Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2023; 19(13):4020-4035. doi:10.7150/ijbs.82012 This issue Cite
Research Paper
ER translocon inhibitor ipomoeassin F inhibits triple-negative breast cancer growth via blocking ER molecular chaperones
1. Cancer Centre, Faculty of Health Sciences, University of Macau, Taipa, Macau SAR, China.
2. Department of Chemistry and Biochemistry, University of Maryland, College Park, Maryland 20742, USA.
3. Shenzhen Laboratory of Tumor Cell Biology, Institute of Biomedicine and Biotechnology, Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, China.
4. Chemical Genomics Leader Research Laboratory, Department of Biotechnology, College of Life Science and Biotechnology, Yonsei University, Seoul 03722, Korea.
5. Department of Biological Sciences, University of Arkansas, Fayetteville, Arkansas 72701, USA.
6. Feinstein Institute for Medical Research, Northwell Health, 350 Community Dr., Manhasset, New York, 11030, USA.
7. Department of Chemistry, Ball State University, Muncie, Indiana 47306, USA.
8. Department of Mathematical Sciences, University of Arkansas, Arkansas 72701, USA.
9. MOE Frontiers Science Centre for Precision Oncology, University of Macau, Taipa, Macau SAR, China.
Abstract
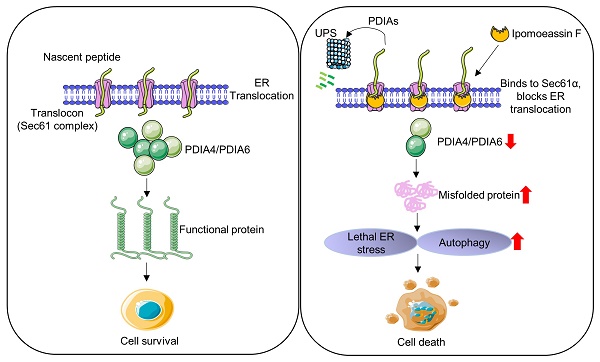
Triple-negative breast cancer (TNBC) is an aggressive type of breast cancer where no effective therapy has been developed. Here, we report that the natural product ER translocon inhibitor ipomoeassin F is a selective inhibitor of TNBC cell growth. A proteomic analysis of TNBC cells revealed that ipomoeassin F significantly reduced the levels of ER molecular chaperones, including PDIA6 and PDIA4, and induced ER stress, unfolded protein response (UPR) and autophagy in TNBC cells. Mechanistically, ipomoeassin F, as an inhibitor of Sec61α-containing ER translocon, blocks ER translocation of PDIA6, inducing its proteasomal degradation. Silencing of PDIA6 or PDIA4 by RNA interferences or treatment with a small molecule inhibitor of the protein disulfide isomerases in TNBC cells successfully recapitulated the ipomoeassin F phenotypes, including the induction of ER stress, UPR and autophagy, suggesting that the reduction of PDIAs is the key mediator of the pharmacological effects of ipomoeassin F. Moreover, ipomoeassin F significantly suppressed TNBC growth in a mouse tumor xenograft model, with a marked reduction in PDIA6 and PDIA4 levels in the tumor samples. Our study demonstrates that Sec61α-containing ER translocon and PDIAs are potential drug targets for TNBC and suggests that ipomoeassin F could serve as a lead for developing ER translocon-targeted therapy for TNBC.
Keywords: Ipomoeassin F, ER translocon, TNBC, PDIA6, PDIA4, ER stress

 Global reach, higher impact
Global reach, higher impact