Impact Factor ISSN: 1449-2288
Int J Biol Sci 2021; 17(14):3968-3980. doi:10.7150/ijbs.58910 This issue Cite
Research Paper
Long non-coding RNA MEG3 promotes cisplatin-induced nephrotoxicity through regulating AKT/TSC/mTOR-mediated autophagy
1. Department of Clinical Laboratory, The Second Hospital of Shandong University, Jinan, 250000, China.
2. Department of Clinical Neuroscience, Karolinska Institutet, S-171 76, Sweden.
3. Department of Oncology, Nanfang Hospital, Southern Medical University, Guangzhou, 510515, China.
4. Department of Oncology-Pathology, Karolinska Institutet, Stockholm, Sweden.
5. Department of Cell and Molecular Biology, Karolinska Institutet, S-171 76, Sweden.
6. Key Laboratory, The Second Hospital of Shandong University, Jinan, 250000, China.
7. Department of Microbiology, Tumor and Cell Biology, Karolinska Institutet, S-171 76, Sweden.
8. Department of Nephrology, Qilu hospital of Shandong University, Jinan, China.
9. Department of Pharmacology, The Second Hospital of Shandong University, Jinan, 250000, China.
Abstract
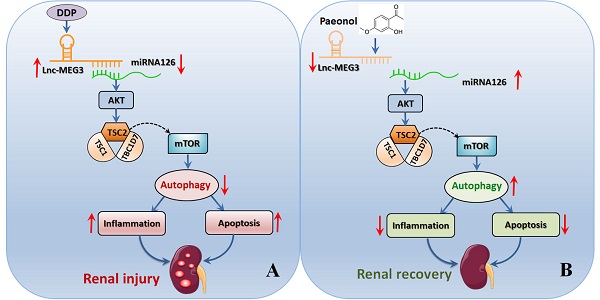
Cis-Diamminedichloroplatinum (II) (DDP)-induced nephrotoxicity (DDPIN) may cause irreversible renal injury associated with high morbidity and mortality. Current standard therapies have not achieved satisfactory clinical outcomes due to unclear molecular and cellular mechanisms. Therefore, exploring potential therapies on DDPIN represents an urgent medical need. Present study characterized the role of lncRNA maternally expressed gene 3 (lnc-MEG3) in the pathogenesis of DDPIN. In both in vitro and in murine models of DDP-induced nephrotoxicity, lnc-MEG3 exacerbated DDPIN by negatively regulating miRNA-126 subsequently causing a decreased AKT/TSC/mTOR-mediated autophagy. By silencing lnc-MEG3 or incorporating miRNA-126 mimetics, the proliferation and migration of DDP-treated cells were restored. In vivo, we identified Paeonol to alleviate DDPIN by the inhibition of lnc-MEG3. Taken together, lnc-MEG3 represents a novel therapeutic target for DDPIN and Paeonol may serve as a promising treatment by inhibiting lnc-MEG3 and its related signaling pathways.
Keywords: lncRNA MEG3, DDPIN, miRNA-126, autophagy

 Global reach, higher impact
Global reach, higher impact