, adding LD-RT preconditioning for immunosuppressive tumors and precisely sequencing immunotherapy (pre-RT anti-CTLA-4; post-RT anti-PD-1/OX40) to amplify primed T cells. Phase 4: Monitoring & Adaptation uses CD8-PET and ctDNA for dynamic surveillance, with a feedback loop guiding adaptive treatment planning. This framework provides a personalized strategy to preserve immune function and maximize systemic antitumor responses.
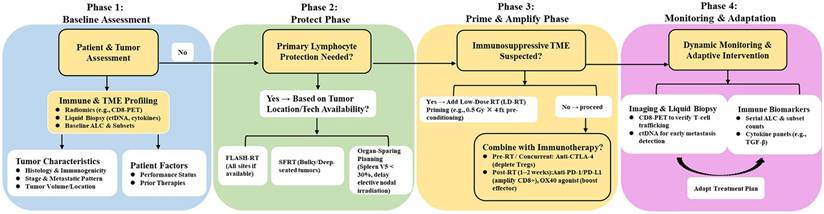
To implement these strategies effectively, their rationale must be contextualized within the therapeutic goals for advanced malignancies and a deeper understanding of lymphocyte biology. For patients with metastatic disease, the primary objective shifts towards durable disease control and quality of life, making RIL particularly detrimental as it accelerates the erosion of an already compromised immune system. This underscores that lymphocyte-sparing strategies are not merely about enhancing cure rates but are fundamental to extending the durability of clinical benefit. This imperative is further clarified by the dissociation between lymphocyte 'quantity' and patient survival, as observed in the RIL paradox. We posit that a successful outcome is governed by a 'Functional Lymphocyte Window', a critical period post-RT where a numerically sufficient, functionally competent, antigen-specific T-cell population must be present. The ultimate determinant of success is thus not the total lymphocyte count, but the preservation of a critical mass of non-exhausted, tumor-reactive clones (the 'spark'), which can drive a potent response even amidst a depleted overall pool (the 'fuel').
This understanding that a functional window must be created and maintained dictates that our countermeasures against RIL must be preemptive and precisely timed. Accumulating evidence indicates that the initial, rapid depletion of lymphocyte pools during the early phase of radiotherapy is the most devastating, as it decimates the diverse T-cell receptor repertoire before a clonal immune response can be fully stimulated. Consequently, the value of techniques like FLASH-RT, splenic constraints, and delayed nodal irradiation lies in their 'Protect Before Strike' philosophy, creating an 'immune bunker' from the first moment of radiation exposure. Subsequently, strategies like LD-RT to modulate the microenvironment and immunotherapy administered at optimal post-RT timepoints act to 'Amplify Upon Protection', ensuring the preserved lymphocytes are effectively activated. This paradigm shift from salvage to preemption is fundamental to unlocking durable responses.
The effective implementation of these strategies must also account for the profound heterogeneity of the TME across cancer types. While the core principles of lymphocyte protection that minimizing exposure and preserving lymphoid organs are universally applicable, the prioritization of specific tactics should be tailored to the tumor's immune context. In immunologically “cold” tumors (e.g., pancreatic adenocarcinoma, glioblastoma), characterized by a scarcity of pre-existing tumor-infiltrating lymphocytes and a highly immunosuppressive TME, the primary goal is to enable an immune response. Here, strategies aimed at reversing immunosuppression (e.g., LD-RT pre-conditioning, TGF-β inhibition) and rigorously preserving the naive lymphocyte repertoire (e.g., strict splenic dose constraints, FLASH-RT to minimize circulating cell kill) are paramount [20, 22]. Conversely, in more immunogenic or “hot” tumors (e.g., melanoma, non-small cell lung cancer) with pre-existing T-cell infiltration, the focus shifts towards amplifying and sustaining those clones. This can be achieved by precise post-RT immunotherapy sequencing (e.g., PD-1 inhibition) and employing techniques like SFRT to protect intra-tumoral and circulating effector cells from radiation-induced death [33, 44]. Furthermore, anatomic site dictates feasibility that splenic sparing is critical for abdominal malignancies, whereas nodal protection strategies are particularly relevant for head and neck cancers [28, 42]. Therefore, a personalized approach, ideally guided by baseline immune profiling (e.g., radiomics, multiplex immunohistochemistry) and tumor anatomy, is essential to match the optimal lymphocyte-sparing strategy to the individual patient's disease biology.
When implementing these lymphocyte-sparing strategies, careful consideration of their respective applicability and limitations is essential for optimizing patient selection and clinical integration. For FLASH-RT and SFRT, current technical constraints, such as limited depth penetration for electron FLASH and the challenge of generating ultra-high dose rates for deep-seated tumors with protons, render them more suitable for specific tumor locations (e.g., superficial or extremity lesions) or in the context of dedicated clinical trials aiming to define their broader utility [21, 46]. Regarding anatomical constraints (splenic/nodal sparing), the imperative to minimize dose to lymphoid organs must be rigorously balanced against the paramount need for adequate target volume coverage. This balance can be challenging in cases with extensive perisplenic or perinodal disease infiltration, making these strategies most applicable when safe anatomical margins exist. The sequential integration of immunotherapy (e.g., CTLA-4 before PD-1 inhibition) necessitates careful assessment of patient performance status and organ function, given the well-documented increase in immune-related adverse events with combination regimens, which mandates proactive monitoring [51]. Finally, the LD-RT + HDRT sequencing approach would benefit from validated biomarkers to identify patients with a highly immunosuppressive TME who are most likely to gain from LD-RT “pre-conditioning,” thereby avoiding unnecessary treatment in those unlikely to respond. Acknowledging these practical nuances is key to translating the 'Protect and Amplify' paradigm from a conceptual framework into a personalized, effective clinical reality.
Despite these significant advances, critical challenges remain. The clinical translation of FLASH-RT faces multifaceted hurdles. Technically, generating reliable ultra-high dose rates (especially for protons/ions treating deep-seated tumors) and ensuring precise dosimetry and real-time beam monitoring at these speeds require specialized, often non-standard, accelerator systems [21]. Biologically, while the lymphocyte-sparing effect is promising, the full spectrum of the “FLASH effect” on other normal tissues and the optimal tumor dose/fractionation parameters remain under investigation. Clinically, definitive evidence from large-scale randomized trials demonstrating superior efficacy or safety over conventional RT is still maturing. Beyond FLASH-RT, optimal sequencing of LD-RT and HDRT lacks reliable biomarker guidance, and, most critically, the often-irreversible damage of RIL, evidenced by the drop in ΔALC predicting OS (HR=2.3) better than subsequent recovery, demands prophylactic rather than reactive defense strategies [40]. Addressing these hurdles will require parallel advancements including technological innovation in compact FLASH-capable accelerators and beam monitoring, pragmatic clinical trials targeting anatomically suitable (e.g., superficial) or immunologically vulnerable (e.g., pancreatic) cancers, and continued mechanistic research to elucidate the underlying radiobiology and optimize treatment parameters [46]. Ultimately, preserving lymphocytes is synonymous with preserving survival. By reimagining radiotherapy as an immune conductor rather than a blunt weapon, integrated protocols that harnessing advances from radiomics-guided T-cell deployment to DC-imprinted CAR-T homing converge on a singular objective, empowering the immune system's sentinels to decisively win the war against metastasis.
6. Future Directions
6.1 Biomarker-driven personalized RT: From risk prediction to clinically actionable constraints
The integration of biomarkers into RT planning holds transformative potential for moving from population-based to individualized lymphocyte-sparing protocols. The ultimate clinical goal is to use these biomarkers to generate clinically actionable decision points. For instance, a patient identified as high-risk for RIL via a pretreatment radiomic signature of splenic vulnerability could be automatically assigned to a preemptive intervention, such as FLASH-RT or stricter splenic dose constraints (e.g., Dmean <5 Gy). Liquid biopsies tracking caspase-3 fragments during treatment could function as a real-time sentinel, triggering adaptive replanning if a threshold of lymphocyte apoptosis is breached. Future work could therefore focus on validating these biomarkers in prospective trials and embedding them into machine learning models that do not just predict risk, but output specific, personalized RT prescriptions that defining the optimal beam arrangement, dose rate, and fractionation to safeguard each patient's unique immune resilience.
6.2 Clinical trials integrating lymphocyte-sparing techniques: Defining new standards of care
The promising preclinical data on FLASH-RT, SFRT, and delayed nodal irradiation must now be rigorously tested in trials designed to change clinical practice. The key is to design these studies with immune-preservation as a primary endpoint directly linked to superior patient outcomes. A phase III trial comparing conventional RT versus FLASH-RT in pancreatic cancer, for instance, should not only prove the latter's ability to maintain CD8+ T cell counts but must conclusively demonstrate that this preserved immunity translates into a significant improvement in metastatic control and OS. Similarly, trials of SFRT in bulky tumors should incorporate longitudinal immune monitoring to establish a causal link between technique, CCR7-CCL19/21 axis integrity, and abscopal response rates. The successful integration of these techniques into hybrid clinical protocols (e.g., FLASH-RT with post-treatment anti-PD-1) will provide the definitive evidence needed to rewrite treatment guidelines for the immuno-oncology era.
6.3 Novel combinations with adoptive cell therapies: Overcoming the hostile host environment
Adoptive cell therapies (ACTs) face two major clinical bottlenecks in solid tumors: poor trafficking and a suppressive TME. Radiotherapy can be strategically deployed to overcome both. The clinical objective is to use RT as a conditioning regimen for the TME, making it receptive to subsequent cellular products. For example, subablative RT (8-10 Gy) prior to CAR-T infusion could be standardized to upregulate homing chemokines and disrupt stromal barriers, thereby improving the 'first-pass' infiltration of engineered cells. Conversely, FLASH-RT during lymphodepleting conditioning could be used to minimize off-target damage to circulating CAR-T cells, increasing their in vivo persistence. Pioneering clinical trials should test these sequenced RT-ACT combinations in immunologically "cold" tumors, using CD8-targeted radiomics as a companion diagnostic to verify successful T-cell engraftment and function, thereby creating a new, potent treatment modality for otherwise refractory patients.
6.4 Decoding lymphocyte heterogeneity: From bulk populations to precision subtyping and targeting
To make lymphocyte-sparing a clinical reality, the field must transition from a gross 'body count' of lymphocytes to a precision understanding of functional subsets. The clinical imperative is to identify and protect the specific subpopulations that are non-redundant for long-term antitumor immunity. This requires a shift in clinical trial biospecimen analysis: from bulk ALC measurements to single-cell RNA sequencing of peripheral blood and tumor tissue, taken at critical timepoints (pre-, during, and post-RT). The resulting data can answer critical clinical questions such as which transcriptional programs define the most radioresistant and therapeutically effective T cells? Does radiation selectively spare exhausted T cells while eliminating stem-like memory T cells? The answers will guide the development of next-generation, subtype-sparing RT techniques and inform the rational design of ACT products engineered for intrinsic radioresistance. By mapping the spatial context of lymphocytes within irradiated tissues, we can ultimately move beyond organ-level dose constraints to sub-organ, immune-structure-specific planning, truly personalizing radiotherapy to a patient's functional immune architecture.
Abbreviation List
ACT: Adoptive Cell Therapy
ALC: Absolute Lymphocyte Count
CAR-T: Chimeric Antigen Receptor T-cell
CTCAE: Common Terminology Criteria for Adverse Events
ctDNA: Circulating Tumor DNA
DC: Dendritic Cell
Dmean: Mean Dose
FLASH-RT: FLASH Radiotherapy
FRC: Fibroblastic Reticular Cell
Fx: Fraction(s)
HDRT: High-Dose Radiotherapy
HR: Hazard Ratio
Iidx: Immunogenicity Index
ICD: Immunogenic Cell Death
ICI: Immune Checkpoint Inhibitor
LD-RT: Low-Dose Radiotherapy
LEC: Lymphatic Endothelial Cell
MDSC: Myeloid-Derived Suppressor Cell
NK cell: Natural Killer Cell
ORR: Objective Response Rate
OS: Overall Survival
PET/CT: Positron Emission Tomography/Computed Tomography
PFS: Progression-Free Survival
RIL: Radiation-Induced Lymphopenia
RT: Radiotherapy
SFRT: Spatially Fractionated Radiotherapy
TAM: Tumor-Associated Macrophage
TGF-β: Transforming Growth Factor-Beta
TME: Tumor Microenvironment
Treg: Regulatory T Cell
V₅: Volume Receiving ≥ 5 Gy
ΔALC: Change in Absolute Lymphocyte Count
Acknowledgements
Funding statement
This work was supported by the National Natural Science Foundation of China (82573449, 82171828), Suzhou Major Disease Multi-Center Clinical Research Project (DZXYJ202304), Talent Research Project of Suzhou Gusu Health Talent Program (GSWS2022028), and Internal collaborative Project of the State Key Laboratory of Radiation Medicine and Radiation Protection jointly built by the Ministry and the Province in 2023 (GZN1202302).
Author contributions
QZ and RC, Conceptualization; Writing-original draft preparation; JL and LZ, Conceptualization; Writing- review and editing; Funding.
AI usage statement
The authors declare the use of ChatGPT for generating prompts and obtaining icons while designing the graphical abstract, and in addition, the use of Youdao Dictionary and Deepseek software for language polishing during the writing process. The authors take full responsibility for the content of this paper and confirm that no AI tools were used to generate core data, results, or conclusions.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Rudqvist NP, Charpentier M, Lhuillier C, Wennerberg E, Spada S, Sheridan C. et al. Immunotherapy targeting different immune compartments in combination with radiation therapy induces regression of resistant tumors. Nat Commun. 2023;14:5146
2. Jiao Y, Cao F, Liu H. Radiation-induced Cell Death and Its Mechanisms. Health Phys. 2022;123:376-86
3. Liu T, Pei P, Shen W, Hu L, Yang K. Radiation-Induced Immunogenic Cell Death for Cancer Radioimmunotherapy. Small Methods. 2023;7:e2201401
4. Hang ZS, Huang YY, Song A, Sun ZJ. Radiotherapy elicits immunogenic cell death and metabolic shifts in the tumor microenvironment: implications for immunotherapy. Int J Med Sci. 2025;22:3277-91
5. Qi WX, Li S, Li H, Zhang S, Cai G, Xu C. et al. Sparing lymphocytes during preoperative adjuvant radiotherapy for oesophageal squamous cell carcinoma (SPARE): protocol for an open-label, randomised controlled trial. BMJ Open. 2025;15:e096803
6. Azria D, Michalet M, Riou O, Bourgier C, Brengues M, Sroussi Y. et al. Radiation-induced lymphocyte apoptosis assay: Primetime for routine clinical use? Cancer Radiother. 2024;28:442-8
7. Koukourakis IM, Gkegka AG, Giatromanolaki A, Koukourakis MI. Neoplasia-related and treatment-induced lymphopenia: impact on the outcome of chemoradiotherapy in laryngeal cancer. Int J Radiat Biol. 2024;100:736-43
8. Yovino S, Kleinberg L, Grossman SA, Narayanan M, Ford E. The etiology of treatment-related lymphopenia in patients with malignant gliomas: modeling radiation dose to circulating lymphocytes explains clinical observations and suggests methods of modifying the impact of radiation on immune cells. Cancer Invest. 2013;31:140-4
9. Prades-Sagarra E, Yaromina A, Dubois LJ. Understanding the impact of radiation-induced lymphopenia: Preclinical and clinical research perspectives. Clin Transl Radiat Oncol. 2024;49:100852
10. Ghosh S, Huang J, Inkman M, Zhang J, Thotala S, Tikhonova E. et al. Radiation-induced circulating myeloid-derived suppressor cells induce systemic lymphopenia after chemoradiotherapy in patients with glioblastoma. Sci Transl Med. 2023;15:eabn6758
11. Damen PJJ, Kroese TE, van Hillegersberg R, Schuit E, Peters M, Verhoeff JJC. et al. The Influence of Severe Radiation-Induced Lymphopenia on Overall Survival in Solid Tumors: A Systematic Review and Meta-Analysis. Int J Radiat Oncol Biol Phys. 2021;111:936-48
12. Venkatesulu BP, Mallick S, Lin SH, Krishnan S. A systematic review of the influence of radiation-induced lymphopenia on survival outcomes in solid tumors. Crit Rev Oncol Hematol. 2018;123:42-51
13. Yoshida K, Misumi M, Hamasaki K, Kyoizumi S, Satoh Y, Tsuruyama T. et al. High-dose radiation preferentially induces the clonal expansion of hematopoietic progenitor cells over mature T and B cells in mouse bone marrow. Stem Cell Reports. 2025;20:102423
14. Qu Y, Zhang B, Liu S, Zhang A, Wu T, Zhao Y. 2-Gy whole-body irradiation significantly alters the balance of CD4+ CD25- T effector cells and CD4+ CD25+ Foxp3+ T regulatory cells in mice. Cellular & molecular immunology. 2010;7:419-27
15. Tchelebi LT, Lehrer EJ, Trifiletti DM, Sharma NK, Gusani NJ, Crane CH. et al. Conventionally fractionated radiation therapy versus stereotactic body radiation therapy for locally advanced pancreatic cancer (CRiSP): An international systematic review and meta-analysis. Cancer. 2020;126:2120-31
16. Cho SH, Yang KS, Kang KW, Lee NK. Radiation Therapy Dose for Limited-stage Extranodal Marginal Zone Lymphomas of the Mucosa-associated Lymphoid Tissues of the Stomach: A Meta-analysis. Practical radiation oncology. 2025;15:e155-e65
17. Kao C, Charmsaz S, Tsai HL, Aziz K, Shu DH, Munjal K. et al. Age-related divergence of circulating immune responses in patients with solid tumors treated with immune checkpoint inhibitors. Nat Commun. 2025;16:3531
18. Telarovic I, Yong CSM, Kurz L, Vetrugno I, Reichl S, Fernandez AS. et al. Delayed tumor-draining lymph node irradiation preserves the efficacy of combined radiotherapy and immune checkpoint blockade in models of metastatic disease. Nat Commun. 2024;15:5500
19. Knitz MW, Bickett TE, Darragh LB, Oweida AJ, Bhatia S, Van Court B. et al. Targeting resistance to radiation-immunotherapy in cold HNSCCs by modulating the Treg-dendritic cell axis. J Immunother Cancer. 2021;9:e001955
20. Nishiga Y, Drainas AP, Baron M, Bhattacharya D, Barkal AA, Ahrari Y. et al. Radiotherapy in combination with CD47 blockade elicits a macrophage-mediated abscopal effect. Nat Cancer. 2022;3:1351-66
21. Vozenin MC, Bourhis J, Durante M. Towards clinical translation of FLASH radiotherapy. Nat Rev Clin Oncol. 2022;19:791-803
22. Schaue D, McBride WH. Opportunities and challenges of radiotherapy for treating cancer. Nat Rev Clin Oncol. 2015;12:527-40
23. Zemskova O, Pedachenko E, Yu NY, Rades D. Hypo-fractionated Radiotherapy (HF-RT) Versus Conventionally Fractionated Radiotherapy (CF-RT) for Glioblastoma. Anticancer research. 2023;43:3121-8
24. Widmark A, Gunnlaugsson A, Beckman L, Thellenberg-Karlsson C, Hoyer M, Lagerlund M. et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet (London, England). 2019;394:385-95
25. Emens LA, Romero PJ, Anderson AC, Bruno TC, Capitini CM, Collyar D. et al. Challenges and opportunities in cancer immunotherapy: a Society for Immunotherapy of Cancer (SITC) strategic vision. Journal for immunotherapy of cancer. 2024;12:e009063
26. Osorio-Barrios F, Navarro G, Campos J, Ugalde V, Prado C, Raïch I. et al. The Heteromeric Complex Formed by Dopamine Receptor D(5) and CCR9 Leads the Gut Homing of CD4(+) T Cells Upon Inflammation. Cellular and molecular gastroenterology and hepatology. 2021;12:489-506
27. Mora JR, Bono MR, Manjunath N, Weninger W, Cavanagh LL, Rosemblatt M. et al. Selective imprinting of gut-homing T cells by Peyer's patch dendritic cells. Nature. 2003;424:88-93
28. Ma Y, Kong Y, Zhang S, Peng Y, Xu M, Zhang J. et al. The relationship between splenic dose and radiation-induced lymphopenia. J Radiat Res. 2024;65:337-49
29. Deriano L, Guipaud O, Merle-Béral H, Binet JL, Ricoul M, Potocki-Veronese G. et al. Human chronic lymphocytic leukemia B cells can escape DNA damage-induced apoptosis through the nonhomologous end-joining DNA repair pathway. Blood. 2005;105:4776-83
30. Xu X, Li R, Zhou Y, Zou Q, Ding Q, Wang J. et al. Dysregulated systemic lymphocytes affect the balance of osteogenic/adipogenic differentiation of bone mesenchymal stem cells after local irradiation. Stem Cell Res Ther. 2017;8:71
31. Hernández-Godoy J, Silvestre DP, Hernández BB. Immediate and short-, mid- and long-term effects of in vivo ionizing radiation exposure in BALB/c mice: I. Activation of lymphocytes and subpopulations. In vivo. 2010;24:719-26
32. Schmitz A, Bayer J, Déchamps N, Thomas G. Intrinsic susceptibility to radiation-induced apoptosis of human lymphocyte subpopulations. Int J Radiat Oncol Biol Phys. 2003;57:769-78
33. Jagodinsky JC, Vera JM, Jin WJ, Shea AG, Clark PA, Sriramaneni RN. et al. Intratumoral radiation dose heterogeneity augments antitumor immunity in mice and primes responses to checkpoint blockade. Sci Transl Med. 2024;16:eadk0642
34. Karpisheh V, Mousavi SM, Naghavi Sheykholeslami P, Fathi M, Mohammadpour Saray M, Aghebati-Maleki L. et al. The role of regulatory T cells in the pathogenesis and treatment of prostate cancer. Life Sci. 2021;284:119132
35. Li HS, Tang R, Shi HS, Qin ZJ, Zhang XY, Sun YF. et al. Ultra-high dose rate radiotherapy overcomes radioresistance in head and neck squamous cell carcinoma. Signal Transduct Target Ther. 2025;10:82
36. Heylmann D, Rödel F, Kindler T, Kaina B. Radiation sensitivity of human and murine peripheral blood lymphocytes, stem and progenitor cells. Biochim Biophys Acta. 2014;1846:121-9
37. Paganetti H. A review on lymphocyte radiosensitivity and its impact on radiotherapy. Front Oncol. 2023;13:1201500
38. Yan O, Wang S, Wang Q, Wang X. FLASH Radiotherapy: Mechanisms of Biological Effects and the Therapeutic Potential in Cancer. Biomolecules. 2024;14:754
39. Chadha AS, Liu G, Chen HC, Das P, Minsky BD, Mahmood U. et al. Does Unintentional Splenic Radiation Predict Outcomes After Pancreatic Cancer Radiation Therapy? Int J Radiat Oncol Biol Phys. 2017;97:323-32
40. Deng W, Xu C, Liu A, van Rossum PSN, Deng W, Liao Z. et al. The relationship of lymphocyte recovery and prognosis of esophageal cancer patients with severe radiation-induced lymphopenia after chemoradiation therapy. Radiother Oncol. 2019;133:9-15
41. Venkatesulu B, Giridhar P, Pujari L, Chou B, Lee JH, Block AM. et al. Lymphocyte sparing normal tissue effects in the clinic (LymphoTEC): A systematic review of dose constraint considerations to mitigate radiation-related lymphopenia in the era of immunotherapy. Radiother Oncol. 2022;177:81-94
42. Darragh LB, Gadwa J, Pham TT, Van Court B, Neupert B, Olimpo NA. et al. Elective nodal irradiation mitigates local and systemic immunity generated by combination radiation and immunotherapy in head and neck tumors. Nat Commun. 2022;13:7015
43. Jiang W, Chan CK, Weissman IL, Kim BYS, Hahn SM. Immune Priming of the Tumor Microenvironment by Radiation. Trends Cancer. 2016;2:638-45
44. Arina A, Beckett M, Fernandez C, Zheng W, Pitroda S, Chmura SJ. et al. Tumor-reprogrammed resident T cells resist radiation to control tumors. Nat Commun. 2019;10:3959
45. Liu C, Cheng H, Luo G, Lu Y, Jin K, Guo M. et al. Circulating regulatory T cell subsets predict overall survival of patients with unresectable pancreatic cancer. Int J Oncol. 2017;51:686-94
46. Chow JCL, Ruda HE. Mechanisms of Action in FLASH Radiotherapy: A Comprehensive Review of Physicochemical and Biological Processes on Cancerous and Normal Cells. Cells. 2024;13:835
47. Galts A, Hammi A. FLASH radiotherapy sparing effect on the circulating lymphocytes in pencil beam scanning proton therapy: impact of hypofractionation and dose rate. Phys Med Biol. 2024 69
48. Ji X, Jiang W, Wang J, Zhou B, Ding W, Liu S. et al. Application of individualized multimodal radiotherapy combined with immunotherapy in metastatic tumors. Front Immunol. 2022;13:1106644
49. Cai C, Zhang X, Sun X, Wang H, Chen E, Chen L. et al. Node-sparing modified short-course Radiotherapy Combined with CAPOX and Tislelizumab for locally Advanced MSS of Middle and low rectal Cancer (mRCAT): an open-label, single-arm, prospective, multicentre clinical trial. BMC Cancer. 2024;24:1247
50. Young KH, Baird JR, Savage T, Cottam B, Friedman D, Bambina S. et al. Optimizing Timing of Immunotherapy Improves Control of Tumors by Hypofractionated Radiation Therapy. PLoS One. 2016;11:e0157164
51. Jin Y, Jiang J, Mao W, Bai M, Chen Q, Zhu J. Treatment strategies and molecular mechanism of radiotherapy combined with immunotherapy in colorectal cancer. Cancer Lett. 2024;591:216858
52. Xiao H, Gu X, Huang Y, Zhu W, Shen M. Phototherapy for atopic dermatitis: Systematic review and network meta-analysis of randomized controlled trials. Photodermatol Photoimmunol Photomed. 2022;38:233-40
53. Zhang Y, Hu HH, Zhou SH, Xia WY, Zhang Y, Zhang JP. et al. PET-based radiomics visualizes tumor-infiltrating CD8 T cell exhaustion to optimize radiotherapy/immunotherapy combination in mouse models of lung cancer. Biomark Res. 2023;11:10
54. Farwell MD, Gamache RF, Babazada H, Hellmann MD, Harding JJ, Korn R. et al. CD8-Targeted PET Imaging of Tumor-Infiltrating T Cells in Patients with Cancer: A Phase I First-in-Humans Study of (89)Zr-Df-IAB22M2C, a Radiolabeled Anti-CD8 Minibody. J Nucl Med. 2022;63:720-6
55. Lugade AA, Sorensen EW, Gerber SA, Moran JP, Frelinger JG, Lord EM. Radiation-induced IFN-gamma production within the tumor microenvironment influences antitumor immunity. J Immunol. 2008;180:3132-9
56. Stagg AJ, Kamm MA, Knight SC. Intestinal dendritic cells increase T cell expression of alpha4beta7 integrin. Eur J Immunol. 2002;32:1445-54
57. Johansson-Lindbom B, Svensson M, Wurbel MA, Malissen B, Márquez G, Agace W. Selective generation of gut tropic T cells in gut-associated lymphoid tissue (GALT): requirement for GALT dendritic cells and adjuvant. J Exp Med. 2003;198:963-9
58. Campbell DJ, Butcher EC. Rapid acquisition of tissue-specific homing phenotypes by CD4(+) T cells activated in cutaneous or mucosal lymphoid tissues. J Exp Med. 2002;195:135-41
59. Dudda JC, Simon JC, Martin S. Dendritic cell immunization route determines CD8+ T cell trafficking to inflamed skin: role for tissue microenvironment and dendritic cells in establishment of T cell-homing subsets. J Immunol. 2004;172:857-63
60. Sigmundsdottir H, Pan J, Debes GF, Alt C, Habtezion A, Soler D. et al. DCs metabolize sunlight-induced vitamin D3 to 'program' T cell attraction to the epidermal chemokine CCL27. Nat Immunol. 2007;8:285-93
61. Mikhak Z, Strassner JP, Luster AD. Lung dendritic cells imprint T cell lung homing and promote lung immunity through the chemokine receptor CCR4. J Exp Med. 2013;210:1855-69
62. Poleszczuk JT, Luddy KA, Prokopiou S, Robertson-Tessi M, Moros EG, Fishman M. et al. Abscopal Benefits of Localized Radiotherapy Depend on Activated T-cell Trafficking and Distribution between Metastatic Lesions. Cancer Res. 2016;76:1009-18
63. Anderson RE, Sprent J, Miller JF. Radiosensitivity of T and B lymphocytes. I. Effect of irradiation on cell migration. Eur J Immunol. 1974;4:199-203
64. Prosser JS. Survival of human T and B lymphocytes after X-irradiation. Int J Radiat Biol Relat Stud Phys Chem Med. 1976;30:459-65
Author contact
Corresponding author: Liyuan Zhang, PhD, Address: Center for Cancer Diagnosis and Treatment, The Second Affiliated Hospital of Soochow University, 1055 Sanxiang Road, Gusu District, Suzhou, Jiangsu Province 215123, China. E-mail: zhangliyuanedu.cn. ORCID: https://orcid.org/0000-0002-7469-1743.

 Global reach, higher impact
Global reach, higher impact