Impact Factor ISSN: 1449-2288
Int J Biol Sci 2026; 22(6):3144-3173. doi:10.7150/ijbs.129709 This issue Cite
Review
Mitochondria transfer in tissue homeostasis and diseases
1. Department of Orthopaedics, Shenzhen Nanshan People's Hospital, Affiliated Nanshan Hospital of Shenzhen University, Shenzhen, Guangdong Province, China.
2. Department of Orthopaedics & Rehabilitation, Yale University School of Medicine, New Haven, USA.
3. Department of Biochemistry, Homeostatic Medicine Institute, School of Medicine, Guangdong Provincial Key Laboratory of Cell Microenvironment and Disease Research, Shenzhen Key Laboratory of Cell Microenvironment, Southern University of Science and Technology, Shenzhen, Guangdong Province, China.
#These authors contributed equally.
Received 2025-12-9; Accepted 2026-2-5; Published 2026-2-26
Abstract
Mitochondria serve as the essential powerhouse for virtually all eukaryotic cells and have been implicated in other crucial functions in both physiological and disease contexts. As cytoplasmic organelles, mitochondria are segregated and transported from parent to daughter cells during division or differentiation, a process known as vertical mitochondria transfer (VMT). A growing body of literature indicates that various cell types can export mitochondria for delivery to developmentally unrelated cell types without division, a process termed horizontal mitochondria transfer (HMT). In this review, we summarize current understanding of the modes of mitochondria transfer and illustrate the phenomenon of HMT across different tissue backgrounds, including the immune, cardiovascular, respiratory, hepatic, renal, musculoskeletal, adipose, and reproductive systems. Moreover, updated applications and functions of mitochondria transfer are discussed. Additionally, we also highlight the therapeutic potential of mitochondria transfer in current preclinical and clinical trials for inherited mitochondrial diseases, cancer, wound healing, and injuries of the respiratory and central nervous systems.
Keywords: vertical mitochondria transfer, horizontal mitochondria transfer, intercellular mitochondria transfer, tunneling nanotubes (TNT), extracellular vesicles (EVs), gap junctions (GJs)
Introduction
Mitochondria are essential double-membrane subcellular organelles that can be found in nearly all eukaryotic cells. Considering their important regulations in continuously supplying eukaryotes with energy in the form of adenosine triphosphate (ATP), mitochondria are often referred to as the “powerhouse of the cell”[1]. In addition to energy supply, these ubiquitously distributed intracellular organelles encompassing wide ranges of essential cellular functions in reactive oxygen species (ROS) control[2, 3], calcium signaling and homeostasis[4], and immune responses[5]. Furthermore, mitochondria can regulate the cellular state from normal to stress[6], thus involve in cell death[7, 8].
Mitochondria are unique organelles in cell. They undergo dynamic cycles of fission and fusion, enabling their functions and adaptations to metabolic changes or cellular stress during physical or pathological conditions[9, 10]. Moreover, they are the only organelles that harbor their own DNA (mtDNA), which consists 37 genes and is uniformly distributed within the mitochondrial matrix[11]. Mitochondrial proteomes encoded by mtDNA consist of more than 1000 proteins[12], which include proteins that involved in a variety of important functions as protein synthesis, amino acid and nucleotide metabolism, fatty-acid catabolism, apoptosis, ion homeostasis and etc. Moreover, mitochondrial biogenesis enables a self-renewal route for mitochondrial repair and regeneration[12]. Importantly, the per-cell numbers, size, structure, distribution of mitochondria in individual cell are under active and dynamic modeling, which may vary depending on the cell type, cell cycle and environmental conditions. Studies show that the respiration rate and mitochondrial protein content are different among different species and tissues[13]. Moreover, the cristae structure also vary across the eukaryotic domain. The typical flat, lamellar cristae structure is commonly observed in most mammalian tissues, whereas tubular cristae have been seed in budding yeast such as Saccharomyces cerevisiae and discoidal cristae have been identified in protists such as Trypanosoma[14].
As mitochondria located in the cytoplasm, when cells divide, parent cells pass mitochondria to daughter cells through a process called vertical inheritance or vertical mitochondria transfer (VMT)[15]. For the past twenty years, emerging evidence started to show that mitochondria can be transfer or delivered from donor cells to recipient cells between developmentally unrelated cell types. This process is called intercelluar or horizontal mitochondria transfer (HMT), which has been reported both in vitro conditions and in vivo systems. HMT can be achieved between the same types of cells or different types of cells, between the same types of tissues/organs or even in different systems, and between health or pathology-related circumstances. In this review, we introduce the origin of mitochondria, describe the mechanisms and functions of mitochondria transfer, and summarize updated understandings of HMT functions in different tissue backgrounds. We also highlight the current experimental and clinical trials of mitochondria transfer and discuss it as a new therapeutic potential in cancer, wound healing and other human diseases.
The discovery, structure and evolution of mitochondria
The discovery of mitochondria was achieved by a German scientist Richard Altmann in around 1890[16, 17], who developed a new approach for tissue fixation and staining. He observed strings of granules within nearly all cells he examined. He called this new structures “bioblasts” and proposed that these granules could be responsible for metabolic processes. Later on, another German scientist, Carl Benda published results with a different crystal violet staining and named the Richard Altmann's bioblasts as "mitochondrion" in 1898[16]. Mitochondrion is from the Greek words mitos (meaning thread) and chondros (meaning granule) with the plural being mitochondria. In 1900, Leonor Michaelis published his findings with dye Janus green stained mitochondria in living cells, proving that they were real and dynamic organelles in cells[16, 18].
Mitochondria are ubiquitous organelles found in almost all eukaryotic cells, ranging from single-celled organisms and plants to vertebrates, including human being[19, 20]. Eukaryotic cell commonly has multiple mitochondria per cell, from several in single somatic cell to hundreds or thousands in multi-nuclear cells or mature oocytes[21]. Among all the tissues, skeletal muscle cells contain the greatest number of mitochondria due to the fused mucltinucleated myofibers and their high energy demands for contraction (Figure 1. a). The typical structure of a mitochondrion is a round to oval-shaped organelle with an average size of 0.75-3 μm[2, 22]. Single mitochondrion includes four essential parts (Figure 1. b, c): outer membrane, inner membrane, the intermembrane space between outer and inner membranes, and the matrix inside a mitochondrion.
Typical mitochondrial morphology in mouse skeletal muscles. The typical mitochondrial morphology is illustrated with transmission electron microscopy (TEM) images detected from the cross-section of sole muscles from 4-month-old adult male mouse (a, b) and cartoon image for the inner structures of single mitochondrion (c).
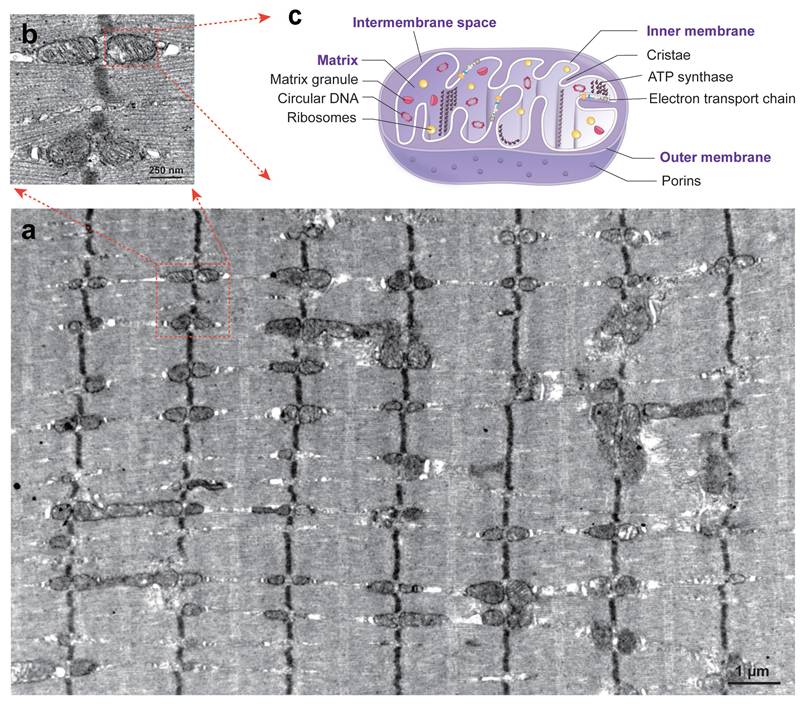
The two membranes of mitochondria are largely different in nature: i.e., the outer membrane is lipid-rich and contains a rather low number of proteins, whereas the inner membrane contains hundreds of different integral membrane proteins[23]. The outer membrane contains multiple voltage-dependent anion channels (VDAC) called porins to control membrane permeability in mediating ion exchanges and metabolites between mitochondria and the cytosol. There are three isoforms of VDAC have been identified in mammals with different functions: VDAC1 is an important mediator for mitochondria-driven apoptosis; while VDAC2, conversely, serves an anti-apoptotic function; VDAC3 participates in the regulation of ROS production and mitochondrial quality control[24]. Another mitochondrial translocase of the outer membrane (TOM) complex mediates retrotranslocation of damaged mitochondrial proteins across the outer membrane in the mitochondria-associated degradation pathway (MAD) pathway for mitochondrial protein quality control[25]. Compared to the outer membrane, the inner membrane of mitochondria has been reported with a protein-to-lipid ratio of about 4 : 1, accounting for one of the most protein-rich membranes of the eukaryotic cell[26]. The inner membrane forms folds known as cristae. This peculiar membrane architecture is essential for diverse mitochondrial functions, including oxidative phosphorylation and the biosynthesis of cellular building blocks[27]. During the cristae formation, the mitochondrial contact site and cristae-organizing system (MICOS) synthesize key mitochondrial phospholipids which cooperate with cristae-shaping proteins and contribute to establish the inner membrane architecture[14]. The cristae increases the surface area of inner membrane and make it available for ATP productions through the electron transport chain and other chemical reactions[28]. The intermembrane space is the most constricted sub-mitochondrial compartment, which hosts only around 5% of the mitochondrial proteome and is involved in the regulation of ion concentrations[29]. Matrix is the innermost compartment of a mitochondrion, which contains mtDNA, ribosomes, various metabolic enzymes for oxidative phosphorylation. Two major proteases of the mitochondrial matrix, LON and ClpXP, have been recognized with essential functions in mitochondrial protein quality control, mitochondrial gene expression and respiratory chain function maintenance[30].
The double membranes and unique genome of mitochondria distinct them from other intracellular organelles, which indicates the special origin of mitochondria during evolution. Several features of mitochondria are similar to bacteria. For instance, their circular genomic DNA are characterized by hypomethylated CpG residues and formylated N-terminus methionine peptides (fMPs)[31]. Moreover, their binary fission and antibiotic sensitivity supporting the endosymbiotic theory of the shared evolutionary past between mitochondria and bacteria[32]. The idea that mitochondria may originate from bacteria has been proposed in the early 20th century[33, 34]. In 1967, American scientist Lynn Margulis (then Lynn Sagan) published the famous article “On the Origin of Mitosing Cells”, in which mitochondria and chloroplasts are proposed to be evolved from endosymbiotic bacteria[35]. Since them, growing evidence from high-throughput gene sequencing and proteomic data support that mitochondria originated from an endosymbiotic event involving the uptake of a Gram-negative bacterium capable of respiring aerobically by a fermentative Asgard rchaeal host cell[36-38]. This endosymbiosis theory of mitochondria origin define the important evolutionary event of eukaryotic cells, as this organelle originated at the same time as the nuclear component of eukaryotic cell[39]. According to the fossil records, this endosymbiotic event likely took place prior to the divergence of all known eukaryotes between 1.5 and 2 billion years ago[40]. The co-evolutionary changes between mitochondria and eukaryotic cells result in the transition of the endosymbiotic bacterium into a permanent organelle.
As a result of endosymbiosis, the evolution of this cooperative relationship between mitochondria and eukaryotic cells made fundamental changes for both two. From one side, mitochondria retained a part of the genome originated from their bacterial ancestors. The propel mitochondrial functions require more than 1000 proteins encoded by nuclear genome[41], and only 14-16% of the modern mitochondrial proteome can be traced back to the ancient bacteria[42]. In mammalian cells, mitochondria generally have a single copy of mtDNA and mitochondrial nucleoids are compacted by mitochondrial transcription factor A (TFAM) into an irregular ellipsoidal shape[43]. In general, mtDNA (16,569-base pair) codes for 13 mRNAs, 22 tRNAs and 2 rRNAs[44-46], encoding 13 proteins that are components of the respiratory chain located in the inner mitochondrial membrane. From the other side, most eukaryotic cells possess a population of mitochondria, therefore multiple mtDNA copies. This intracellular mitochondrial heterogeneity lead to the intercellular mitochondrial heterogeneity, therefore cellular heterogeneity[47]. The mitochondrial heterogeneity contributes to the tissue specific energy demands, cell fate determination and tissue remodeling[48, 49].
Mechanisms of mitochondria transfer
As co-opted for energy production and cellular metabolism, mitochondria have been long considered as autonomous organelles with cytoplasmic inheritance[50]. It has been widely accepted that mitochondria and mitochondrial DNA genome can be transfer or segregated from parent to daughter cells during cell division. In 2004, a novel model of in vitro cell-to-cell communication by tunneling nanotubes (TNT) which facilitate organelle transport was reported by Gerdes and colleagues[51]. After this report, growing evidence continuously show that this TNT-like bridges can transfer mitochondria and other organelles between living cells[52, 53]. In 2015, Brridge and his colleagues utilized mouse tumor models and reported the primary evidence of mitochondria transfer in vivo, in which mtDNA from healthy host cells were transferred into tumor cells and partially restored the mitochondrial functions in tumor cells[54].
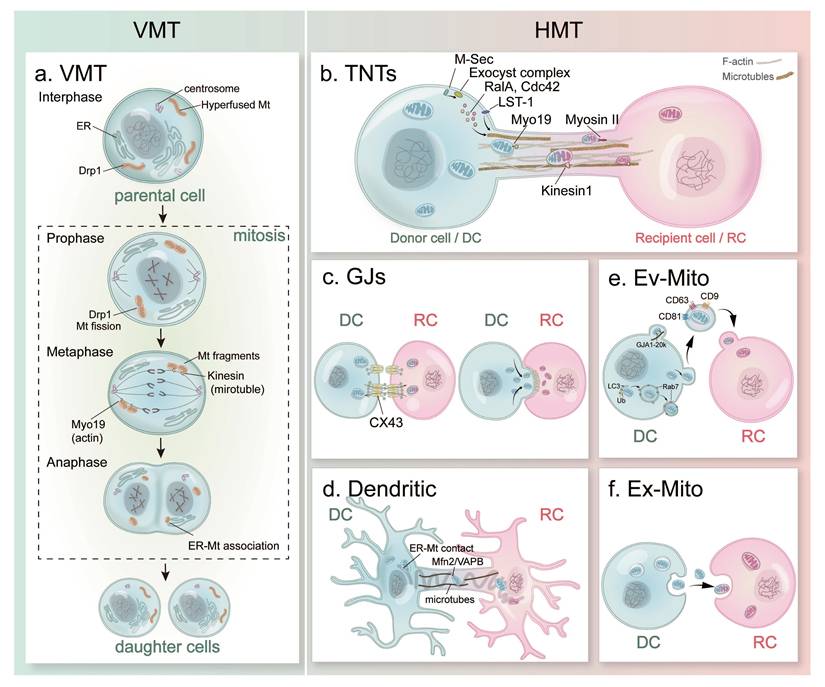
To identify these two modes of mitochondria transfer, vertical mitochondria transfer (VMT) is referring to the parent-daughter mitochondrial inheritance, and horizontal mitochondria transfer (HMT) represents the mitochondrial trafficking between living cells. Since these pioneering discoveries, increasing evidence showed that mitochondrial communications between cells occur more frequently than previously understood. Detailed mechanisms behind VMT were reported (Figure 2. a), and the diverse modes of HMT were discovered (Figure 2. b-f).
Models of mitochondria transfer. (a) Simplified schematic representation of the VMT mode between parental cell and daughter cells; (b) TNT-mediated HMT mode between donor cell and recipient cell; (c) GJ-mediated HMT mode; (d) Dendrite-mediated HMT mode; (e) Ev-Mito transfer; (f) Ex-Mito transfer. Abbreviations: VMT, vertical mitochondria transfer; HMT, horizontal mitochondria transfer; TNTs, tunneling nanotubes; GJs, gap junctions; Ev-Mito, extracellular vesicle-associated mitochondria transfer; Ex-Mito, extracellular mitochondria transfer; DC, donor cell; RC, recipient cell.
Vertical mitochondria transfer (VMT)
During cell mitosis, VMT is critical to properly partition the functional organelles as well as nuclear acid information to daughter cell. In general, mitochondria are typically dispersed throughout the cell cytoplasm with special accumulation only during distinct cellular events, such as synaptic transmission[55] or germ cell formation[56]. Moreover, mitochondria also undergo active intracellular movement on the actin or microtubule-based cytoskeletal network by dynein, kinesin and myosin-based motor proteins[57-59]. Considering the special feature of mitochondria, passing mtDNA and the functional sets of mitochondria is important to achieve successful division[15].
Mitochondrial dynamics are achieved by a balance between mitochondrial fusion and fission: Mitochondrial fusion is the physical merging of the outer and then the inner mitochondrial membranes of two originally distinct mitochondria; whereas mitochondrial fission is the process of division of a single mitochondria into two or more independent structures. The continual fusion and fission of mitochondria is extremely critical to maintain mitochondrial integrity and homeostasis, which were systematically summarized and reviewed in published paper[60-64]. Here, we focused on the mitochondrial segregation during VMT process in mitosis.
Mitosis ensures genome integrity by mediating precise segregation of the duplicated genetic material, including mtDNA (Figure 2. a). During the interphase (G1, S, G2 phases), mitochondria form a extensively fused and interconnected tubule structure[65]. Mitochondrial fission from mitochondrial fragment in early mitotic phase by the phosphorylation of Drp1 through both Cdk1-cyclin B[66] and Auora A mediation[67]. When cells enter the prophase, mitochondria start to prepare for their division by recruiting the pro-fission dynamin-related GTPase Drp1 and its receptors (including Mff, MiD49 and MiD51) localized on the outer membrane of mitochondria[68]. Mitochondrial highly fragmented morphology which is even more pronounced when cells reach the metaphase and anaphase stages. During the whole cell division processes, mitochondria interact with microtubules and actin cytoskeleton, as well as endoplasmic reticulum (ER) to achieve propel partitioning. On one hand, mitochondria associate with the growing microtubule tips through interactions between mitochondrial protein Miro and the cytoskeletal-associated protein Cenp-F[69]. On the other hand, Myo19 actin-based motor helps mitochondria movement and contribute to faithful segregation during division[70]. In addition, mitochondria form a membrane contact sites with ER in cells, which expand and display a tighter physical association between these two organelles during division[71]. This ER-MT association could support mitochondrial functions during mitosis and co-segregation of the two organelles to the daughter cells. At the end of cytokinesis during the telophase, mitochondria begin to recruit the pro-fusion proteins MNF1/2 at the outer membrane and OPA1 at the inner membrane and achieve elongated structures[15, 65]. By the time of cell division completion, the fragmented mitochondria re-form the filamentous network structure in the daughter cells after division[72]. In short summary, mitochondria remodel through complex structural changes and dynamic associations with the cytoskeleton during VMT process.
Horizontal mitochondria transfer (HMT)
Compared to VMT, an emerging body of literature indicates that a wide range of cell types export their mitochondria for delivery to developmentally unrelated cell types through HMT approach. Even though the detailed molecular and signaling mechanisms underlying HMT are not fully understood, it has been reported to occur between adjacent cells of the same type, between distant cells of different types, and even across remote, distinct tissues. In general, HMT can be classified into two main categories: contact-dependent and contact-independent transfer.
Contact-dependent mitochondria transfer
The contact-dependent HMT mode refers to the formation of transient cellular connections between donor and receptor cells, through which mitochondria can move from one cell to another. Currently reported contact-dependent mitochondria transfer mechanisms include tunneling nanotubes (TNTs), gap junction (GJ)-medicated transfer and dendritic structure-mediated transfer (Figure 2. b-d).
Tunneling nanotubes (TNTs)-mediated transfer
TNTs were first reported and referred as “highways” for intercellular organelle transport in 2004 between the same type of cells, including rat pheochromocytoma PC12 cells[51] and immune cells[73]. Later on, TNT-mediated mitochondria transfer was reported between two cells from different types and species, transferring from human mesenchymal stem cells (hMSCs) to co-cultured rat cardiac myocytes[74]. From in vitro to in vivo systems, TNTs are now extensive studied with more advanced and diverse detecting approaches, such as tissue-specific labeling method, scanning electron microscopy (SEM), atomic force microscopy (AFM), cryo-electron microscopy, in vivo live imaging, and others.
Mitochondria have been shown as the most frequently reported organelle that are unidirectionally or bidirectionally transferred by TNTs[75]. The components of cytoskeleton and the size (including length and diameter) of TNTs vary depending on the cell type, micro-environment and cellular status[76]. The diameter of TNTs can vary between 50 to 1500 nm, and their length can range from several tens to hundreds of micro[76, 77]. Reported data suggest that various stress factors, for example p53 activation[78], that induce mitochondrial damage could facilitate TNT formation and associated mitochondria transfer.
The formation of TNTs involves a variety of molecules (Figure 2. b)[79], including M-Sec, small GTPases, exocyst complex, and leukocyte specific transcript 1 (LST1). M-Sec, a mammalian protein as known as TNF alpha induced protein 2, was reported to induce de novo formation of plasma membrane protrusions through Ral-exocyst pathway during TNT formation[80]. The exocyst complex is an octameric protein complex, which has been reported with functions during the fusion of secretory vesicles derived from Golgi body with plasma membrane in exocytosis[81]. During TNT formation, M-Sec interacts with exocyst complex and promotes its assembly, during which small GTPase RalA and Cdc42 are recruited and lead to the remodeling of actin cytoskeleton[80]. LST1, a transmembrane protein, recruits RalA and the actin cross-linked protein filamin to the submembrane region, which accelerates the interaction between RalA and the exocyst complex, ultimately leading to TNT formation[82]. Cdc42 is reported with functions during the prolongation stage of TNTs[80]. Compared to the mechanism behind TNT formation, the molecular regulation of the membrane fusion between the donor and the target cells remains unclear.
Structurally, TNTs are thin membrane tubes that are filled with cytoskelston filaments, mostly F-actin and microtubles[76]. F-actin is the most common cytoskeleton found in TNTs, whose labeling is widely used to mark TNTs in cells. Besides F-actin, microtubules also participate in TNT functions. For example, cultured hippocampal neurons generated protrusions towards astrocytes and formed microtubule-containing TNTs, in which 35% of TNTs were F-actin negative under fluorescence microscopy[83]. Moreover, cells may from different types of TNTs with diverse functions. For example, human monocyte-derived macrophages can form two types of TNTs, one is thin nanotubes containing only F-actin, the other is thick nanotubes (diameter larger than 0.7 μm) containing both F-actin and microtubules[84]. Interestingly, in these cells, bacteria could be trapped and only surf along thin nanotubes after be phagocytosed; mitochondria and intracellular vesicles were detected and transferred only in thick nanotubes[84].
Since the main cytoskeletons in TNTs include F-actin and microtubes, the shuttling of mitochondria requires these cytoskeletons associated motors for intercellular transportation. For example, F-actin polymerization and F-actin-associated myosin II motors are essential for mitochondria transfer in TNTs[85]. Selectively blocking F-actin polymerization by cytochalasin B, or treatment of myosin II specific inhibitor S-(-)-Blebbistatin inhibit TNT formation and related intercellular organelle transfer[85, 86]. Moreover, Miro1, a calcium-sensitive Rho-GTPase located in the outer mitochondrial membrane, is essential for F-actin and microtubule-mediated mitochondria transfer. Miro1 has been reported to interact with Myosin XIX (Myo19), an actin-based motor, which contributes to mitochondria cristae structure and potentially mitochondria transfer[58, 87]. Miro1 also can interact with mitofusion1/2 and combines with the kinesin-1 molecular motor through TRAK1/2 and OIP106/98, forming a mitochondria-loaded vesicle and enabling the mitochondrial transfer on microtubules in neurons[77, 88, 89]. Miro1 has been reported with essential functions in mitochondria transfer from mesenchymal stem cells (MSCs) to epithelial cells during epithelial injury and recovery[90]. In vivo experimental mouse study further suggests that both Miro1 and Miro2 are required for TRAK2-mediated mitochondrial motility and positioning on actin and microtubule cytoskeleton[91].
The transport of mitochodnria transfer via TNTs is bi-directional. Interestingly, functional mitochondria in healthy cells are preferably released and transferred to damaged or injured cells. For example in brain environment, TNTs were observed between neurons, between glia cells, and between neurons and glia cells. In TNTs formed between astrocytes, an extracellular molecule S100A4 seems control TNT direction. In astrocytes, p53 activates caspase-3, which leads to the cleavage and decreased concentration of S100A4 in cells[92]. As a result, a gradient of S100A4 was formed in astrocyte population, and TNTs are initiated in low concentration cells towards high concentration cells[92]. Moreover, another team reported that when neurons were subjected to oxygen-glucose deprivation/reoxygenation exposure damage, TNT formation and mitochondrial transportation from astrocytes to injured neurons were facilitated[93]. In addition, TNTs between neurons and microglia are bi-directional, and microglia preferred transfer mitochondria to α-Synuclein burdened neurons over the healthy ones[94].
Till now, TNT-mediated mitochondria transfer is the most common transfer route of HMT, which has been reported during physiology and pathological conditions in the immune system, cardiovascular system, respiratory system, epithelial tissues, central nervous system (CNS), and cancers[95]. Detailed mechanisms behind TNT-mediated HMTs may vary with reflection of their distinct tissue backgrounds.
Gap junction-mediated transfer
Gap junctions (GJs) are specialized intercellular channels between adjacent cells, allowing direct cell-cell transfer of cytoplasmic contents[96]. GJs are encoded by connexin gene family in mammalian, which has been extensively studied in the past century. Among all the connexin members, connexin 43 (Cx43) is widely expressed in mammalian tissues. Like most trans-membrane proteins, Cx43 protein is synthesized by ribosomes, bounded to the ER membrane, trafficked via the ER-Golgi mechanism, and finally inserted into the plasma membrane (PM)[97]. Once delivered to the PM, Cx43 proteins are assembled into hexamers and function as hemichannels in one cell[97]. When two cells connect, opposed Cx43 connnexins from two cells form a complete gap junction facilitating direct intercellular communication. Reported studies showed that Cx43-mediated GJs are gated open more often than closed, which opposites to Cx43-mediated hemichannels[98].
Several reports illustrate the importance of Cx43-mediated GJs in the mitochondria transfer process[99]. In bone marrow environment, Cx43 in hematopoietic stem and progenitor cells (HSPC) mediates the mitochondria transfer from HSPCs to MSCs through cell-contact dependent approach[100]. Moreover, MSCs deliver mitochondria to damaged chondrocytes under chemical, environmental, and mechanical stress through Cx43-associated GJs in OA cartilage[101, 102]. In vitro co-culture and in vivo mitochondria labeling method indicated that bone marrow stromal cells (BMSCs) transfer mitochondria to injured motor neurons, which is blocked by GJ inhibitor 18β glycyrrhetinic acid[103]. In addition, ultra-purified BMSCs transfer a greater number of mitochondria into mitochondria-deficient (ρ0) cells mainly through Cx43-GJs[104].
These Cx43-mediated mitochondria transfer may achieved by different mechanisms (Figure 2. c). First, Cx43 can facilitate mitochondria transfer through direct GJ-mediated cell-contact[95]. The Cx43-mediated GJ channels are non-selective pores between cells, allowing the exchanges of many intracellular molecules (up to ~1kDa), such as inorganic salts, sugars, amino acids, and nucleotides pass through connecting cells[97, 98]. Giving the size of Cx43-mediated GJs within a range of 10-16 Å/mesh[105] (~1190-2000 μm) and typical mitochondria size with 0.5-3 μm[106], it is possible that mitochondria can directly pass through these channels. Second, Cx43 may function as a stabilizer to adhere mitochondria to the docked membrane structure of donor and receptor cells[97, 107], which further facilitate mitochondria transfer between cells. Third, a truncated Cx43 isoforms, GJA1-20k has been implicated in mitochondrial motility by mobilizing mitochondria along microtubules during mitochondria transfer from human mesenchymal stromal cells to stressed chondrocytes[102].
In addition to GJ-mediated mitochondria transfer, Cx43 also contribute to TNTs and extracellular vesicles (EV)-mediated transfer. Studies showed that human induced pluripotent stem cell (iPSC)-derived MSCs form TNTs and transfer mitochondria to epithelial cells, which is dependent on the expression level of Cx43 in iPSC-MSCs and attenuates the mitochondrial dysfunctions of epithelial cells[108]. In lipopolysaccharide (LPS)-induced acute lung injury (ALI) model, BMSCs formed Cx43-containing GJs with the alveolar epithelia and released the mitochondria-containing EVs, which have been engulfed by the epithelia, thus restituting alveolar bioenergetics[107]. Interestingly, Cx43 is found present inside of the mitochondria as mitocondrial connexin 43 (mtCx43) in many pathological conditions, particularly in the heart[109]. mtCx43 is reported at the inner membrane of mitochondria in cardiomyocytes and contributes to mitochondrial K(+) flux, potentially by forming hemichannel-like structures[110]. Furthermore, mtCx43 participates in shRNA-Rictor-induced mitochondrial dysfunctions in the mouse embryonic stem cell-derived cardiomyocytes (ESC-CMs)[111].
In short summary, GJ-mediated mitochondria transfer facilitates a direct and fast intercellular communications between adjacent cells. The mechanism behind Cx43-mediated mitochondrial transfer and mtCx43 functions shall be further studied in various physical and pathological conditions[99].
Dendritic connection-mediated transfer
In addition to TNTs and GJs, dendritic connection has been reported to involved in mitochondria transfer in a special set of cell, osteocytes, in bone (Figure 2. d). Osteocytes are the terminal differentiated bone cells embedded in the mineralized bone matrix, which are inter-connected with each other with dense dendritic networks[112]. The length, density and dynamics of osteocyte dendrites are tightly associated with osteocyte functions during bone remodeling and mechanotransduction[112-115]. These are two main types of cytoskeleton composed in osteocyte dendrites, i.e., F-actin filaments and microtubules. Recently, Gao and his research team observed the distribution of mitochondria along the tubulin track of dendrites in the primary osteocytes, whose number decreased with aging[116]. Under live imaging microscopy, they further reported that the dynamic mitochondria transfer takes place between osteocytes through their dendrite connections, and the transferred mitochondria are able to restore the cellular metabolism in the stressed osteocytes[116]. This process requires tethering proteins, including mitofusion 2 (Mfn2) and vesicle-associated membrane protein B (VAPB), which facilitates ER-mitochondria contact formation and mediate the transfer of mitochondria between osteocytes[116]. Published reported revealed that ER can slide on stable acetylated microtubles, and the ER-mitochodria contact maintained during dynamic morphological processes in cell[117]. These results may suggest that the mitochondria transfer is achieved by the interaction between ER, mitochondria and microtubules through the cytoskeleton extension during osteocyte dendritic formation process[77].
Even though F-actin and microtube are largely involved in HMT transfer, large numbers of HMT reports based on these cytoskeleton structures were through TNT-mediated mode, but mere study reported the cell dendrite-mediated mitochondria transfer. Till now, mitochondria transfer between osteocytes[116], transfer from osteocytes to endothelial cells[118], and from astrocytes to neurons[119], are reported through cell dendrites. Structually, it is difficult to distinguish TNT-mediate and dendrite-mediate HMT mode[120]. More evidence is needed to demonstrate this dendrite-mediated transfer approach may server as a more universal mode for mitochondria communications between other types of cells with dense dendries. In addition, it is of importance for future studies is to distinguish the fundamental regulatory and transnational differences between dendritic connections and other forms of mitochondria transfer.
Contact-independent mitochondria transfer
Besides the cell-contact dependent mechanisms listed above, mitochondria and their DNA components can be released outside the cell with in intact, fragmented, and vesicle encapsulated forms under both physiological and pathological conditions[1, 121-123]. These contact-independent mitochondria transfer can be further categorized as two modes: i.e., extracellular vesicles with mitochondria (Ev-Mito) and extracellular mitochondria (Ex-Mito) (Figure 2. e, f).
EV-associated mitochondria (Ev-Mito) transfer
Intercelluar mitochondria transfer can be achieved by the release and capture of extracellular vesicles (EVs), i.e., Ev-Mito mode, as the second common mechanism reported beside TNTs (Figure 2. e).
Based on their size, EVs can be further divided in to three subtypes: exsosomes (30 - 100 nm), microvesicles (MVs, 100 nm - 1 μm), and apoptotic bodies (˃ 1 μm)[77, 124]. As limited in size, exsosomes derived from endosomal cell membranes only contain mtDNA and other small molecules[125]. Small EVs (100-200 nm), marked with the tetraspanins CD63, CD9, and CD81, also contains damaged mitochondrial components[95, 126, 127]. Large MVs (~ 1 μm size) can load functional mitochondria, which may formed via multivesicular bodies (MVBs) or bud off from the plasma membrane[95]. This Ev-Mito transfer mode has been reported in various types of cells under both physical and pathological condition. Brown adipocytes remove damaged mitochondrial components through Ev-Mito process[127]. In bone, osteoblasts release Ev-Mito to osteoprogenitor cells and promote bone formation[128]. In the brain following ischemic stroke, astrocytes release Ev-Mito to the hypoxic neurons to support neuronal survival[119]. Moreover, cardiomyocytes also release 200-600 nm mitochondria in EVs[129].
Even though EVs from Ev-Mito transfer are all marked with the tetraspanins CD63, CD9, and CD81, but their export from donor cells may through various export mechanisms[95]. Considering the size of extracellular mitochondria, it is originally suggested that Ev-Mito was released as microveriscles directly through plasma membrane blebbing[130, 131]. Moreover, live cell imaging also indicates that Ev-Mito transfer can be achieved through an exosomal/endolysosomal mechanism, in which Ev-Mito released from MSCs is incorportated into LC3-positive autophagosomes before being released[132]. Recent studies also suggest that the formation and transportation of Ev-Mito is associated with mitophagy process, the key regulation for mitochondrial quality control by promoting lysosomal degradation of damaged mitochondria, through both PINK1/Parkin-dependent and -independent pathway[133]. Data showed that the activation of Parkin targets damaged mitochondrial content to lysosomes for degradation thus preventing their inclusion into EVs[134]. Moreover, thermogenically stressed brown adipocytes release Ev-Mito that contain oxidatively damaged mitochondrial proteins through PINK1-dependent manner and exportation requires PARKIN protein[127, 135]. Moreover, overexpression of Parkin-independent mitophagy regulator, BNIP3 (BCL2 interacting protein 3) and BNIP3L/NIX (BCL2 interacting protein 3 like) prevents the release of Ev-Mito[136]. In addition, another study reported that small GTPase Rab7 is essential for the secretion of mitochondria-contained multivesicular bodies (MVB) in the heart of aged mouse during cardiac pathophysiology[129].
Ev-Mito transfer has emerging as the second common HMT mode reported in various tissues and organs. Since the EV-mediated communications are not limited to two adjacent cells, their influence can be reach out to broad internal environment and achieve fundamental homeostasis.
Extracellular mitochondria (Ex-Mito) transfer
Free or naked mitochondria can be transferred between donor and recipient cells, which is called extracellular mitochondria (Ex-Mito) transfer (Figure 2. f). Since mitochondria, as independent organelle, have its own double membranes, cell-free mitochondria can be released or extruded into the circulating system as Ex-Mito[95]. Reported data showed that free Ex-Mito have a size approximately 0.5-1 μm in diameter, which lack EVs and contain a full-length of mtDNA genome[121, 137]. The free mitochondria were first identified in the blood from both mice and humans, in which the activated platelets contain both free Ex-Mito and Ev-Mito in a ~2:1 ratio[138].
Mechanistically, the extrusion and internalization of free mitochondria mostly occur during the process for mitochondrial quality control or when cells are under stress[77]. For example, cytoplasmic vacuoles engulf mitochondria and subsequently fuse with the plasma membrane, leading to Ex-Mito release to the extracellular medium during tumor necrosis factor α (TNFα)-induced cell death in a caspase-dependent fashion[139]. Moreover, Hela cells extrude fragments of mitochondria under high rates of ROS conditions[140]. In vivo, plates extrude Ex-Mito to enhance inflammatory responses[138]. Moreover, adipocytes also generate Ex-Mito locally and transferred to macrophages or into the circulation for distant organs[137]. With growing evidence to link free mitochondria or mitochondrial contents with pathological conditions, the study of Ex-Mito could provide a new way to find mitochondrial associated biomarkers for diseases.
Mitochondria transfer in tissue homeostasis
Communication between cells is crucial for maintaining life activities and coordinating the normal operation of various organs and systems. Mitochondria transfer is an important form of intercellular communication, which supports the metabolism of recipient cells, controls the mitochondrial mass of donor cells, regulates the immune system, and maintains metabolic homeostasis in various tissue/organ backgrounds[123, 141]. Here, we summarize current progresses of mitochondria transfer in different systems (Table 1) and highlight their essential regulatory mechanisms during physiological conditions and pathogenesis.
Mitochondria transfer reported in different tissue backgrounds.
| Tissues/Organs | Donor cells (Cell type/species) | Recipient cells (Cell type/species) | Mode of MT transfer | Functions | Ref. |
|---|---|---|---|---|---|
| Immune systems | MSC/Human | Alveolar macrophages/Murine | EVs | Ameliorate lung injury in vivo | [148] |
| M1-like macrophage/Mouse | MSCs/Mouse | TNTs and EVs | Induce ROS bursts, impair the osteogenic differentiation of MSCs | [253] | |
| Breast cancer cells/Human | T-cells/Human | TNTs | Lead to T cell exhaustion | [246] | |
| T cells/Mouse | Breast cancer cells/Human, Breast cancer cells/Mouse | TNTs | Facilitate immune evasion | [153] | |
| Nervous system | M2-like macrophage/Mouse | Sensory neurons/Mouse | EVs | Reduce inflammatory pain | [152] |
| BMSCs/Rat | Cortical neurons/Rat | GJs | Protect neural cells from apoptosis | [103] | |
| Microglia/Mouse | Neurons/Mouse | TNTs | Restore neuronal health | [308] | |
| Astrocytes/Human | Glioblastoma/Human | Intercellular transfer through network-forming | Upregulate metabolic pathways related to proliferation and tumorigenicity | [309] | |
| Astrocytes/Mouse | Neurons/Rat | TNTs | Support neuronal mitochondrial metabolism and survival | [119] | |
| Cardiovascular system | Cardiomyoblast cells/Rat | H9c2 cardiomyocytes/Mouse | Ex-Mito | Alleviates myocardial ischemia-reperfusion injury | [183] |
| M2-like macrophage/Mouse | H9c2 cardiomyocytes/Rat | Cell fusion | Prevent doxorubicin-induced cardiotoxicity | [310] | |
| Colon carcinoma cell line/Human | Neonatal cardiomyocytes/Mouse | Ex-Mito | Protect cardiomyocytes from doxorubicin-induced mitochondrial dysfunction | [181] | |
| hUC-MSC/Human | Cardiomyocytes/Mouse | Ex-Mito | Repair myocardial injury | [182] | |
| Osteocytes/Mouse | Endothelial cells/Mouse | Ex-Mito | Regulate the vascularization of transcortical vessels | [118] | |
| MSCs/Human | Endothelial cells/Human | TNTs | Promote EC transplantation and stimulating angiogenesis | [184] | |
| Respiratory system | MSCs/Human | Bronchial epithelial cells/Human | TNTs | Mediate the repair of damaged bronchial epithelial cells | [186] |
| Asthmatics' airway myeloid-derived regulatory cell/Human | CD4+ T/Human | EVs | Reprogramme the function of T cells and inhibite their inflammatory responses | [187] | |
| Airway smooth muscle cells (ASMCs)/Human | Airway smooth muscle cells (ASMCs)/Human | EVs | Regulate bioenergetics and cellular functions within the airway | [189] | |
| hP-MSC/Human | TC-1 epithelial/Mouse, HUVEC endothelial/Human, Hs888Lu fibroblast/Human | Ex-Mito | Repair the damaged mitochondria, restore the inhibited mitochondrial autophagy process | [191] | |
| BMSCs/Mouse | Pulmonary microvascular endothelial cells/Mouse | TNTs | Restore the function of mitochondria, reduce the levels of ROS and cell apoptosis, recovery the barrier function of ECs | [192] | |
| Liver | hUC- MSCs/Human | AML12 liver/Mouse | TNTs | Restored mitochondrial function, activate the Nrf2/HO-1 signaling pathway in hepatocytes, suppress ferroptosis and fibrosis | [199] |
| hUC-MSCs/Human | Neutrophils/Murine | EVs | regulate neutrophil NETs formation | [200] | |
| BMMSCs/Human | Primary hepatocytes/Mouse | TNTs | Enhance lipid metabolism and facilitate the restoration of tissue homeostasis | [197] | |
| MIHA cells (Immortalized hepatocytes)/Human | NaAsO2-treated MIHA cells | TNTs | Enable the mutual supplementation of mtDNA between cells, compensating for the mitochondrial dysfunction caused by arsenic and alleviates cellular senescence | [198] | |
| PLC/PRF/5 cells/Human, MHCC-97H cells/Human | Hep3B cells/Human | TNTs | Enhance the migration and invasion abilities of the less invasive cells | [201] | |
| Kidney | BMSCs/Rat | Glomerular endothelial cells/Rat | Contact-independent Mt transfer( co-culture, the specific mode was not explicitly stated. ) | Anti-apoptotic | [210] |
| BMSCs/Rat | Proximal tubular epithelial cells/Rats | Contact-dependent Mt transfer (co-culture, the specific mode was not explicitly stated. ) | Inhibit cell apoptosis, inhibit ROS production, restore transporter expression, and repair renal tubular structure | [311] | |
| Musculoskeletal system | MLO-Y4 osteocyte/Mouse | MLO-Y4 osteocyte/Mouse | Dendiritic network | Restore the metabolic function of injured mitochondria and maintain the homeostasis of bone cells | [116] |
| RAW 264.7 macrophage/Mouse | BMSCs/Mouse | TNTs, EVs | Promote the osteogenic differentiation of MSCs and regulate the homeostasis of bone. | [253] | |
| BMSCs/Mouse | Chondrocytes/Mouse | TNTs, GJs | Improve the mitochondrial function of chondrocytes | [312] | |
| BMSCs/Rat | Chondrocytes/Rat | Contact-dependent Mt transfer (co-culture, the specific mode was not explicitly stated. ) | Improve mitochondrial function, enhance cell proliferation, and inhibit apoptosis | [217] | |
| Adipose tissues | Adipocytes/Mouse | Macrophages/Mouse | Not well understood | Regulate metabolic homeostasis | [221] |
| Adipocytes/Mouse | Cardiomyocytes/Mouse | EVs | Protect cardiomyocytes from acute oxidative stress | [127] | |
| ADSCs/Rat | Dendritic cells/Rat | EVs | Reduce inflammation | [224] | |
| ADSCs/Mouse | Macrophages/Mouse | TNTs | Promote barrier restoration | [223] | |
| Macrophages/Mouse | Adipocytes/mouse | EVs | Promote Adipocyte-myofibroblast transition | [222] | |
| ADSCs/Human | Cardiomyocytes/Rat | TNTs | Improve the cardiac function | [226] | |
| ADSCs/Human | islet β-cells human | TNTs and EVs | Improve insulin secretory function | [225] | |
| ADSCs/Human | Breast cancer cells/Human | TNTs | multi-drug resistance (MDR) | [227] | |
| Reproductive system | ADSCs/Mouse | Oocytes/Mouse | microinjection | Enhance oocyte quality, promote embryonic development | [228] |
| EnMSCs/Mouse | Oocytes/Mouse | microinjection | Enhance oocyte quality, promote embryonic development | [229] | |
| iPSCs/Mouse | Oocytes/Mouse | microinjection | Enhance oocyte quality, promote embryonic development | [230] | |
| USCs/Human | Oocytes/Mouse | microinjection | Improve embryonic development and metabolism | [231] | |
| ASCs/Mouse | Oocytes/Mouse | microinjection | Improve the developmental potential of cryopreserved oocytes | [232] |
Immune systems
Inflammation has been associated with various degenerative and acute diseases, as well as the aging process, with mitochondrial alterations playing a central role in these phenomena[142]. Beyond their bioenergetic and biosynthetic functions, mitochondria are increasingly acknowledged as a key trigger of innate immunity that influence antimicrobial responses, autoimmune diseases, metabolic disorders, and cancers[143]. When mitochondria are compromised due to damage or mutations, excess ROS are produced, and cellular stress cannot be effectively resolved. Consequently, mitochondria serve as principal regulators of the pro-inflammatory states, modulating the innate immunity through redox-sensitive inflammatory pathways or by directly activating the inflammasome[144].
Reported studies showed that mitochondria transfer is involved in immune responses. For example, platelet-derived mitochondria that transfer to human dermal fibroblasts elicit an anti-inflammatory process, potentially by suppressing both intracellular and mitochondrial ROS production and facilitating the wound repair[145]. In bone marrow, stromal cell-derived mitochondria are delivered to progenitor cells, inducing a leukocyte proliferative response that defends against acute bacterial infections at distant sites[146]. MSC-mediated mitochondrial transfer through EVs, which can alter alveolar macrophage phenotype, enhancing their anti-inflammatory and phagocytic functions, while promoting tissue regeneration[147, 148].
Moreover, mitochondria transfer also participates in pathological immune-responses. Data showed that extracellular mtDNA can promote NLRP3 inflammasome activation and induce acute lung injury through TLR9 and NF-κB[149]. In autoimmune diseases, mitochondria transfer from MSCs is associated with enhanced regulatory T cell activation, leading to increased expression of FOXP3, IL2RA, CTLA4, and TGF-β1, thus contributing to the suppression of immune responses and the enhance of immune tolerance[150]. Macrophages, typically responsible for digesting dysfunctional mitochondria to maintain homeostasis, can also donate mitochondria in pathological contexts. In osteoporosis, macrophages with an M1-like phenotype transfer mitochondria to MSCs, inducing ROS bursts that impair the osteogenic differentiation of MSCs[151]. Conversely, M2-like macrophages facilitate the resolution of inflammatory pain via transferring mitochondria to sensory neurons, mediated by the CD200 receptor (CD200R) on macrophages and its noncanonical ligand iSEC on sensory neurons[152]. Natural killer T cells transfer mitochondria via TNTs, enhancing cancer cell basal respiration, spare respiratory capacity, and growth[153].
Nervous system
The central nervous system (CNS) comprises the brain and spinal cord. At the cellular level, CNS contains neurons, glial cells, and other cell types[154]. Mitochondria are integral to the function of the CNS as they produce the majority of the energy required for membrane ATPases, the influx and efflux of neurotransmitters, and the formation of new neural circuits[155]. Mitochondria exhibit an adaptive response to the fluctuating demands of their host cells to maintain bioenergetic and oxidative homeostasis[156]. In the context of a damaged or diseased brain, mitochondrial dysfunction results in decreased ATP levels[157], which further triggers an increase in ROS production and the activation of related apoptosis pathways[158]. Therefore, various forms of mitochondrial dysfunction have been identified in conditions such as schizophrenia (SZ)[159], spinal cord injury[160], Parkinson's disease[161], and Alzheimer's disease[162].
To achieve the homeostasis in nervous systems, mitochondria transfer has been reported between different types of cells. Data showed that multipotential mesenchymal stem cells (MMSCs) can transfer mitochondria to both neuron and glial cells[163]. Moreover, astrocytes can transfer mitochondria to neurons, which further modify the calcium concentration in damaged neurons[164]. In response to ischemic injury, glial cells can transfer healthy mitochondria into injured neurons, thereby enhancing the survival rate of the damaged cells[165]. Furthermore, mitochondria can be transferred from astrocytes to microglia, influencing their phagocytic functions; conversely, microglia can also transfer mitochondria to astrocytes, which may either promote or inhibit neuronal inflammation[94].
Currently, the restoration of normal biological function in damaged mitochondria is one of the research hotspots for treating neurological diseases[166]. Numerous studies have confirmed that mitochondria transfer can alter the biological activity and physiological state of recipient cells in spinal cord injury (SCI) models[167]. The transfer of normal mitochondria from donor cells to recipient cells with abnormal mitochondrial functions can enhance mitochondria-related biosynthesis in recipient cells, and affect their biological functions[168]. Studies have demonstrated that transplanted BMSCs can prevent neuronal apoptosis and facilitate locomotor functional recovery by transferring mitochondria to injured neurons following SCI[103]. Furthermore, result indicate that photobiomodulation (PBM) intervention facilitate the transfer of platelet-derived mitochondria to neurons in the treatment of SCI, which is more effective than single treatment in terms of motor function recovery, tissue repair, and inhibition of neuronal apoptosis[169]. Therefore, mitochondrial transplantation represents a promising therapeutic approach for spinal cord injury. In context of neurological ischemic diseases or neurodegenerative diseases caused by ischemia and hypoxia, the transfer of mitochondria from healthy cells to damaged cells can often restore cellular activity, rescue mitochondrial respiratory function, promote oxidative phosphorylation, reduce lactic acid production, and ultimately prevent apoptosis[170]. Regarding SZ, isolated active normal mitochondria can enter various cell types without any manipulations, thereby improving impaired mitochondrial functions in SZ-derived cells[171]. In neurological disease, cerebrospinal fluid (CSF) lactate levels are elevated in stroke patients and show a negative correlation with astrocytic mitochondria. Inhibition of mitochondria transfer from astrocytes to injured neurons exacerbates ischemia-reperfusion injury in mouse models of ischemic stroke, whereas promoting the transfer of astrocytic mitochondria to neurons can counteract ischemic stroke[119, 172, 173].
Cardiovascular system
Mitochondria make up to approximately 29% to 36% of cardiac myocyte volume, thus providing energy for the contraction of myocardial cells and critical for cardiac functions[174]. Therefore, maintaining the structural integrity and functional stability of mitochondria is of crucial significance for the overall health of the cardiovascular system[175]. The balance of mitochondrial homeostasis is regulated by dynamic processes, such as mitochondrial dynamics, crest remodeling, biosynthesis, autophagy, and oxidative stress. When mitochondrial homeostasis is disrupted, the contractile function of myocardial cells is affected and their activities are hindered, which can lead to diseases, such as cardiomyopathy, atherosclerosis, hypertension and heart failure[176].
Mitochondria transfer is a key example of intercellular communication in cardiomyocytes[177], which supports the metabolism of recipient cells and regulate tissue homeostasis[120]. The methods for treating cardiovascular diseases through mitochondrial transplantation include naked mitochondria transfer and cell-mediated mitochondrial transplantation, the latter of which is mainly mediated by TNTs, EVs and cell fusion[178]. In the heart, following myocardial ischemia or reperfusion injury, cardiomyocyte-derived small EVs deliver mitochondrial components to cardiac fibroblasts, activating the cGAS-STING pathway and promoting ischemic cardiac fibrosis[179]. Additionally, after myocardial infarction, cardiac fibroblasts transfer damaged mitochondria via small EVs to macrophages, activating the NLRP3 pathway and exacerbating tissue inflammation and adverse ventricular remodeling[180].
Moreover, the potential of mitochondria transfer therapy in restoring the cellular function of myocardial cells has been tested as an important approach in the treatment of heart failure. For example, Zhang A injected human colon carcinoma cell lines into the hearts of wild-type mice to ensure that human mitochondria were absorbed by mouse cardiomyocytes. These results indicated that mitochondria transfer had a protective effect on doxorubicin-induced heart failure and could protect cardiomyocytes from doxorubicin-induced mitochondrial dysfunction[181]. Jin N transferred mitochondria from human umbilical cord mesenchymal stem cells (ucMSCs) into cardiomyocytes, which protects the cardiac function and prevents cardiomyocyte apoptosis, providing a new strategy for the treatment of heart failure[182]. Moreover, Sun X constructed myocardial cell injury models in vitro and implemented different mitochondrial transplants to explore new therapeutic measures for improving cardiovascular diseases. Studies have found that Alda-1 stimulation significantly enhances the respiratory and mechanical functions of myocardial cells caused by mitochondria transfer, showing great potential in the treatment of myocardial ischemia-reperfusion injury[183]. In addition, Lin RZ reported that mitochondria transfer between mesenchymal and endothelial cells through TNTs had a protective effect on the function of endothelial cells by triggering mitochondrial autophagy[184].
Respiratory system
Mitochondria transfer provides a new therapy for treating respiratory diseases caused by mitochondrial dysfunctions, such as chronic obstructive pulmonary disease and pulmonary fibrosis[185]. In vitro, Sinclair KA co-cultured epithelial cells with MSCs, and found that MSCs from lung tissue can transfer mitochondria to bronchial epithelial cells through multiple mechanisms, contributing to the repair of damaged bronchial epithelial cells[186]. In vivo, reported experimental animal studies demonstrate that mitochondria transfer serves as functional treatments in various respiratory diseases.
Asthma is a chronic respiratory disease, in which exosomes may mediate important immunomodulatory roles in asthma. To explore the potential mechanism of immune regulation by airway myeloid-derived regulatory cells (MDRC) in asthma, Hough KP transferred mitochondria to T cells through exosomes. Their data indicated that the mitochondria transfer between airway myeloid-derived regulatory cells represented a new immunomodulatory pattern in asthma, and also confirmed that exosomes were likely involved in the immune response of MDRCs in asthma[187]. This study provides a theoretical basis for the development of treatments for asthma targeting inflammatory responses. Moreover, chronic obstructive pulmonary disease (COPD), as the eighth leading cause affecting global health, is a common chronic disease characterized by airflow obstruction. Reported studies show that there are variations in the mtDNA genome in the lung tissue and peripheral blood of patients with COPD, and these variations may lead to abnormal mitochondrial functions[188]. Frankenberg GJ transferred mitochondria from MSCs to airway smooth muscle cells and found that they could regulate bioenergetics and cellular functions[189]. This also proves that the uptake of MSCs-derived mitochondria by airway smooth muscle cells can reverse mitochondrial dysfunction induced by oxidative stress, providing a theoretical basis that mitochondrial transfer can be used as a research method to explore mitochondrial dysfunction in COPD. Furthermore, pulmonary fibrosis (PF) is a serious lung disease characterized by fibroblast proliferation and a large amount of extracellular matrix deposition. Previous studies have shown that mitochondrial dysfunction in lung cells is one of the main causes promoting PF[190]. The use of exogenous mitochondria to supplement damaged mitochondria has been proposed as a strategy for treating PF. Huang T treated human placenta-derived MSCs with a combination of pioglitazone (Pg) and iron oxide nanoparticles, which could effectively transfer mitochondria to damaged lung cells and restore mitochondrial homeostasis[191]. In addition, Zhang F found that the regulation of TFAM expression in MSCs plays a key role in improving the permeability barrier of pulmonary microvascular endothelial cells (PMVECs) by mediating mitochondria transfer through TNTs, providing a new therapeutic strategy for the treatment of acute lung injury caused by sepsis[192].
Liver
The liver is an important metabolic organ responsible for detoxification, bile production, nutrient storage, and regulation of diverse metabolic processes. Mitochondrial quality control (MQC) plays a crucial role in maintaining liver health and function, and its disruption has been implicated in a wide range of liver diseases[193]. For example, impaired mitochondrial function is associated with hepatic lipid accumulation, oxidative stress, insulin resistance, and inflammation[194]. These pathological factors contribute to the development and progression of liver diseases such as nonalcoholic fatty liver disease (NASH), alcohol-associated liver disease (ALD), drug-induced liver injury (DILI), and viral hepatitis. Studies have shown that the AMPK pathway plays a key role in regulating hepatic energy metabolism and cellular viability[195], and transferring healthy mitochondria can improve the symptoms of metabolic syndrome, including hypertension, hyperlipidemia, and fatty liver[196].
Mitochondria-targeted interventions have emerged as promising therapeutic strategies for NASH, focusing on improving energy metabolism, antioxidant effects, and mitochondrial quality control. MSCs transfer mitochondria to liver cells through TNTs, which can further reduce the lipid load of the liver, promote the transformation of lipids from storage to utilization, and improve the ability of tissue homeostasis[196, 197]. In arsenic-induced liver injury, functional mitochondria were transferred between cells through TNTs, achieving mutual supplementation of mtDNA between cells. This compensated for the mitochondrial dysfunction caused by arsenic and hepatocyte senescence and liver damage[198]. Umbilical cord-derived MSCs (UC-MSCs) have been found to alleviate liver damage by restoring mitochondrial functions through activation of the Nrf2/HO-1 pathway[199]. In hepatic ischemia-reperfusion injury, MSC-derived EVs also deliver functional mitochondria to neutrophils, repair their mitochondrial function, and inhibit local neutrophil extracellular traps (NETs) formation, exhibiting significant nanotherapeutic effects[200]. Additionally, HMT occurs between hepatocellular carcinoma (HCC) cells via TNTs; the transfer of mitochondria from highly invasive to less invasive cells can increase the migratory and invasive capacity of recipient cells, with hypoxia further promoting this process[201].
Kidney
The kidney is one of the most energy-consuming organs in the human body, second only to the heart in mitochondrial content and oxygen consumption[202, 203]. Due to the exceptionally high oxidative activity of renal mitochondria, the kidney is highly susceptible to oxidative stress damage, thereby promoting the onset and progression of kidney diseases and potentially resulting in renal failure. Mitochondrial dysfunction inevitably leads to energy deficiency in renal tubular epithelial cells, consequently causing renal dysfunction[204]. Mitochondrial dysfunction in kidney cells can lead to various pathological conditions, including acute kidney injury (AKI) and chronic kidney disease (CKD), by affecting cellular energetics, redox balance, and cell death pathways[203, 205]. Therefore, maintaining mitochondrial homeostasis and quality control is essential for preserving normal renal functions.
Mitochondria play a central role in the pathogenesis and progression of kidney diseases primarily through several mechanisms, including the regulation of ROS production, immune responses, and energy metabolism[206, 207]. Recent studies have revealed that EVs possess multiple biological effects in kidney diseases, such as anti-inflammatory, anti-apoptotic, pro-angiogenic, and anti-fibrotic actions[208, 209]. Furthermore, Naoto K showed that BM-MSCs improve impaired proximal tubular epithelial cells (PTECs) in diabetic nephropathy (DN) via mitochondria transfer[210]. In this study, transferred mitochondria inhibited PTECs apoptosis, regulated related factors to inhibit ROS production, restored transporter expression, and repaired renal tubular structure. Moreover, MSCs also transfer mitochondria to macrophages, enhancing their anti-inflammatory properties and alleviating kidney injury in diabetic nephropathy through PGC-1α activation[211].
Musculoskeletal system
Mitochondria play a crucial role in the musculoskeletal system, influencing cellular metabolism, energy production, and homeostasis. Mitochondrial dysfunction has been implicated in various musculoskeletal conditions, including rheumatic diseases, age-related muscle loss, and osteoporosis[212]. In recent years, research on HMT within the musculoskeletal system has primarily focused on osteocytes and chondrocytes.
In osteocytes, mitochondria transfer occurs through dendritic networks, mediated by endoplasmic reticulum (ER)-mitochondrial contact and regulated by Mitofusin 2 (Mfn2)[116]. Osteocytes exchange mitochondria through interconnected dendritic networks[116], and this feed-forward mechanism promotes bone matrix formation and restores metabolic function in stressed cells. Moreover, clinical data showed that bone is one of the most common sites of tumor metastasis. During bone metastasis, osteocytes transfer mitochondria to metastatic cancer cells and trigger cGAS/STING-mediated anti-tumor responses[116, 213].
In chondrocytes, mitochondrial dysfunction is closely associated with cartilage damage. Elevated mitochondrial electron transport chain activity and increased ROS levels in injured chondrocytes result in excessive free radical and metabolite release, thereby triggering apoptosis and necrosis[214]. Due to the avascular nature of cartilage, its intrinsic ability for self-repair is limited, making restoration of chondrocyte function particularly critical for tissue repair[215]. MSC transplantation not only alleviates symptoms of osteoarthritis (OA) but also restores mitochondrial functions in damaged cells by transferring mitochondria to chondrocytes through EVs or GJs[101, 216]. The transfer of healthy mitochondria from MSCs to OA chondrocytes has been demonstrated to improve mitochondrial function, reduce cell apoptosis, and increase the production of cartilage-specific proteins[217]. In addition, mitochondrial transplantation can upregulate the expression of PGC-1α, downregulate Mfn2 expression, and increase mitochondrial DNA content, suggesting that mitochondria transfer promotes both mitochondrial function and biogenesis in chondrocytes[218].
Besides osteocytes and chondrocytes, recent studies have shown that macrophages can enhance the osteogenic differentiation of MSCs through mitochondria transfer and regulation of ROS. Macrophages deliver functional mitochondria to MSCs, promoting their osteogenic differentiation and regulating bone homeostasis[219]. Moreover, osteoblasts transfer mitochondria to osteoprogenitor cells, stimulating their differentiation into mature osteoblasts[128]. In short conclusion, mitochondrial transfer in musculoskeletal system not only facilitates the osteogenic differentiation and bone formation, but also prevents osteocyte damage and suppresses chondrocyte apoptosis, highlighting its therapeutic potential in skeletal and joint diseases.
Adipose tissue
Adipose, a connective tissue composed of mature adipocytes, fibroblasts, endothelial cells, macrophages, matrix cells, immune cells, and mesenchymal stem cells, plays a key role in regulating pathophysiological processes by influencing insulin sensitivity, blood pressure, endothelial function, fibrinolytic activity, and inflammatory responses[220]. This is dependent on both its endocrine capacity and intercellular mitochondrial communication. Under normal conditions, adipocytes can share their mitochondria with adjacent macrophages in white adipose tissue, promoting the differentiation of specific macrophage subgroups and supporting metabolic homeostasis. The ability of macrophages to take up mitochondria may be related to the expression level of heparan sulfate on their cell surface[221]. Moreover, macrophages also transfer mitochondria to adipocytes via EVs. Studies showed that M2 macrophages can transfer more mitochondria to adipocytes than M1 macrophages, promoting adipocyte-myofibroblast transition through the activation of TGF-β and PAI-1 pathways[222]. However, under obesity conditions, adipocytes release EVs containing damaged mitochondria in response to intense energy stress. These vesicles are taken up by cardiomyocytes through the circulatory system. The EV-Mito induce transient mitochondrial oxidative stress in cardiac tissue, preconditioning the heart and protecting it from ischemia or reperfusion injury[127].
Growing evidence has shown that mitochondria from adipose-derived mesenchymal stem cells (ADSCs) are beneficial for certain diseases. For instance, ADSCs connected with macrophages through TNTs to deliver mitochondria to synovial macrophages, alleviating rheumatoid arthritis[223]. Moreover, ADSCs transferred mitochondria to dendritic cells (DCs) via EVs. After internalizing the functional mitochondria from EVs, DCs activated the MAPK/ERK1/2/FOXO1/autophagy pathway, altering cell metabolism. This reprogramming facilitated the shift of DCs from an activated state to a tolerant state, thereby reducing inflammation[224]. In an in vitro insulin secretion study, ADSC mitochondria were transferred to pancreatic islet cells via TNTs to improve insulin secretion function[225]. Moreover, mitochondria were transferred through TNTs into rat cardiomyocytes three days after ADSCs were transplanted onto the surface of ischemic hearts, improving the cardiac function in rat models of ischemic cardiomyopathy[226]. However, the HMTs of ADSCs is not always beneficial, but displays a harmful effect. For example, ADSCs cocultured with different breast cancer cells (BCCs) transferred mitochondria to BCCs via TNTs, increasing ATP production and driving ABC transporter-mediated multidrug resistance (MDR)[227].
Reproductive system
The reproductive system plays a pivotal role in species continuation and genetic transmission. Mitochondria, serving as the cellular powerhouses and subject to maternal inheritance, critically influence gamete quality and embryonic development. Age-associated mitochondrial dysfunction is a significant contributor to declining gamete quality and infertility. Recent studies demonstrates that autologous mitochondrial transplantation (MRT) - the supplementation of viable mitochondria into aged or compromised oocytes - is an effective strategy for rescuing fertility potential.
In aged mouse models, transplantation of mitochondria derived from autologous sources such as adipose-derived stem cells (ADSCs)[228], endometrial mesenchymal stem cells (EnMSCs)[229], or induced pluripotent stem cells (iPSCs)[230] significantly enhanced oocyte quality, promoted embryonic development, and increased live birth rates. Human studies further evaluated mitochondria from various autologous stem cells (ADSCs, BMSCs, urine-derived stem cells (USCs), and ovarian granulosa cells (GCs)). Notably, mitochondria from USCs exhibited superior characteristics, including a characteristic non-fused spherical morphology, high abundance, low ROS levels, and a robust coupled metabolic profile reliant on both glycolysis and oxidative phosphorylation (OXPHOS). Following transplantation into human oocytes, USC mitochondria effectively restored mitochondrial content and function, ameliorated metabolic status, improved embryonic euploidy rates, and supported normal embryonic development[231]. Furthermore, MRT has proven effective in mitigating mitochondrial damage and oxidative stress induced by oocyte cryopreservation - a routine procedure in assisted reproductive technology (ART) - thereby restoring developmental competence and live birth potential of frozen-thawed oocytes[232].
These collective findings highlight the considerable promise of autologous mitochondrial transplantation, particularly utilizing optimized sources like USCs, for: (i) improving the function of aged or damaged oocytes, (ii) treating associated infertility, and (iii) enhancing the success rates of ART procedures, including oocyte cryopreservation.
In short summary, HMT has been emerged as a promising mechanism for tissue revitalization, offering potential therapeutic strategies for treating tissue damage and degeneration. Understanding the dual nature of mitochondria transfer in health and disease provides new opportunities for targeted intervention. Mitochondria transfer has a unique advantage in the study of tissue homeostasis and in the treatment of systematic diseases, providing researchers with a new perspective to explore treatment plans for various diseases.
Functions of mitochondria transfer
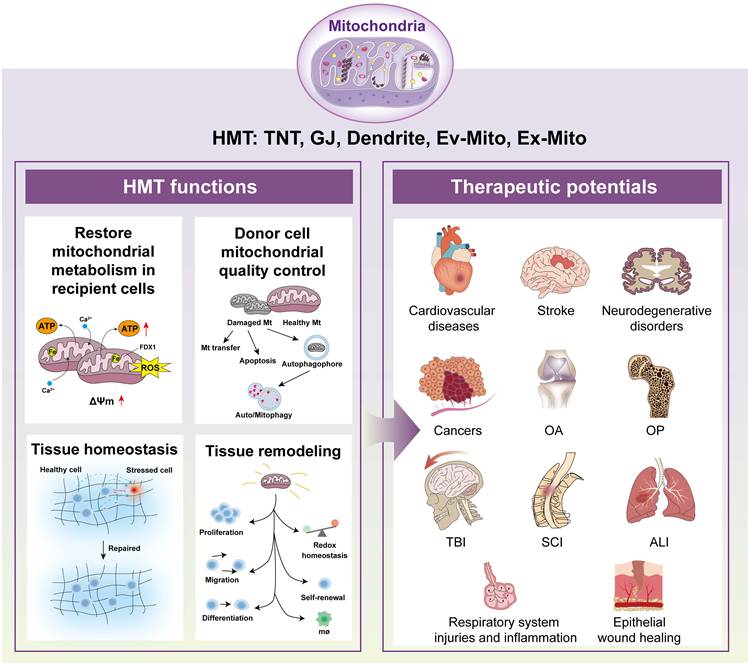
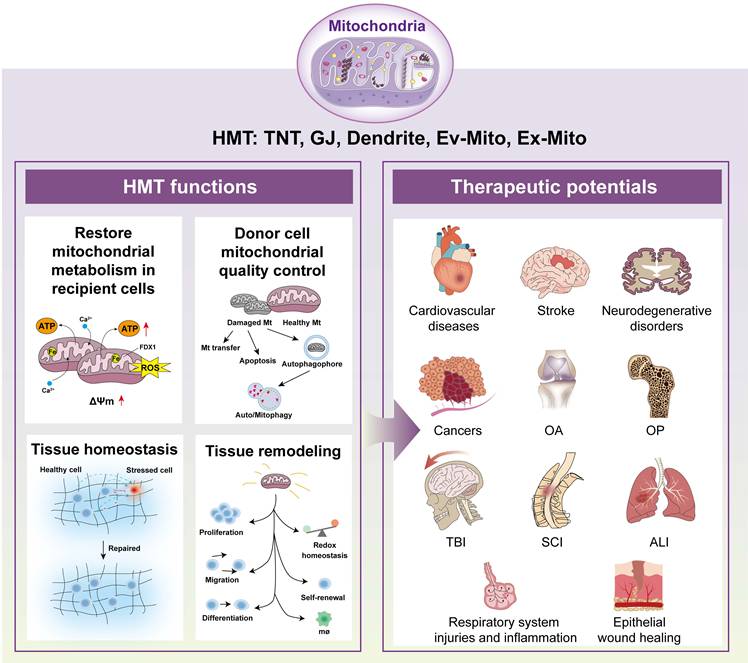
The dynamic intercellular exchange of mitochondria plays a crucial role in cell and tissue physiology[95,233]. Mitochondria transfer serves multiple functions, including restoring mitochondrial metabolism, maintaining donor cell quality control, promoting tissue homeostasis, and promoting tissue remodeling[77]. In the following sections, we explore these roles in detail and schematically summarize the mechanisms and outcomes of mitochondrial transfer on recent advances (Figure 3).
Schematic diagram of functions and therapeutic potentials of mitochondria transfer. Mitochondria transfer has been shown with essential functions in the pathology and treatment of various tissues and diseases. These functions include restoration mitochondrial metabolism in recipient cells, donor cell mitochondria quality control, tissue homeostasis and tissue remodeling, which have been demonstrated with essential roles in various disease pathogenesis and treatment. Abbreviations: VMT, vertical mitochondria transfer; HMT, horizontal mitochondria transfer; TNTs, tunneling nanotubes; GJs, gap junctions; Ev-Mito, extracellular vesicle-associated mitochondria transfer; Ex-Mito, extracellular mitochondria transfer; OA, osteoarthritis; OP, osteoporosis; TBI, traumatic brain injury; SCI, spinal cord injury; ALI, acute lung injury.
Restore mitochondrial metabolism in recipient cells
HMT represents a fundamental mechanism by which donor cells—including MSCs, astrocytes, and immune cells—restore or augment mitochondrial function in stressed or injured recipient cells. This process rescues mitochondrial metabolism by replenishing the mitochondrial pool, restoring membrane potential, ATP production, and reducing oxidative stress, thereby mitigating cell death across various pathological contexts such as myocardial infarction, stroke, acute lung injury, and neurodegenerative disorders[2, 234]. Transferring mitochondria from neighboring cells or foreign cells can alleviate mitochondrial dysfunction and restore its biological function[235, 236]. Transfer of mitochondria or mitochondrial components from MSCs or their EVs to alveolar cells restores bioenergetic function, as evidenced by increased alveolar ATP levels, enhanced mtDNA content, improved mitochondrial membrane potential, and elevated OXPHOS activity. Notably, only EVs containing mitochondria confer significant protection in models of lung injury[107, 147, 237].
Mitochondria transfer to recipient cells can restore mitochondrial metabolism by replacing damaged mitochondria with healthy ones, thereby improving energetic balance, calcium and iron homeostasis, and reducing oxidative stress—functions that are particularly beneficial in neurodegenerative disease[238]. For example, CD8⁺T cells acquiring exogenous mitochondria display enhanced mitochondrial respiration and spare respiratory capacity, leading to improved expansion, tumor infiltration, and reduced exhaustion in tumor-bearing hosts[239]. Moreover, intercellular nanotube-mediated mitochondria transfer from immune cells to cancer cells markedly enhances mitochondrial metabolism in recipient cancer cells, as reflected by increased mitochondrial respiration, basal respiration, and spare respiratory capacity[153]. Traumatic brain injury impairs mitochondrial respiration in recipient neurons, resulting in reduced oxygen consumption and compromised ATP production[240]. These findings suggest that donor cells can induce long-term behavioral changes in recipient cells or tissues by transferring relatively small numbers of mitochondria.
Donor cell mitochondrial quality control
In addition to being involved in supporting the metabolism of recipient cells, mitochondria transfer also serves as a quality control mechanism for donor cells, allowing them to maintain their own mitochondrial health[95]. By offloading damaged or dysfunctional mitochondria into recipient cells or extracellular vesicles, donor cells can reduce oxidative stress and prevent the accumulation of defective organelles[241]. This process is tightly regulated and often involves mitophagy pathways, where damaged mitochondria are tagged for transfer or degradation[242]. This process has been observed in stem cells, neurons, immune cells, and cancer cells, and may involve mechanisms such as mitophagy, autophagy, and lysosomal degradation. For example, brown adipocytes and cardiomyocytes release damaged mitochondrial components in extracellular vesicles, which are subsequently captured and degraded by tissue-resident macrophages[243, 244]. Neurons can also release damaged mitochondria and transfer them to astrocytes for disposal and recycling[119]. During acute ischemic stroke, loss of blood flow impairs mitochondrial oxidative phosphorylation and causes bioenergetic stress. HMT helps maintain mitochondrial function and supports neurovascular unit homeostasis[245]. This intercellular mitochondrial quality control adds a new layer to the regulation of cellular homeostasis and may underlie certain aspects of stem cell function and tumor progression.
Tissue homeostasis
Beyond individual cellular effects, mitochondria transfer contributes to the maintenance of tissue homeostasis. By distributing functional mitochondria among heterogeneous cell populations, tissues can buffer local metabolic fluctuations, enhance stress resistance, and facilitate coordinated responses to injury or disease. In the central nervous system, astrocyte-to-neuron mitochondria transfer has been implicated in neuroprotection and functional recovery after ischemic insult. In hematopoietic and immune tissues, HMT helps balance redox status, sustain stem cell self-renewal, and regulate immune cell activation —processes essential for preserving tissue architecture and function under both physiological and pathological conditions.
Emerging data also indicate a role for mitochondria transfer in tissue regeneration and repair, promoting survival and proliferation of resident or recruited progenitor cells. For example, under healthy conditions, adipocytes transfer mitochondria to macrophages in white adipose tissue via macrophage surface heparan sulfate, supporting energy homeostasis[221]. Disruption of this pathway—such as during obesity or high dietary long-chain fatty acid exposure—impairs mitochondrial transfer, leading to metabolic dysfunction and release of adipocyte-derived mitochondria into the circulation, where they help counteract stresses such as metabolic and ischemic stress in distant organs such as the heart[137].
Mitochondria transfer is also involved in disease progression and immune regulation. Transfer of mitochondria harboring cancer cell-specific mtDNA mutations to tumor-infiltrating lymphocytes induces metabolic dysfunction and senescence in T cells, impairs memory formation, and compromises antitumor immunity, thereby facilitating immune evasion[246]. In addition, through the PINK1/Parkin-Mfn2 pathway, cancer cells acquire platelet mitochondria, which reprogram them toward a metastatic phenotype by modulating the GSH/GSSG ratio and ROS levels, thereby promoting metastasis of osteosarcoma[247]. Mitochondrial transfer from MSCs restores tumor cell mitochondrial function, driving chemoresistance, heterogeneity, and metastasis in several cancers[248-250].
Tissue remodeling
Mitochondria transfer is increasingly recognized as a central mechanism in tissue remodeling—a process fundamental to development, wound healing, and adaptation to physiological stress. During tissue remodeling, cells undergo metabolic reprogramming, with mitochondria transfer providing critical energy and signaling cues to support cellular proliferation, migration, and differentiation, particularly in regenerative contexts. Notably, adipocytes have been shown to extrude damaged mitochondria into the extracellular space, where they may circulate and be taken up by recipient cells, thereby shaping the local microenvironment and modulating tissue remodeling[251].
Emerging studies have further elucidated the complexity of mitochondria transfer in various tissue contexts. For example, platelets enhance the wound-healing capacity of MSCs by transferring platelet-derived mitochondria and inducing metabolic reprogramming[252]. Moreover, in the context of bone homeostasis, mitochondria transfer from macrophages to MSCs regulates the metabolism and osteogenic differentiation of MSCs, whereas impaired mitochondria transfer in osteoporosis disrupts this process and contributes to disease pathology[253]. These findings highlight the dual role of mitochondria transfer in tissue remodeling, with implications for both regenerative and disease processes.
Therapeutic potentials of mitochondria transfer in diseases
Mitochondria transfer has emerged as a promising therapeutic strategy in a wide spectrum of diseases, spanning from rare mitochondrial disorders to common conditions such as cancer, tissue injury, and aging[254]. Harnessing the power of healthy mitochondria to restore metabolic capacity, modulate cellular signaling, and reshape tissue microenvironment, mitochondria-related therapeutics are reshaping translational medicine. In this section, we review the main disease categories associated with mitochondria transfer, highlight its current preclinical models and clinical therapeutic applications, and discuss key organ systems and clinical settings in which mitochondria transfer is under intensive investigation (Table 2).
Therapeutic impact of mitochondria transfer in pathological conditions.
| Pathological conditions | Disease/ injury models | Mitochondria/ Treatment | Mode/Route of transfer | Outcomes | Ref. |
|---|---|---|---|---|---|
| Inherited mitochondrial diseases | Friedreich ataxia, LHON, MELAS, MERRF, Leigh syndrome, mtDNA depletion syndromes, MNGIE | Mitochondrial transplantation/exogenous mitochondrial delivery Supplementation (CoQ10, idebenone, EPI-743) High-dose L-arginine (MELAS) Allogeneic stem cell transplantation, enzyme replacement (MNGIE) Deoxynucleoside supplementation (mtDNA depletion) Rapamycin (autophagy/mitophagy modulation) NAD⁺ enhancement (nicotinamide riboside) | Mitochondrial transplantation, exogenous mitochondrial delivery Oral or intravenous drug/supplement therapy Cell transplantation | Improved mitochondrial function and metabolism Reduced stroke-like episodes (MELAS) Improved survival and quality of life Correction of underlying defects in preclinical models | [255-261] |
| Cancers | Tumor/cancer models (including preclinical, T cell/CAR-T cell models) | MSC/fibroblast-derived mitochondria TNT inhibitors (e.g., vincristine), selective mitochondrial depletion TrxR2 inhibition MSC mitochondrial transfer to Treg or CAR-T cells | TNTs, EVs Pharmacological/genetic blockade Pharmacological inhibition HLA-dependent cell contact | Restored mitochondrial respiration Reduced tumor growth, increased therapy sensitivity Promoted cancer cell apoptosis Enhanced immunosuppressive function of Tregs | [263-267] |
| Wound healing | Diabetic wound models, diabetic rats with pressure sores, full-thickness skin wounds in mice, cranial defect models | MSC-derived EVs and small EVs carrying mitochondria HucMSC-derived exosomes (HucMSC-Exo) Mitochondria-loaded microvesicles (Mito@euMVs) VEGF-loaded AAV encapsulated in HucMSC-exosome hydrogel Engineered/optimized MSCs for mitochondrial transfer | EV-mediated or exosome injection Microneedle transdermal delivery Hydrogel-based gene delivery Direct cell engineering/local transfer | Restored neutrophil and endothelial mitochondrial function Suppressed NETs and ferroptosis Restored calcium homeostasis Inhibited cell senescence Enhanced angiogenesis and vascularization Accelerated wound/bone defect healing and tissue regeneration | [269-274, 290] |
| Respiratory system: injury and inflammation | ALI (e.g., LPS-induced), COPD models, viral pneumonia in mice, postnatal alveolar development | MSC- or macrophage-derived mitochondria Mitochondrial complex I-dependent NAD⁺ regeneration Plant nanovesicles (Artemisia annua) delivering GABA | TNTs, EVs, microvesicles Nanovesicle-mediated delivery Cell-based or pharmacological modulation | Restored ATP and mitochondrial bioenergetics Reduced inflammation and oxidative stress Repaired epithelial barrier Improved survival and cell fate Decreased cytokine release and apoptosis | [107, 147, 275-278] |
| Central nervous system (CNS) injury | Ischemic stroke, traumatic brain injury (TBI), spinal cord injury (SCI) | Exogenous healthy mitochondria transplantation MSC-based mitochondrial transfer Mitochondria-targeted therapies Cell engineering approaches | Direct mitochondrial transplantation Stem cell-mediated mitochondrial transfer Cell engineering to enhance delivery | Restored ATP levels and aerobic respiration Reduced energy deficits and apoptosis Supported axonal regeneration and functional recovery Improved therapeutic outcomes | [279-286] |
| Other diseases | 1.Muscle injury;2. OA chondrocytes (in vitro), collagenase-induced OA in mice;3. Injured pancreatic acinar cells (severe acute pancreatitis); 4.Tendinopathy (in vitro & in vivo) | Systemic mitochondrial delivery MSC-derived mitochondria/microtubules Hypoxia-conditioned MSC-derived mitochondria Bone marrow MSC-derived healthy mitochondria | Systemic administration Intra-articular injection Extracellular vesicles (EVs) Direct transfer | Enhanced muscle regeneration and function Restored energy status & mitochondrial dynamics increased resistance to oxidative stress & apoptosis Protected against cartilage degeneration Reprogrammed cell metabolism, alleviated pancreatitis injury Restored mitochondrial function, promoted tendon healing | [287, 288, 291, 292] |
Inherited mitochondrial diseases
Inherited mitochondrial diseases are caused by genetic mutations affecting mtDNA or nuclear-encoded mitochondrial genes, resulting in impaired oxidative phosphorylation and multisystem dysfunction[255]. Disorders such as Friedreich's ataxia (FA), Leber hereditary optic neuropathy (LHON), Mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS), Myoclonic epilepsy with ragged red fibers (MERRF), and Leigh syndrome (LS) typically affect high-energy-demand tissues, leading to neurological, muscular, and cardiac symptoms. Traditional treatments are mostly supportive and have limited efficacy in treating the underlying mitochondrial dysfunction[256].
Recent advances in mitochondrial transplantation and exogenous mitochondrial delivery have shown promising results in preclinical models, providing a foundation for the development of novel therapies targeting the root causes of mitochondrial diseases. Current supplementation therapies, such as coenzyme Q10, idebenone, and EPI-743, aim to enhance mitochondrial electron transport and alleviate symptoms in selected disorders. Disease-specific interventions include high-dose L-arginine for MELAS to reduce stroke-like episodes[257], allogeneic stem cell transplantation and enzyme replacement for mitochondrial neurogastrointestinal encephalopathy[258], and deoxynucleoside supplementation for mtDNA depletion syndromes[259]. In addition, emerging experimental approaches focus on improving mitochondrial quality control and metabolism—for example, modulation of autophagy and mitophagy using rapamycin[260], or enhancement of the NAD+ pathway with nicotinamide riboside[261]. Together, these therapies aim to alleviate symptoms, improve quality of life, and address the underlying mitochondrial dysfunction.
Non-inherited mitochondria-related diseases
Emerging evidence suggests that mitochondria transfer also plays a crucial role in acquired and degenerative diseases[262]. In conditions such as cancer, wound healing, ischemic injury, chronic inflammation, and neurodegenerative diseases, intercellular mitochondrial exchange shapes disease progression and recovery. The therapeutic impact of mitochondria transfer in the following several key pathological conditions have been extensively studied in recent years.
Cancers
Cancer cells often exploit mitochondria transfer to enhance metabolic plasticity, evade apoptosis, and acquire resistance to chemotherapy or radiotherapy. Tumor-associated stromal cells, including MSCs and fibroblasts, have been shown to donate mitochondria to cancer cells via TNTs or EVs, thereby restoring mitochondrial respiration and supporting rapid proliferation[263]. Importantly, inhibiting pathological mitochondria transfer—through genetic or pharmacological means—reduces tumor growth and increases therapeutic sensitivity in preclinical models. Strategies such as selective mitochondrial depletion or disruption of TNT formation with microtubule inhibitors like vincristine (VCR) effectively impair the “rescue” function of activated MSCs within the tumor microenvironment[264]. Redox regulation is also intricately involved in this process. Thioredoxin reductase 2 (TrxR2), a key mitochondrial redox enzyme frequently overexpressed in cancer cells, confers resistance to apoptosis. Selective inhibition of TrxR2 increases mitochondrial ROS and drives the redox state toward oxidation, thereby promoting cancer cell apoptosis[265].
Beyond pharmacological interventions, cell engineering is emerging as a promising strategy in tumor therapy. Recent work by Piekarska et al. demonstrates that MSCs can transfer active mitochondria to allogeneic regulatory T cells in an HLA-dependent manner, augmenting their immunosuppressive function and therapeutic potential[266]. Similarly, Court et al. reveal that mitochondria transfer from MSCs protects both native and engineered CAR-T cells from apoptosis, enhancing their metabolic fitness and persistence, and offering a compelling approach to improve the efficacy of T cell-based immunotherapies[267]. Recently, Zhang H et al. introduced a statistical deconvolution method for tracing and quantifying mitochondrial trafficking between cancer and T cells at single-cell level, which largely empowered MT research in a higher resolution[268].
Collectively, these findings underscore the complex role of mitochondria transfer in cancer biology, highlighting both its contribution to tumor progression and its potential as a target for innovative therapeutic strategies.
Wound healing
Mitochondria transfer is increasingly recognized as a pivotal mechanism underpinning tissue repair and regeneration. MSCs, in particular, play an essential role in this process. Through advanced cell engineering approaches, MSCs can be optimized to transfer their mitochondria to local cells at sites of injury, resulting in elevated ATP production, enhanced cellular proliferation and migration, and accelerated tissue repair. This highlights the promise of mitochondria transfer as an innovative strategy for developing next-generation cell-derived therapies in regenerative medicine[269]. Recent studies have further elucidated the multifaceted mechanisms by which MSC-derived products exert their regenerative effects. For example, exosomes isolated from human umbilical cord MSCs (HucMSC-Exo) and injected into full-thickness skin wounds in mice have been shown to reprogram neutrophil mitochondrial metabolism, induce N2 neutrophil polarization, and thereby promote angiogenesis and enhance tissue regeneration[270]. EVs derived from MSCs also serve as efficient vehicles for mitochondria transfer in pathological settings. In diabetic wound models, MSC-derived EVs have been shown to deliver functional mitochondria to neutrophils, restoring their mitochondrial function, suppressing neutrophil extracellular trap (NET) formation and endothelial cell ferroptosis, and ultimately accelerating angiogenesis and wound healing[271]. Moreover, small extracellular vesicles derived from MSCs can restore TRPC6 channel activity and mitochondrial functions, correct calcium homeostasis, and significantly promote healing in diabetic wounds[272].
Furthermore, innovative delivery systems are also emerging. For instance, Dong et al. developed functional mitochondria-loaded microvesicles (Mito@euMVs) from enucleated MSCs using a simple extrusion process. Transdermal delivery of these Mito@euMVs via microneedle patches to diabetic rats with pressure sores effectively inhibited and reversed hyperglycemia-induced cellular senescence, thereby facilitating chronic wound healing[273]. Likewise, Shan He et al. created a biomimetic gene delivery platform combining VEGF-loaded, extracellular vesicle-encapsulated adeno-associated virus (AAV) with human umbilical cord MSC-derived exosomes embedded in a hydrogel. Application of this system to diabetic wound models improved mitochondrial function in endothelial cells, enhanced vascularization, and promoted chronic wound healing by modulating the inflammatory microenvironment[274]. Collectively, these advances underscore the therapeutic potential of mitochondria transfer, whether via direct mitochondrial donation, engineered vesicles, or advanced delivery systems, in the treatment of chronic and acute wounds. As our understanding of mitochondrial dynamics and intercellular communication deepens, mitochondria transfer is poised to become a transformative strategy in regenerative medicine.
Respiratory injury and inflammation
Mitochondria transfer is a critical therapeutic tool for respiratory injuries and inflammatory conditions, such as acute lung injury (ALI) and chronic obstructive pulmonary disease (COPD). Mitochondrial complex I-dependent NAD+ regeneration, potentially influenced by mitochondria transfer, is essential for proper lung epithelial cell fate during postnatal alveolar development by preventing pathological activation of the integrated stress response[275]. In ALI, MSCs transfer mitochondria to damaged alveolar epithelial cells via TNTs or EVs, restoring ATP production, reducing inflammation, and repairing the epithelial barrier[107, 147]. Notably, BMSCs airway-instilled into mice with LPS-induced acute lung injury formed Cx43-dependent GJs with alveolar epithelial cells and transferred mitochondria via microvesicles, thereby increasing alveolar ATP levels and significantly reducing lung injury and mortality[107]. In COPD, mitochondria transfer from MSCs or macrophages protects lung cells against oxidative stress-induced mitochondrial dysfunction and apoptosis and reduces pro-inflammatory cytokine release[276, 277]. Beyond cell-based approaches, pharmacological modulation of mitochondrial function has demonstrated substantial promise for improving local inflammation and tissue repair. For instance, in mouse models of ALI and viral pneumonia, nanovesicles derived from the medicinal plant Artemisia annua have been used to deliver gamma-aminobutyric acid (GABA) to alveolar macrophages, thereby enhancing mitochondrial bioenergetics, dampening lung inflammation, and improving survival outcomes[278]. These findings suggest that mitochondria transfer could transform the management of acute and chronic respiratory diseases.
Central nervous system (CNS) injury
Mitochondrial dysfunction has been identified as a seemingly unifying pathological phenomenon across a wide range of neurodegenerative disorders. Recent studies have shown that mitochondrial transplantation has been demonstrated to replace damaged or dysfunctional mitochondria with exogenous healthy mitochondria for CNS injuries, including stroke, traumatic brain injury (TBI), and spinal cord injury (SCI). The transfer of healthy mitochondria into injured cells has been shown to restore ATP levels and reduce energy deficits following ischemic stroke[279]. Stem cell-based mitochondria transfer thus represents a promising therapeutic strategy for stroke[280]. After ischemia-reperfusion injury, MSCs maintain aerobic respiration and inhibit apoptosis in damaged endothelial cells through mitochondria transfer[280]. Several preclinical and clinical studies have evaluated mitochondria-targeted therapies, indicating that stem cell-based treatments and mitochondrial transplantation are promising novel biotherapeutic strategies for the treatment of traumatic brain injury (TBI)[281-283]. In SCI, mitochondria transfer from MSCs to damaged neurons supports axonal regeneration and functional recovery[284-286]. These approaches are complemented by cell engineering strategies, enhancing delivery efficiency, offering hope for CNS repair.
Other diseases
In addition to the common diseases mentioned above, mitochondria transfer holds significant potential for the treatment of various other pathological conditions and the maintenance of systemic homeostasis. Alway et al. demonstrate that systemic mitochondria delivery can enhance the rate of muscle regeneration and recovery of muscle function after injury[287]. Moreover, in a collagenase-induced mouse model of osteoarthritis, intra-articular injection of MSC-derived mitochondria protects against cartilage degeneration by restoring chondrocyte energy metabolism and mitochondrial dynamics, while enhancing resistance to oxidative stress and apoptosis[288]. In a study focused on the autoimmune tissue inflammation of rheumatoid arthritis, Wu B et al. reported that transfer intact mitochondria into T cells, as well as supplementation of exogenous aspartate, rescued the mitochondria-instructed expansion of ER membranes and suppressed TNF release and rheumatoid tissue inflammation[289]. Similarly, the transfer of mitochondria to BMSCs, followed by in situ implantation at cranial defect sites, has been demonstrated to significantly promote bone defect repair[290]. Moreover, published study demonstrates that MSCs deliver hypoxia-conditioned functional mitochondria to injured pancreatic acinar cells via EVs, reprogramming their metabolism and alleviating severe acute pancreatitis injury[291]. Wei et al. demonstrates that BMMSCs protect against tendinopathy by transferring healthy mitochondria to injured tenocytes both in vitro and in vivo, thereby restoring mitochondrial functions and promoting tendon healing[292].
Current clinical trails
To further review the current clinical applications and outcomes in this field, registered clinical trials and published first-in-human or pilot clinical experience related to mitochondrial transplantation and mitochondria transfer-based therapies were examined and evaluated (Table 3). During data searching and literature review, we found that mitochondria-based interventions have been registered in ClinicalTrials.gov across multiple contexts, including acute ischemic stroke[293], cardiac ischemia or extracorporeal membrane oxygenation (ECMO)-associated myocardial injury (NCT02851758), and inflammatory myopathies (NCT04976140). Moreover, autologous mitochondrial transplantation is currently being evaluated in a registered clinical study for cerebral ischemia (NCT04998357), sponsored by the University of Washington. This ongoing trial further indicates that mitochondrial transplantation has progressed beyond preclinical investigation and is under active clinical evaluation. In addition, a pediatric postcardiotomy cardiogenic shock or ECMO setting has published pilot outcomes associated with autologous mitochondrial transplantation approaches[294]. Collectively, these records support that mitochondria transplantation and transfer-based therapies have entered early clinical exploration, while the current evidence base remains predominantly early-phase, small sample, and frequently non-randomized, and therefore efficacy conclusions remain premature.
Current mitochondria-based therapeutic treatments in clinical trails.
| Diseases | Mt sources and approach | Sample size | Phase / status | Outcomes | Clinical trial# |
|---|---|---|---|---|---|
| Acute ischemic stroke | Autologous mitochondria;intra-arterial/endovascular delivery to ischemic territory. | Early-phase feasibility | Phase 1; recruiting | No major safety signal reported in Phase 1 publication. | Clinical trial NCT04998357 [293] |
| Pediatric cardiac ischemia | Autologous mitochondria; direct injection or infusion to myocardium. | Registry lists pediatric ECMO; publication pilot cohort | Phase NA; unknown | Pilot outcomes reported in pediatric cardiogenic shock/ECMO context. | Clinical trial NCT02851758 [294] |
| Refractory polymyositis | Allogeneic mitochondria product; systemic administration. | Dose-escalation small cohorts described in registry snippet | Phase 1/2a; recruiting | Safety/tolerability and exploratory efficacy. | Clinical trial NCT04976140 |
| Refractory polymyositis (later phase) | Allogeneic mitochondria product; systemic administration. | Not specified | Phase 2; recruiting | Designed to evaluate efficacy/safety. | Clinical trial NCT07122648 |
| Cardiac ischemia | Autologous mitochondria with MSC-exosomes (co-therapy) | Not specified | Phase 1/2; unknown | Study designed to evaluate safety, feasibility, and preliminary efficacy; no results published or posted yet. | Clinical trial NCT05669144 |
| Pearson syndrome | Autologous CD34+ cells with donor-derived mitochondria. | 7 participants | Phase 1/2; Completed. | Trial completed; no peer-reviewed publication of outcomes. | Clinical trial NCT03384420 |
| Reproductive medicine | Autologous mitochondria transfer to oocyte. | Pilot | Pilot; unknown | Designed to assess feasibility of improving oocyte quality by autologous mitochondrial transfer; no published results available. | Clinical trial NCT03639506 |
Current progress in HMT methodology
With growing attentions and great efforts on the fundamental studies and therapeutic trails of mitochondria transfer, different applications regarding the improvement of transferring efficiency by engineered cell therapies, various transplantation strategies, and pharmacological agents are currently enhancing our understanding of mitochondria transfer and their potential as a treatment for different diseases.
Mitochondrial transplantation
Direct transplantation of isolated, functional mitochondria into damaged tissues has been explored in a range of animal models and early clinical studies, revealing its potential for restoring tissue function. Recent findings indicate that specific sub-populations of mitochondrial vesicles (mitoEVs), which are enriched in mitochondrial components, can effectively deliver these elements to recipient cells and modulate their function under various pathological conditions[295]. The concept of HMT has thus inspired the development of innovative mitochondrial transplantation strategies for human disease. Emerging technologies are rapidly expanding the toolkit for mitochondrial transplantation. Artificial mitochondria generated through mitochondrial genome engineering or mitochondrial-nuclear hybridization offer new avenues for treating diseases associated with mitochondrial dysfunction[296]. Notably, Gäbelein et al. developed a dedicated probe based on the FluidFM platform that can minimally invasively access living cells to extract, inject, and transplant mitochondria with subcellular spatial resolution; importantly, the transplanted mitochondria can integrate into the host cell's mitochondrial network and support replication of donor mitochondrial DNA. Recently, Hyslop, L. A., et al. demonstrated that mitochondrial donation through pronuclear transfer was compatible with human embryo viability, which was effective in reducing the transmission of homoplasmic and heteroplasmic pathogenic mtDNA variants[297]. In addition, Kim et al. developed a novel and efficient therapeutic platform consisting of fusogenic liposomes encapsulating mitochondria, which showed promising results in delivering mitochondrial proteins and was validated in the treatment of osteoarthritis[298]. These advanced approaches are opening new perspectives for the study and treatment of mitochondrial diseases, paving the way for innovative clinical applications.
Cell engineering using extracellular mitochondria
Engineered cell therapies using extracellular mitochondria represent a new frontier. Restoring the mitochondrial health of cells with mitochondrial dysfunction using embryonic stem cells, MSCs, iPSCls, stem cell secretions, and EV-mediated mitochondrial transplantation technology can affect important processes such as their proliferation, differentiation, cell metabolism, inflammatory response, cell senescence, cell stress, and cell migration, and enhance their regenerative capacity, survival rate, and therapeutic effect, which is crucial to improving therapeutic effects[299]. It holds great therapeutic potential for the treatment of neurological disorders, tissue repair, lung injury, tumors, and other diseases. Recent evidence showed that the release of haematopoietic cell-derived extracellular mitochondria into circulation, or direct transfer isolated healthy mitochondria from mice or human can extend lifespan, improve neurological function in Leigh syndrome (LS) mouse models[300]. Notably, accumulating evidence indicates that mitochondrial transfer from MSCs plays a dual role in promoting both tissue repair and cancer progression, highlighting its potential as a cell engineering strategy for regenerative medicine and cancer therapy[301, 302].
Drugs and nanomedicine affecting mitochondria transfer
Beyond their well-established role in energy metabolism, mitochondria are now appreciated as integrative hubs where catabolic and anabolic pathways, redox mediators, and diverse signaling networks intersect to sustain cellular homeostasis and orchestrate responses to environmental cues. Thus, understanding how pharmacological agents affect mitochondrial biochemistry and mitochondria transfer is therefore of critical importance and holds substantial promise for the development of innovative therapies targeting mitochondrial-related diseases[303]. Numerous studies have shown that drugs that can modulate mitochondrial transport are under development. Small molecules that promote TNT formation, actin polymerization, or mitochondrial transport can promote beneficial mitochondrial transport in regenerative environments, while TNT inhibitors or vesicle transport blockers may limit pathological metastasis in cancer[304]. Besides pharmaceutic drugs, new materials, especially nanoparticles, have been widely tested and engineered to enhance mitochondria transfer in vitro and in vivo. For instance, Huang T et al. reported that iron oxide nanoparticles (IONPs) can augment the intercellular mitochondrial transfer from hMSCs selectively to diseased cells through enhanced Cx43- mediated GJs[305].
Conclusions and perspectives
For the past two decades, HMT has emerged as a new paradigm in cell biology occurring in vitro and in vivo. More distinct transferring modes have been reported between cells in various tissue background. However, the remaining fundamental questions regarding HMT include: (1) What is the precise onset and triggering information for HMT? (2) What are the molecular players and orchestrated sequences of connection formation and transportation? (3) How to apply the therapeutic potential of HMT in clinic?
During the initiation of HMT, one crucial question is yet to be answered is the trigger of intercellular connections, or the trigger of Ev-Mito or Ex-Mito formation. Since HMT is commonly reported between healthy and stressed cells, one can hypothesize that the potential “signal” triggering the process of HMT arise from the receptor cells, which are in need of functional mitochondria[306]. Current published data showed that the triggering signal is presented by ADP in the stress osteocytes[306]. From the fundamental cell biology, this initiation information is intriguing and could be harnessed for alleviating various pathological states. During HMT formation and mitochondria transportation, directly visualization of mitochondria transfer would unreal its physiological significance. Current studies utilized fluorescence dyes to track mitochondria dynamics. However, the leakage of dyes results in limitations or even false results. The development of novel fluoresceins for mitochondrial tracking and visualizing HMT in real time in vivo remain major challenges in this field. Moreover, separating healthy and damaged mitochondria with different labeling during HMT could be another challenging which could greatly accelerate the progress.
In summary, considering the broad involvement of mitochondria transfer with implications for tissue homeostasis, physiology, and pathophysiology, future therapeutic implications need to enhance desirable transfer, bock non-wanted transfer and achieve functional treatment in receptor cells and tissues. In a broad point of view, segregation of mitochondria derived from various origins may bring new insights into “inter-organ mitohormesis”[127, 307]. These distant mitochondrial communications involved in multiple organs or even entire organism could explain the pathological mechanisms behind some systematic diseases, such as diabetes, stress, aging, lifespan and etc. From this perspective, mitochondria transplantation or mitochondria-related treatment may bring new therapeutics with high translational potential for treating diseases that are linked to mitochondrial dysfunctions.
Acknowledgements
The authors acknowledge the assistance of the Core Research Facilities and the Laboratory Animal Center of Medical Research Center of Shenzhen Nanshan People's Hospital.
Funding
This work was supported by the Shenzhen Medical Research Funds (B2504003, B2402033), the Noncommunicable Chronic Diseases-National Science and Technology Major Project (2025ZD0550400), the National Natural Science Foundation of China Grants (82261160395, 82230081, 82302748, 82430078), the Guangdong Provincial Science and Technology Innovation Council Grant (2017B030301018), the Guangdong Basic and Apllied Basic Research Foundation (2025B0303010), the Projects of Nanshan Health Committee (NSZD2025008, NS2025011, NS2025078), and the Nanshan District Medical Key Discipline Construction Financial Support Project.
Author contributions
LQ, DG, GX, and WY contribute to conceptualization and investigation; LQ, DG, ZC, ZX, MW and BG literature reviewing and writing review; LQ, DG, YL, XM, and WL editing; LQ, WY and GX visualization.
Declaration of generative AI in scientific writing
The authors did not use any AI or AI-assisted technologies in the writing process.
Competing interests
The authors have declared that no competing interest exists.
References
1. Spinelli JB, Haigis MC. The multifaceted contributions of mitochondria to cellular metabolism. Nature Cell Biology. 2018;20:745-54
2. Brady NR, Hamacher-Brady A, Westerhoff HV, Gottlieb RA. A wave of reactive oxygen species (ROS)-induced ROS release in a sea of excitable mitochondria. Antioxid Redox Signal. 2006;8:1651-65
3. Shadel Gerald S, Horvath Tamas L. Mitochondrial ROS Signaling in Organismal Homeostasis. Cell. 2015;163:560-9
4. Cartes-Saavedra B, Ghosh A, Hajnóczky G. The roles of mitochondria in global and local intracellular calcium signalling. Nature Reviews Molecular Cell Biology. 2025;26:456-75
5. Diaz-Meco MT, Linares JF, Moscat J. Hijacking the powerhouse: Mitochondrial transfer and mitophagy as emerging mechanisms of immune evasion. Molecular Cell. 2025;85:1258-9
6. Liu YJ, Sulc J, Auwerx J. Mitochondrial genetics, signalling and stress responses. Nature Cell Biology. 2025;27:393-407
7. Glover HL, Schreiner A, Dewson G, Tait SWG. Mitochondria and cell death. Nature Cell Biology. 2024;26:1434-46
8. Peng B, Zhang H. Mitochondrial homeostasis in stem cell senescence. Oral Science and Homeostatic Medicine. 2025;1:9610006
9. Westermann B. Mitochondrial fusion and fission in cell life and death. Nature Reviews Molecular Cell Biology. 2010;11:872-84
10. Tábara LC, Segawa M, Prudent J. Molecular mechanisms of mitochondrial dynamics. Nat Rev Mol Cell Biol. 2025;26:123-46
11. Chinnery PF, Hudson G. Mitochondrial genetics. Br Med Bull. 2013;106:135-59
12. Pfanner N, Warscheid B, Wiedemann N. Mitochondrial proteins: from biogenesis to functional networks. Nature Reviews Molecular Cell Biology. 2019;20:267-84
13. Hulbert AJ, Turner N, Hinde J, Else P, Guderley H. How might you compare mitochondria from different tissues and different species? J Comp Physiol B. 2006;176:93-105
14. Venkatraman K, Lipp NF, Budin I. Origin and evolution of mitochondrial inner membrane composition. J Cell Sci. 2025 138
15. Mishra P, Chan DC. Mitochondrial dynamics and inheritance during cell division, development and disease. Nat Rev Mol Cell Biol. 2014;15:634-46
16. Pagliarini DJ, Rutter J. Hallmarks of a new era in mitochondrial biochemistry. Genes Dev. 2013;27:2615-27
17. O'Rourke B. From bioblasts to mitochondria: ever expanding roles of mitochondria in cell physiology. Front Physiol. 2010;1:7
18. Mendes D, Peixoto F, Oliveira MM, Andrade PB, Videira RA. Mitochondria research and neurodegenerative diseases: On the track to understanding the biological world of high complexity. Mitochondrion. 2022;65:67-79
19. Møller IM, Rasmusson AG, Van Aken O. Plant mitochondria - past, present and future. Plant J. 2021;108:912-59
20. Xu X, Pang Y, Fan X. Mitochondria in oxidative stress, inflammation and aging: from mechanisms to therapeutic advances. Signal Transduction and Targeted Therapy. 2025;10:190
21. Moura JP, Oliveira PJ, Urbano AM. Mitochondria: An overview of their origin, genome, architecture, and dynamics. Biochim Biophys Acta Mol Basis Dis. 2025;1871:167803
22. Isenberg JS, Delpire E. Mitochondria: an elegant means of partitioning and harnessing energy. Am J Physiol Cell Physiol. 2021;320:C463-c4
23. Kizmaz B, Nutz A, Egeler A, Herrmann JM. Protein insertion into the inner membrane of mitochondria: routes and mechanisms. FEBS Open Bio. 2024;14:1627-39
24. Argueti-Ostrovsky S, Barel S, Kahn J, Israelson A. VDAC1: A Key Player in the Mitochondrial Landscape of Neurodegeneration. Biomolecules. 2024 15
25. Liao PC, Lin TY, Tsang CA, Huang CJ, Filpo K, Boldogh I. et al. Tom40 functions as a channel for protein retrotranslocation in the mitochondria-associated degradation (MAD) pathway. Commun Biol. 2025;8:1122
26. Palmer CS, Lou J, Kouskousis B, Pandzic E, Anderson AJ, Kang Y. et al. Super-resolution microscopy reveals the arrangement of inner membrane protein complexes in mammalian mitochondria. J Cell Sci. 2021 134
27. Daumke O, van der Laan M. Molecular machineries shaping the mitochondrial inner membrane. Nat Rev Mol Cell Biol. 2025;26:706-24
28. Vercellino I, Sazanov LA. The assembly, regulation and function of the mitochondrial respiratory chain. Nat Rev Mol Cell Biol. 2022;23:141-61
29. Edwards R, Eaglesfield R, Tokatlidis K. The mitochondrial intermembrane space: the most constricted mitochondrial sub-compartment with the largest variety of protein import pathways. Open Biol. 2021;11:210002
30. Szczepanowska K, Trifunovic A. Mitochondrial matrix proteases: quality control and beyond. Febs j. 2022;289:7128-46
31. Weiß E, Kretschmer D. Formyl-Peptide Receptors in Infection, Inflammation, and Cancer. Trends in Immunology. 2018;39:815-29
32. Boguszewska K, Szewczuk M, Kaźmierczak-Barańska J, Karwowski BT. The Similarities between Human Mitochondria and Bacteria in the Context of Structure, Genome, and Base Excision Repair System. Molecules. 2020 25
33. Friedman JR, Nunnari J. Mitochondrial form and function. Nature. 2014;505:335-43
34. Focusing on mitochondrial form and function. Nature Cell Biology. 2018; 20: 735-.
35. Sagan L. On the origin of mitosing cells. J Theor Biol. 1967;14:255-74
36. Gray MW. Mitochondrial evolution. Cold Spring Harb Perspect Biol. 2012;4:a011403
37. Raval PK, Martin WF, Gould SB. Mitochondrial evolution: Gene shuffling, endosymbiosis, and signaling. Sci Adv. 2023;9:eadj4493
38. Leger MM, Gawryluk RMR. Evolution: A gene-rich mitochondrial genome sheds light on the last eukaryotic common ancestor. Current Biology. 2024;34:R776-R9
39. Gray MW, Burger G, Lang BF. Mitochondrial evolution. Science. 1999;283:1476-81
40. Brocks JJ, Logan GA, Buick R, Summons RE. Archean molecular fossils and the early rise of eukaryotes. Science. 1999;285:1033-6
41. Andersson SG, Karlberg O, Canbäck B, Kurland CG. On the origin of mitochondria: a genomics perspective. Philos Trans R Soc Lond B Biol Sci. 2003;358:165-77 discussion 77-9
42. Gabaldón T, Huynen MA. Shaping the mitochondrial proteome. Biochim Biophys Acta. 2004;1659:212-20
43. Kukat C, Davies KM, Wurm CA, Spåhr H, Bonekamp NA, Kühl I. et al. Cross-strand binding of TFAM to a single mtDNA molecule forms the mitochondrial nucleoid. Proc Natl Acad Sci U S A. 2015;112:11288-93
44. Anderson S, Bankier AT, Barrell BG, de Bruijn MH, Coulson AR, Drouin J. et al. Sequence and organization of the human mitochondrial genome. Nature. 1981;290:457-65
45. Gu J, Wu M, Guo R, Yan K, Lei J, Gao N. et al. The architecture of the mammalian respirasome. Nature. 2016;537:639-43
46. Dong LF, Rohlena J, Zobalova R, Nahacka Z, Rodriguez AM, Berridge MV. et al. Mitochondria on the move: Horizontal mitochondrial transfer in disease and health. J Cell Biol. 2023 222
47. Aryaman J, Johnston IG, Jones NS. Mitochondrial Heterogeneity. Front Genet. 2018;9:718
48. Monzel AS, Enríquez JA, Picard M. Multifaceted mitochondria: moving mitochondrial science beyond function and dysfunction. Nature Metabolism. 2023;5:546-62
49. Granath-Panelo M, Kajimura S. Mitochondrial heterogeneity and adaptations to cellular needs. Nat Cell Biol. 2024;26:674-86
50. Birky CW Jr. The inheritance of genes in mitochondria and chloroplasts: laws, mechanisms, and models. Annu Rev Genet. 2001;35:125-48
51. Rustom A, Saffrich R, Markovic I, Walther P, Gerdes HH. Nanotubular highways for intercellular organelle transport. Science. 2004;303:1007-10
52. Gerdes HH, Carvalho RN. Intercellular transfer mediated by tunneling nanotubes. Curr Opin Cell Biol. 2008;20:470-5
53. Abounit S, Zurzolo C. Wiring through tunneling nanotubes-from electrical signals to organelle transfer. J Cell Sci. 2012;125:1089-98
54. Tan AS, Baty JW, Dong LF, Bezawork-Geleta A, Endaya B, Goodwin J. et al. Mitochondrial genome acquisition restores respiratory function and tumorigenic potential of cancer cells without mitochondrial DNA. Cell Metab. 2015;21:81-94
55. Sheng ZH, Cai Q. Mitochondrial transport in neurons: impact on synaptic homeostasis and neurodegeneration. Nat Rev Neurosci. 2012;13:77-93
56. Cox RT, Spradling AC. Milton controls the early acquisition of mitochondria by Drosophila oocytes. Development. 2006;133:3371-7
57. Morris RL, Hollenbeck PJ. Axonal transport of mitochondria along microtubules and F-actin in living vertebrate neurons. J Cell Biol. 1995;131:1315-26
58. Quintero OA, DiVito MM, Adikes RC, Kortan MB, Case LB, Lier AJ. et al. Human Myo19 is a novel myosin that associates with mitochondria. Curr Biol. 2009;19:2008-13
59. Schwarz TL. Mitochondrial trafficking in neurons. Cold Spring Harb Perspect Biol. 2013 5
60. Adebayo M, Singh S, Singh AP, Dasgupta S. Mitochondrial fusion and fission: The fine-tune balance for cellular homeostasis. Faseb j. 2021;35:e21620
61. Xu W, Xu X, Zhang Y, Li F, Xia D. Mitochondrial fission and fusion in inflammatory diseases: mechanisms and therapeutic implications. J Transl Med. 2025
62. Preminger N, Schuldiner M. Beyond fission and fusion-Diving into the mysteries of mitochondrial shape. PLoS Biol. 2024;22:e3002671
63. Liu BH, Xu CZ, Liu Y, Lu ZL, Fu TL, Li GR. et al. Mitochondrial quality control in human health and disease. Mil Med Res. 2024;11:32
64. Landoni JC, Kleele T, Winter J, Stepp W, Manley S. Mitochondrial Structure, Dynamics, and Physiology: Light Microscopy to Disentangle the Network. Annu Rev Cell Dev Biol. 2024;40:219-40
65. Pangou E, Sumara I. The Multifaceted Regulation of Mitochondrial Dynamics During Mitosis. Front Cell Dev Biol. 2021;9:767221
66. Taguchi N, Ishihara N, Jofuku A, Oka T, Mihara K. Mitotic phosphorylation of dynamin-related GTPase Drp1 participates in mitochondrial fission. J Biol Chem. 2007;282:11521-9
67. Kashatus DF, Lim KH, Brady DC, Pershing NL, Cox AD, Counter CM. RALA and RALBP1 regulate mitochondrial fission at mitosis. Nat Cell Biol. 2011;13:1108-15
68. Kamerkar SC, Liu A, Higgs HN. Mitochondrial fission - changing perspectives for future progress. J Cell Sci. 2025 138
69. Kanfer G, Courthéoux T, Peterka M, Meier S, Soste M, Melnik A. et al. Mitotic redistribution of the mitochondrial network by Miro and Cenp-F. Nat Commun. 2015;6:8015
70. Rohn JL, Patel JV, Neumann B, Bulkescher J, McHedlishvili N, McMullan RC. et al. Myo19 ensures symmetric partitioning of mitochondria and coupling of mitochondrial segregation to cell division. Curr Biol. 2014;24:2598-605
71. Yu F, Courjaret R, Assaf L, Elmi A, Hammad A, Fisher M. et al. Mitochondria-ER contact sites expand during mitosis. iScience. 2024;27:109379
72. Iwata R, Casimir P, Vanderhaeghen P. Mitochondrial dynamics in postmitotic cells regulate neurogenesis. Science. 2020;369:858-62
73. Onfelt B, Nedvetzki S, Yanagi K, Davis DM. Cutting edge: Membrane nanotubes connect immune cells. J Immunol. 2004;173:1511-3
74. Plotnikov EY, Khryapenkova TG, Vasileva AK, Marey MV, Galkina SI, Isaev NK. et al. Cell-to-cell cross-talk between mesenchymal stem cells and cardiomyocytes in co-culture. J Cell Mol Med. 2008;12:1622-31
75. Vignais ML, Caicedo A, Brondello JM, Jorgensen C. Cell Connections by Tunneling Nanotubes: Effects of Mitochondrial Trafficking on Target Cell Metabolism, Homeostasis, and Response to Therapy. Stem Cells Int. 2017;2017:6917941
76. Austefjord MW, Gerdes HH, Wang X. Tunneling nanotubes: Diversity in morphology and structure. Commun Integr Biol. 2014;7:e27934
77. Liu D, Gao Y, Liu J, Huang Y, Yin J, Feng Y. et al. Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduct Target Ther. 2021;6:65
78. Wang Y, Cui J, Sun X, Zhang Y. Tunneling-nanotube development in astrocytes depends on p53 activation. Cell Death Differ. 2011;18:732-42
79. Qin Y, Jiang X, Yang Q, Zhao J, Zhou Q, Zhou Y. The Functions, Methods, and Mobility of Mitochondrial Transfer Between Cells. Front Oncol. 2021;11:672781
80. Hase K, Kimura S, Takatsu H, Ohmae M, Kawano S, Kitamura H. et al. M-Sec promotes membrane nanotube formation by interacting with Ral and the exocyst complex. Nat Cell Biol. 2009;11:1427-32
81. Kee Y, Yoo JS, Hazuka CD, Peterson KE, Hsu SC, Scheller RH. Subunit structure of the mammalian exocyst complex. Proc Natl Acad Sci U S A. 1997;94:14438-43
82. Schiller C, Diakopoulos KN, Rohwedder I, Kremmer E, von Toerne C, Ueffing M. et al. LST1 promotes the assembly of a molecular machinery responsible for tunneling nanotube formation. J Cell Sci. 2013;126:767-77
83. Wang X, Bukoreshtliev NV, Gerdes HH. Developing neurons form transient nanotubes facilitating electrical coupling and calcium signaling with distant astrocytes. PLoS One. 2012;7:e47429
84. Onfelt B, Nedvetzki S, Benninger RK, Purbhoo MA, Sowinski S, Hume AN. et al. Structurally distinct membrane nanotubes between human macrophages support long-distance vesicular traffic or surfing of bacteria. J Immunol. 2006;177:8476-83
85. Gurke S, Barroso JF, Hodneland E, Bukoreshtliev NV, Schlicker O, Gerdes HH. Tunneling nanotube (TNT)-like structures facilitate a constitutive, actomyosin-dependent exchange of endocytic organelles between normal rat kidney cells. Exp Cell Res. 2008;314:3669-83
86. Bukoreshtliev NV, Wang X, Hodneland E, Gurke S, Barroso JF, Gerdes HH. Selective block of tunneling nanotube (TNT) formation inhibits intercellular organelle transfer between PC12 cells. FEBS Lett. 2009;583:1481-8
87. Attia A, Majstrowicz K, Shembekar S, Honnert U, Nikolaus P, Lohmann B. et al. Myosin-19 and Miro Regulate Mitochondria-Endoplasmic Reticulum Contacts and Mitochondria Inner Membrane Architecture. Cells. 2025 p. 1657
88. Glater EE, Megeath LJ, Stowers RS, Schwarz TL. Axonal transport of mitochondria requires milton to recruit kinesin heavy chain and is light chain independent. J Cell Biol. 2006;173:545-57
89. Misko A, Jiang S, Wegorzewska I, Milbrandt J, Baloh RH. Mitofusin 2 is necessary for transport of axonal mitochondria and interacts with the Miro/Milton complex. J Neurosci. 2010;30:4232-40
90. Ahmad T, Mukherjee S, Pattnaik B, Kumar M, Singh S, Kumar M. et al. Miro1 regulates intercellular mitochondrial transport & enhances mesenchymal stem cell rescue efficacy. Embo j. 2014;33:994-1010
91. López-Doménech G, Covill-Cooke C, Ivankovic D, Halff EF, Sheehan DF, Norkett R. et al. Miro proteins coordinate microtubule- and actin-dependent mitochondrial transport and distribution. Embo j. 2018;37:321-36
92. Sun X, Wang Y, Zhang J, Tu J, Wang XJ, Su XD. et al. Tunneling-nanotube direction determination in neurons and astrocytes. Cell Death Dis. 2012;3:e438
93. Xi XR, Zhang ZQ, Li YL, Liu Z, Ma DY, Gao Z. et al. Hypothermia promotes tunneling nanotube formation and the transfer of astrocytic mitochondria into oxygen-glucose deprivation/reoxygenation-injured neurons. Brain Res. 2024;1831:148826
94. Chakraborty R, Nonaka T, Hasegawa M, Zurzolo C. Tunnelling nanotubes between neuronal and microglial cells allow bi-directional transfer of α-Synuclein and mitochondria. Cell Death & Disease. 2023 14
95. Borcherding N, Brestoff JR. The power and potential of mitochondria transfer. Nature. 2023;623:283-91
96. Meşe G, Richard G, White TW. Gap junctions: basic structure and function. J Invest Dermatol. 2007;127:2516-24
97. Zhu Y. Gap Junction-Dependent and -Independent Functions of Connexin43 in Biology. Biology (Basel). 2022 11
98. Goldberg GS, Valiunas V, Brink PR. Selective permeability of gap junction channels. Biochim Biophys Acta. 2004;1662:96-101
99. Gervasi A, D'Aprile S, Denaro S, Amorini MA, Vicario N, Parenti R. Connexin 43 Role in Mitochondrial Transfer and Homeostasis in the Central Nervous System. J Cell Physiol. 2025;240:e70086
100. Golan K, Singh AK, Kollet O, Bertagna M, Althoff MJ, Khatib-Massalha E. et al. Bone marrow regeneration requires mitochondrial transfer from donor Cx43-expressing hematopoietic progenitors to stroma. Blood. 2020;136:2607-19
101. Fahey M, Bennett M, Thomas M, Montney K, Vivancos-Koopman I, Pugliese B. et al. Mesenchymal stromal cells donate mitochondria to articular chondrocytes exposed to mitochondrial, environmental, and mechanical stress. Scientific Reports. 2022;12:21525
102. Irwin RM, Thomas MA, Fahey MJ, Mayán MD, Smyth JW, Delco ML. Connexin 43 regulates intercellular mitochondrial transfer from human mesenchymal stromal cells to chondrocytes. Stem Cell Research & Therapy. 2024;15:359
103. Li H, Wang C, He T, Zhao T, Chen Y-y, Shen Y-l. et al. Mitochondrial Transfer from Bone Marrow Mesenchymal Stem Cells to Motor Neurons in Spinal Cord Injury Rats via Gap Junction. Theranostics. 2019;9:2017-35
104. Yang J, Liu L, Oda Y, Wada K, Ago M, Matsuda S. et al. Extracellular Vesicles and Cx43-Gap Junction Channels Are the Main Routes for Mitochondrial Transfer from Ultra-Purified Mesenchymal Stem Cells, RECs. Int J Mol Sci. 2023 24
105. Weber PA, Chang HC, Spaeth KE, Nitsche JM, Nicholson BJ. The permeability of gap junction channels to probes of different size is dependent on connexin composition and permeant-pore affinities. Biophys J. 2004;87:958-73
106. Shami GJ, Cheng D, Verhaegh P, Koek G, Wisse E, Braet F. Three-dimensional ultrastructure of giant mitochondria in human non-alcoholic fatty liver disease. Sci Rep. 2021;11:3319
107. Islam MN, Das SR, Emin MT, Wei M, Sun L, Westphalen K. et al. Mitochondrial transfer from bone-marrow-derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med. 2012;18:759-65
108. Yao Y, Fan XL, Jiang D, Zhang Y, Li X, Xu ZB. et al. Connexin 43-Mediated Mitochondrial Transfer of iPSC-MSCs Alleviates Asthma Inflammation. Stem Cell Reports. 2018;11:1120-35
109. Boengler K, Leybaert L, Ruiz-Meana M, Schulz R. Connexin 43 in Mitochondria: What Do We Really Know About Its Function? Front Physiol. 2022;13:928934
110. Miro-Casas E, Ruiz-Meana M, Agullo E, Stahlhofen S, Rodríguez-Sinovas A, Cabestrero A. et al. Connexin43 in cardiomyocyte mitochondria contributes to mitochondrial potassium uptake. Cardiovasc Res. 2009;83:747-56
111. Wang JD, Shao Y, Liu D, Liu NY, Zhu DY. Rictor/mTORC2 involves mitochondrial function in ES cells derived cardiomyocytes via mitochondrial Connexin 43. Acta Pharmacol Sin. 2021;42:1790-7
112. Qin L, Liu W, Cao H, Xiao G. Molecular mechanosensors in osteocytes. Bone Res. 2020;8:23
113. Qin L, Fu X, Ma J, Lin M, Zhang P, Wang Y. et al. Kindlin-2 mediates mechanotransduction in bone by regulating expression of Sclerostin in osteocytes. Commun Biol. 2021;4:402
114. Qin L, He T, Yang D, Wang Y, Li Z, Yan Q. et al. Osteocyte β1 integrin loss causes low bone mass and impairs bone mechanotransduction in mice. J Orthop Translat. 2022;34:60-72
115. Qin L, Chen Z, Yang D, He T, Xu Z, Zhang P. et al. Osteocyte β3 integrin promotes bone mass accrual and force-induced bone formation in mice. J Orthop Translat. 2023;40:58-71
116. Gao J, Qin A, Liu D, Ruan R, Wang Q, Yuan J. et al. Endoplasmic reticulum mediates mitochondrial transfer within the osteocyte dendritic network. Sci Adv. 2019;5:eaaw7215
117. Friedman JR, Webster BM, Mastronarde DN, Verhey KJ, Voeltz GK. ER sliding dynamics and ER-mitochondrial contacts occur on acetylated microtubules. J Cell Biol. 2010;190:363-75
118. Liao P, Chen L, Zhou H, Mei J, Chen Z, Wang B. et al. Osteocyte mitochondria regulate angiogenesis of transcortical vessels. Nature Communications. 2024;15:2529
119. Hayakawa K, Esposito E, Wang X, Terasaki Y, Liu Y, Xing C. et al. Transfer of mitochondria from astrocytes to neurons after stroke. Nature. 2016;535:551-5
120. Brestoff JR SK, Aquilano K, Becker LB, Berridge MV, Boilard E, Caicedo A, Crewe C, Enríquez JA, Gao J, Gustafsson ÅB, Hayakawa K, Khoury M, Lee YS, Lettieri-Barbato D, Luz-Crawford P, McBride HM, McCully JD, Nakai R, Neuzil J, Picard M, Rabchevsky AG, Rodriguez AM, Sengupta S, Sercel AJ, Suda T, Teitell MA, Thierry AR, Tian R, Walker M, Zheng M. Recommendations for mitochondria transfer and transplantation nomenclature and characterization. Nat Metab. 2025;7:53-67
121. Al Amir Dache Z, Otandault A, Tanos R, Pastor B, Meddeb R, Sanchez C. et al. Blood contains circulating cell-free respiratory competent mitochondria. Faseb j. 2020;34:3616-30
122. Caicedo A, Zambrano K, Sanon S, Luis Vélez J, Montalvo M, Jara F. et al. The diversity and coexistence of extracellular mitochondria in circulation: A friend or foe of the immune system. Mitochondrion. 2021;58:270-84
123. Li X, Jiang O, Chen M, Wang S. Mitochondrial homeostasis: shaping health and disease. Oral Science and Homeostatic Medicine. 2024 3
124. Mittelbrunn M, Sánchez-Madrid F. Intercellular communication: diverse structures for exchange of genetic information. Nat Rev Mol Cell Biol. 2012;13:328-35
125. Guescini M, Genedani S, Stocchi V, Agnati LF. Astrocytes and Glioblastoma cells release exosomes carrying mtDNA. J Neural Transm (Vienna). 2010;117:1-4
126. Phinney DG, Di Giuseppe M, Njah J, Sala E, Shiva S, St Croix CM. et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nature Communications. 2015;6:8472
127. Crewe C, Funcke JB, Li S, Joffin N, Gliniak CM, Ghaben AL. et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 2021;33:1853-68.e11
128. Suh J, Kim NK, Shim W, Lee SH, Kim HJ, Moon E. et al. Mitochondrial fragmentation and donut formation enhance mitochondrial secretion to promote osteogenesis. Cell Metab. 2023;35:345-60.e7
129. Liang W, Sagar S, Ravindran R, Najor RH, Quiles JM, Chi L. et al. Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nature Communications. 2023;14:5031
130. Liao Z, Tong B, Ke W, Yang C, Wu X, Lei M. Extracellular vesicles as carriers for mitochondria: Biological functions and clinical applications. Mitochondrion. 2024;78:101935
131. Marinković M, Novak I. A brief overview of BNIP3L/NIX receptor-mediated mitophagy. FEBS Open Bio. 2021;11:3230-6
132. Phinney DG, Di Giuseppe M, Njah J, Sala E, Shiva S, St Croix CM. et al. Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat Commun. 2015;6:8472
133. Narendra DP, Youle RJ. The role of PINK1-Parkin in mitochondrial quality control. Nature Cell Biology. 2024;26:1639-51
134. Todkar K, Chikhi L, Desjardins V, El-Mortada F, Pépin G, Germain M. Selective packaging of mitochondrial proteins into extracellular vesicles prevents the release of mitochondrial DAMPs. Nat Commun. 2021;12:1971
135. Rosina M, Ceci V, Turchi R, Li C, Borcherding N, Sciarretta F. et al. Ejection of damaged mitochondria and their removal by macrophages ensure efficient thermogenesis in brown adipose tissue. Cell Metabolism. 2022;34:533-48.e12
136. Choong CJ, Okuno T, Ikenaka K, Baba K, Hayakawa H, Koike M. et al. Alternative mitochondrial quality control mediated by extracellular release. Autophagy. 2021;17:2962-74
137. Borcherding N, Jia W, Giwa R, Field RL, Moley JR, Kopecky BJ. et al. Dietary lipids inhibit mitochondria transfer to macrophages to divert adipocyte-derived mitochondria into the blood. Cell Metab. 2022;34:1499-513.e8
138. Boudreau LH, Duchez AC, Cloutier N, Soulet D, Martin N, Bollinger J. et al. Platelets release mitochondria serving as substrate for bactericidal group IIA-secreted phospholipase A2 to promote inflammation. Blood. 2014;124:2173-83
139. Nakajima A, Kurihara H, Yagita H, Okumura K, Nakano H. Mitochondrial Extrusion through the cytoplasmic vacuoles during cell death. J Biol Chem. 2008;283:24128-35
140. Lyamzaev KG, Nepryakhina OK, Saprunova VB, Bakeeva LE, Pletjushkina OY, Chernyak BV. et al. Novel mechanism of elimination of malfunctioning mitochondria (mitoptosis): formation of mitoptotic bodies and extrusion of mitochondrial material from the cell. Biochim Biophys Acta. 2008;1777:817-25
141. Torralba D BF, Sánchez-Madrid F. Mitochondria Know No Boundaries: Mechanisms and Functions of Intercellular Mitochondrial Transfer. Front Cell Dev Biol. 2016;4:107
142. López-Armada MJ, Riveiro-Naveira RR, Vaamonde-García C, Valcárcel-Ares MN. Mitochondrial dysfunction and the inflammatory response. Mitochondrion. 2013;13:106-18
143. Weinberg Samuel E, Sena Laura A, Chandel Navdeep S. Mitochondria in the Regulation of Innate and Adaptive Immunity. Immunity. 2015;42:406-17
144. Morán M, Moreno-Lastres D, Marín-Buera L, Arenas J, Martín MA, Ugalde C. Mitochondrial respiratory chain dysfunction: Implications in neurodegeneration. Free Radical Biology and Medicine. 2012;53:595-609
145. Kim S, Kim Y, Yu SH, Lee SE, Park JH, Cho G, Choi C, Han K, Kim CH, Kang YC. Platelet-derived mitochondria transfer facilitates wound-closure by modulating ROS levels in dermal fibroblasts. Platelets. 2022;34:2151996
146. Wang Y, Li W, Guo Y, Huang Y, Guo Y, Song J. et al. Mitochondria Transplantation to Bone Marrow Stromal Cells Promotes Angiogenesis During Bone Repair. Advanced Science. 2024 11
147. Xia L, Zhang C, Lv N, Liang Z, Ma T, Cheng H. et al. AdMSC-derived exosomes alleviate acute lung injury via transferring mitochondrial component to improve homeostasis of alveolar macrophages. Theranostics. 2022;12:2928-47
148. Morrison TJ, Jackson MV, Cunningham EK, Kissenpfennig A, McAuley DF, O'Kane CM. et al. Mesenchymal Stromal Cells Modulate Macrophages in Clinically Relevant Lung Injury Models by Extracellular Vesicle Mitochondrial Transfer. American Journal of Respiratory and Critical Care Medicine. 2017;196:1275-86
149. Wu G, Zhu Q, Zeng J, Gu X, Miao Y, Xu W. et al. Extracellular mitochondrial DNA promote NLRP3 inflammasome activation and induce acute lung injury through TLR9 and NF-κB. Journal of Thoracic Disease. 2019;11:4816-28
150. Court AC, Le-Gatt A, Luz-Crawford P, Parra E, Aliaga-Tobar V, Bátiz LF. et al. Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO reports. 2020 21
151. Cai W, Zhang J, Yu Y, Ni Y, Wei Y, Cheng Y. et al. Mitochondrial Transfer Regulates Cell Fate Through Metabolic Remodeling in Osteoporosis. Advanced Science. 2022 10
152. van der Vlist M, Raoof R, Willemen HLDM, Prado J, Versteeg S, Martin Gil C. et al. Macrophages transfer mitochondria to sensory neurons to resolve inflammatory pain. Neuron. 2022;110:613-26.e9
153. Saha T, Dash C, Jayabalan R, Khiste S, Kulkarni A, Kurmi K. et al. Intercellular nanotubes mediate mitochondrial trafficking between cancer and immune cells. Nat Nanotechnol. 2022;17:98-106
154. Hayakawa K, Bruzzese M, Chou SHY, Ning M, Ji X, Lo EH. Extracellular Mitochondria for Therapy and Diagnosis in Acute Central Nervous System Injury. JAMA Neurology. 2018 75
155. Stadelmann C, Timmler S, Barrantes-Freer A, Simons M. Myelin in the Central Nervous System: Structure, Function, and Pathology. Physiological Reviews. 2019;99:1381-431
156. Manoli I, Alesci S, Blackman MR, Su YA, Rennert OM, Chrousos GP. Mitochondria as key components of the stress response. Trends in Endocrinology & Metabolism. 2007;18:190-8
157. Nakamura Y, Park J-H, Hayakawa K. Therapeutic use of extracellular mitochondria in CNS injury and disease. Experimental Neurology. 2020 324
158. Dewanjee S, Chakraborty P, Bhattacharya H, Chacko L, Singh B, Chaudhary A. et al. Altered glucose metabolism in Alzheimer's disease: Role of mitochondrial dysfunction and oxidative stress. Free Radical Biology and Medicine. 2022;193:134-57
159. Kondo MA, Norris AL, Yang K, Cheshire M, Newkirk I, Chen X. et al. Dysfunction of mitochondria and GABAergic interneurons in the anterior cingulate cortex of individuals with schizophrenia. Neuroscience Research. 2022;185:67-72
160. Fatima G, Sharma VP, Das SK, Mahdi AA. Oxidative stress and antioxidative parameters in patients with spinal cord injury: implications in the pathogenesis of disease. Spinal Cord. 2015;53:3-6
161. Choong C-J, Mochizuki H. Involvement of Mitochondria in Parkinson's Disease. International Journal of Molecular Sciences. 2023 24
162. Reiss AB, Gulkarov S, Jacob B, Srivastava A, Pinkhasov A, Gomolin IH. et al. Mitochondria in Alzheimer's Disease Pathogenesis. Life. 2024 14
163. Liu Q, Zhang X, Zhu T, Xu Z, Dong Y, Chen B. Mitochondrial transfer from mesenchymal stem cells: Mechanisms and functions. Mitochondrion. 2024 79
164. Berridge Michael V, Schneider Remy T, McConnell Melanie J. Mitochondrial Transfer from Astrocytes to Neurons following Ischemic Insult: Guilt by Association? Cell Metabolism. 2016;24:376-8
165. Hernández IH, Villa-González M, Martín G, Soto M, Pérez-Álvarez MJ. Glial Cells as Therapeutic Approaches in Brain Ischemia-Reperfusion Injury. Cells. 2021 10
166. Machihara K, Oki S, Maejima Y, Kageyama S, Onda A, Koseki Y. et al. Restoration of mitochondrial function by Spirulina polysaccharide via upregulated SOD2 in aging fibroblasts. iScience. 2023 26
167. Al Amir Dache Z, Thierry AR. Mitochondria-derived cell-to-cell communication. Cell Reports. 2023 42
168. Li H, Sun W, Gong W, Han Y. Transfer and fates of damaged mitochondria: role in health and disease. The FEBS Journal. 2024;291:5342-64
169. Zhu Z, Li X, Wang X, Zuo X, Ma Y, Gao X. et al. Photobiomodulation augments the effects of mitochondrial transplantation in the treatment of spinal cord injury in rats by facilitating mitochondrial transfer to neurons via Connexin 36. Bioengineering & Translational Medicine. 2022 8
170. Bustamante-Barrientos FA, Luque-Campos N, Araya MJ, Lara-Barba E, de Solminihac J, Pradenas C. et al. Mitochondrial dysfunction in neurodegenerative disorders: Potential therapeutic application of mitochondrial transfer to central nervous system-residing cells. Journal of Translational Medicine. 2023 21
171. Robicsek O, Ene HM, Karry R, Ytzhaki O, Asor E, McPhie D. et al. Isolated Mitochondria Transfer Improves Neuronal Differentiation of Schizophrenia-Derived Induced Pluripotent Stem Cells and Rescues Deficits in a Rat Model of the Disorder. Schizophrenia Bulletin. 2018;44:432-42
172. Zhou J, Zhang L, Peng J, Zhang X, Zhang F, Wu Y. et al. Astrocytic LRP1 enables mitochondria transfer to neurons and mitigates brain ischemic stroke by suppressing ARF1 lactylation. Cell Metab. 2024;36:2054-68.e14
173. Ni XC, Wang HF, Cai YY, Yang D, Alolga RN, Liu B. et al. Ginsenoside Rb1 inhibits astrocyte activation and promotes transfer of astrocytic mitochondria to neurons against ischemic stroke. Redox Biol. 2022;54:102363
174. Anastacio MM, Kanter EM, Makepeace CM, Keith AD, Zhang H, Schuessler RB. et al. Relationship between mitochondrial matrix volume and cellular volume in response to stress and the role of ATP-sensitive potassium channel. Circulation. 2013;128:S130-5
175. Karwi QG JA, Lopaschuk GD. Allosteric, transcriptional and post-translational control of mitochondrial energy metabolism. Biochem J. 2019;476:1695-712
176. Bisaccia G RF, Gallina S, Di Baldassarre A, Ghinassi B. Mitochondrial Dysfunction and Heart Disease: Critical Appraisal of an Overlooked Association. Int J Mol Sci. 2021 22
177. Shanmughapriya S LD, Natarajaseenivasan K. Inter and Intracellular mitochondrial trafficking in health and disease. Ageing Res Rev. 2020;62:101128
178. Sun M JW, Mu N, Zhang Z, Yu L, Ma H. Mitochondrial transplantation as a novel therapeutic strategy for cardiovascular diseases. J Transl Med. 2023;21:347
179. Zhang C, Hao H, Wang Y, Mu N, Jiang W, Zhang Z. et al. Intercellular mitochondrial component transfer triggers ischemic cardiac fibrosis. Sci Bull (Beijing). 2023;68:1784-99
180. Zhao Y, Hu Y, Wang Y, Qian H, Zhu C, Dong H. et al. Cardiac fibroblast-derived mitochondria-enriched sEVs regulate tissue inflammation and ventricular remodeling post-myocardial infarction through NLRP3 pathway. Pharmacol Res. 2025;214:107676
181. Zhang A LY, Pan J, Pontanari F, Chia-Hao Chang A, Wang H, Gao S, Wang C, Chang AC. Delivery of mitochondria confers cardioprotection through mitochondria replenishment and metabolic compliance. Mol Ther. 2023;31:1468-79
182. Jin N ZM, Zhou L, Jin S, Cheng H, Li X, Shi Y, Xiang T, Zhang Z, Liu Z, Zhao H, Xie J. Mitochondria transplantation alleviates cardiomyocytes apoptosis through inhibiting AMPKα-mTOR mediated excessive autophagy. Faseb j. 2024;38:e23655
183. Sun X GR, Li W, Zhao Y, Yang H, Chen H, Jiang H, Dong Z, Hu J, Liu J, Zou Y, Sun A, Ge J. Alda-1 treatment promotes the therapeutic effect of mitochondrial transplantation for myocardial ischemia-reperfusion injury. Bioact Mater. 2021;6:2058-69
184. Lin RZ IG, Luo AC, Zhu Y, Hong X, Neumeyer J, Tang HW, Perrimon N, Melero-Martin JM. Mitochondrial transfer mediates endothelial cell engraftment through mitophagy. Nature. 2024;629:660-8
185. Paliwal S CR, Agrawal A, Mohanty S. Regenerative abilities of mesenchymal stem cells through mitochondrial transfer. J Biomed Sci. 2018;25:31
186. Sinclair KA YS, Hopkins PM, Chambers DC. Characterization of intercellular communication and mitochondrial donation by mesenchymal stromal cells derived from the human lung. Stem Cell Res Ther. 2016;7:91
187. Hough KP TJ, Strenkowski JG, Wang Y, Chacko BK, Tousif S, Chanda D, Steele C, Antony VB, Dokland T, Ouyang X, Zhang J, Duncan SR, Thannickal VJ, Darley-Usmar VM, Deshane JS. Exosomal transfer of mitochondria from airway myeloid-derived regulatory cells to T cells. Redox Biol. 2018;18:54-64
188. Riou M AA, Charles AL, Andrès E, Pistea C, Charloux A, Geny B. New Insights into the Implication of Mitochondrial Dysfunction in Tissue, Peripheral Blood Mononuclear Cells, and Platelets during Lung Diseases. J Clin Med. 2020 9
189. Frankenberg Garcia J RA, Mak JCW, Halayko AJ, Hui CKM, Xu B, Chung KF, Rodriguez T, Michaeloudes C, Bhavsar PK. Mitochondrial Transfer Regulates Bioenergetics in Healthy and Chronic Obstructive Pulmonary Disease Airway Smooth Muscle. Am J Respir Cell Mol Biol. 2022;67:471-81
190. Malsin ES KD. The mitochondria in lung fibrosis: friend or foe? Transl Res. 2018;202:1-23
191. Huang T LR, Su Y, Sun H, Zheng X, Zhang J, Lu X, Zhao B, Jiang X, Huang L, Li N, Shi J, Fan X, Xu D, Zhang T, Gao J. Efficient intervention for pulmonary fibrosis via mitochondrial transfer promoted by mitochondrial biogenesis. Nat Commun. 2023;14:5781
192. Zhang F ZX, Zhao F, Li L, Ren Y, Li L, Huang H, Yin H. TFAM-Mediated mitochondrial transfer of MSCs improved the permeability barrier in sepsis-associated acute lung injury. Apoptosis. 2023;28:1048-59
193. Li J, Liu W, Zhang J, Sun C. The Role of Mitochondrial Quality Control in Liver Diseases: Dawn of a Therapeutic Era. International Journal of Biological Sciences. 2025;21:1767-83
194. Mansouri A, Gattolliat C-H, Asselah T. Mitochondrial Dysfunction and Signaling in Chronic Liver Diseases. Gastroenterology. 2018;155:629-47
195. Yang YM. AMPK-associated signaling to bridge the gap between fuel metabolism and hepatocyte viability. World Journal of Gastroenterology. 2010;16:3731
196. Paliwal S, Jain S, Mudgal P, Verma K, Paliwal S, Sharma S. Mitochondrial transfer restores impaired liver functions by AMPK/ mTOR/PI3K-AKT pathways in metabolic syndrome. Life Sciences. 2023;332:122116
197. Hsu M-J, Karkossa I, Schäfer I, Christ M, Kühne H, Schubert K. et al. Mitochondrial Transfer by Human Mesenchymal Stromal Cells Ameliorates Hepatocyte Lipid Load in a Mouse Model of NASH. Biomedicines. 2020;8:350
198. Wang Q, Zhu K, Zhang A. SIRT1-mediated tunnelling nanotubes may be a potential intervention target for arsenic-induced hepatocyte senescence and liver damage. Science of The Total Environment. 2024;947:174502
199. Xiong Z, Chen P, Wang Z, Yao L, Yuan M, Liu P. et al. Human umbilical cord-derived mesenchymal stem cells attenuate liver fibrosis by inhibiting hepatocyte ferroptosis through mitochondrial transfer. Free Radical Biology and Medicine. 2025;231:163-77
200. Lu T, Zhang J, Cai J, Xiao J, Sui X, Yuan X. et al. Extracellular vesicles derived from mesenchymal stromal cells as nanotherapeutics for liver ischaemia-reperfusion injury by transferring mitochondria to modulate the formation of neutrophil extracellular traps. Biomaterials. 2022;284:121486
201. Jing M, Xiong X, Mao X, Song Q, Zhang L, Ouyang Y. et al. HMGB1 promotes mitochondrial transfer between hepatocellular carcinoma cells through RHOT1 and RAC1 under hypoxia. Cell Death & Disease. 2024;15:155
202. Reidy KJ, Ross MJ. Re-energizing the kidney: targeting fatty acid metabolism protects against kidney fibrosis. Kidney International. 2021;100:742-4
203. Zhang X, Agborbesong E, Li X. The Role of Mitochondria in Acute Kidney Injury and Chronic Kidney Disease and Its Therapeutic Potential. International Journal of Molecular Sciences. 2021;22:11253
204. Zhang Y, Cao H, Yang F, Wang X, Xu Z, Cheng M. et al. Comprehensive analyses of nuclear mitochondria-related genes in the molecular features, immune infiltration, and drug sensitivity of clear cell renal cell carcinoma. Medicine Advances. 2024;2:238-53
205. Doke T, Susztak K. The multifaceted role of kidney tubule mitochondrial dysfunction in kidney disease development. Trends in Cell Biology. 2022;32:841-53
206. Aranda-Rivera AK, Cruz-Gregorio A, Aparicio-Trejo OE, Pedraza-Chaverri J. Mitochondrial Redox Signaling and Oxidative Stress in Kidney Diseases. Biomolecules. 2021;11:1144
207. Konari N, Nagaishi K, Kikuchi S, Fujimiya M. Mitochondria transfer from mesenchymal stem cells structurally and functionally repairs renal proximal tubular epithelial cells in diabetic nephropathy in vivo. Scientific Reports. 2019;9:5184
208. Lee SA, Choi C, Yoo T-H. Extracellular vesicles in kidneys and their clinical potential in renal diseases. Kidney Research and Clinical Practice. 2021;40:194-207
209. Lee SA, Yoo TH. Therapeutic application of extracellular vesicles for various kidney diseases: a brief review. BMB Reports. 2022;55:3-10
210. Tang L-X, Wei B, Jiang L-Y, Ying Y-Y, Li K, Chen T-X. et al. Intercellular mitochondrial transfer as a means of revitalizing injured glomerular endothelial cells. World Journal of Stem Cells. 2022;14:729-43
211. Yuan Y, Yuan L, Li L, Liu F, Liu J, Chen Y. et al. Mitochondrial transfer from mesenchymal stem cells to macrophages restricts inflammation and alleviates kidney injury in diabetic nephropathy mice via PGC-1α activation. STEM CELLS. 2021;39:913-28
212. Chen T-H, Koh K-Y, Lin KM-C, Chou C-K. Mitochondrial Dysfunction as an Underlying Cause of Skeletal Muscle Disorders. International Journal of Molecular Sciences. 2022;23:12926
213. Zhou H, Zhang W, Li H, Xu F, Yinwang E, Xue Y. et al. Osteocyte mitochondria inhibit tumor development via STING-dependent antitumor immunity. Sci Adv. 2024;10:eadi4298
214. Liu H, Li Z, Cao Y, Cui Y, Yang X, Meng Z. et al. Effect of chondrocyte mitochondrial dysfunction on cartilage degeneration: A possible pathway for osteoarthritis pathology at the subcellular level. Molecular Medicine Reports. 2019
215. Liu Y, Shah KM, Luo J. Strategies for Articular Cartilage Repair and Regeneration. Frontiers in Bioengineering and Biotechnology. 2021;9:770655
216. Paliwal S, Chaudhuri R, Agrawal A, Mohanty S. Regenerative abilities of mesenchymal stem cells through mitochondrial transfer. Journal of Biomedical Science. 2018;25:31
217. Wang R, Maimaitijuma T, Ma Y-Y, Jiao Y, Cao Y-P. Mitochondrial transfer from bone-marrow-derived mesenchymal stromal cells to chondrocytes protects against cartilage degenerative mitochondrial dysfunction in rats chondrocytes. Chinese Medical Journal. 2021;134:212-8
218. Yu M, Wang D, Chen X, Zhong D, Luo J. BMSCs-derived Mitochondria Improve Osteoarthritis by Ameliorating Mitochondrial Dysfunction and Promoting Mitochondrial Biogenesis in Chondrocytes. Stem Cell Reviews and Reports. 2022;18:3092-111
219. Cai W, Zhang J, Yu Y, Ni Y, Wei Y, Cheng Y. et al. Mitochondrial Transfer Regulates Cell Fate Through Metabolic Remodeling in Osteoporosis. Advanced Science. 2022;10:2204871
220. Maniyadath B, Zhang Q, Gupta RK, Mandrup S. Adipose tissue at single-cell resolution. Cell Metab. 2023;35:386-413
221. Brestoff JR, Wilen CB, Moley JR, Li Y, Zou W, Malvin NP. et al. Intercellular Mitochondria Transfer to Macrophages Regulates White Adipose Tissue Homeostasis and Is Impaired in Obesity. Cell Metab. 2021;33:270-82.e8
222. Hua F, Sun J, Shi M, Mei R, Song Z, Liu J. et al. Macrophage-derived extracellular vesicles transfer mitochondria to adipocytes and promote adipocyte-myofibroblast transition in epidural fibrosis. NPJ Regen Med. 2024;9:43
223. Wang L, Hao M, Xu Y, Wang Z, Xie H, Zhang B. et al. Adipose-derived stem cells attenuate rheumatoid arthritis by restoring CX(3)CR1(+) synovial lining macrophage barrier. Stem Cell Res Ther. 2025;16:111
224. Mei Z, Li H, Huang C, Ma S, Li Y, Deng P. et al. Extracellular vesicles from adipose-derived stromal/stem cells reprogram dendritic cells to alleviate rat TMJOA by transferring mitochondria. J Nanobiotechnology. 2025;23:389
225. Rackham CL, Hubber EL, Czajka A, Malik AN, King AJF, Jones PM. Optimizing beta cell function through mesenchymal stromal cell-mediated mitochondria transfer. Stem Cells. 2020;38:574-84
226. Mori D, Miyagawa S, Kawamura T, Yoshioka D, Hata H, Ueno T. et al. Mitochondrial Transfer Induced by Adipose-Derived Mesenchymal Stem Cell Transplantation Improves Cardiac Function in Rat Models of Ischemic Cardiomyopathy. Cell Transplant. 2023;32:9636897221148457
227. Del Vecchio V, Rehman A, Panda SK, Torsiello M, Marigliano M, Nicoletti MM. et al. Mitochondrial transfer from Adipose stem cells to breast cancer cells drives multi-drug resistance. J Exp Clin Cancer Res. 2024;43:166
228. Wang ZB, Hao JX, Meng TG, Guo L, Dong MZ, Fan LH. et al. Transfer of autologous mitochondria from adipose tissue-derived stem cells rescues oocyte quality and infertility in aged mice. Aging (Albany NY). 2017;9:2480-8
229. Zhang Q, Hao JX, Liu BW, Ouyang YC, Guo JN, Dong MZ. et al. Supplementation of mitochondria from endometrial mesenchymal stem cells improves oocyte quality in aged mice. Cell Prolif. 2023;56:e13372
230. Zhang C, Tao L, Yue Y, Ren L, Zhang Z, Wang X. et al. Mitochondrial transfer from induced pluripotent stem cells rescues developmental potential of in vitro fertilized embryos from aging females†. Biol Reprod. 2021;104:1114-25
231. Jiang Z, Shi C, Han H, Fu M, Zhu H, Han T. et al. Autologous non-invasively derived stem cells mitochondria transfer shows therapeutic advantages in human embryo quality rescue. Biol Res. 2023;56:60
232. Kankanam Gamage US, Hashimoto S, Miyamoto Y, Nakano T, Yamanaka M, Koike A. et al. Mitochondria Transfer from Adipose Stem Cells Improves the Developmental Potential of Cryopreserved Oocytes. Biomolecules. 2022 12
233. Liu Y, Fu T, Li G, Li B, Luo G, Li N. et al. Mitochondrial transfer between cell crosstalk - An emerging role in mitochondrial quality control. Ageing Res Rev. 2023;91:102038
234. Zorov DB, Juhaszova M, Sollott SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94:909-50
235. Huang Y, Li W, Sun H, Guo X, Zhou Y, Liu J. et al. Mitochondrial transfer in the progression and treatment of cardiac disease. Life Sci. 2024;358:123119
236. Ikeda G, Santoso MR, Tada Y, Li AM, Vaskova E, Jung JH. et al. Mitochondria-Rich Extracellular Vesicles from Autologous Stem Cell-Derived Cardiomyocytes Restore Energetics of Ischemic Myocardium. J Am Coll Cardiol. 2021;77:1073-88
237. Dutra Silva J, Su Y, Calfee CS, Delucchi KL, Weiss D, McAuley DF. et al. Mesenchymal stromal cell extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur Respir J. 2021 58
238. Bustamante-Barrientos FA, Luque-Campos N, Araya MJ, Lara-Barba E, de Solminihac J, Pradenas C. et al. Mitochondrial dysfunction in neurodegenerative disorders: Potential therapeutic application of mitochondrial transfer to central nervous system-residing cells. J Transl Med. 2023;21:613
239. Baldwin JG, Heuser-Loy C, Saha T, Schelker RC, Slavkovic-Lukic D, Strieder N. et al. Intercellular nanotube-mediated mitochondrial transfer enhances T cell metabolic fitness and antitumor efficacy. Cell. 2024;187:6614-30.e21
240. Sakas R, Dan K, Edelman D, Abu-Ata S, Ben-Menashe A, Awad-Igbaria Y. et al. Hyperbaric Oxygen Therapy Alleviates Memory and Motor Impairments Following Traumatic Brain Injury via the Modulation of Mitochondrial-Dysfunction-Induced Neuronal Apoptosis in Rats. Antioxidants (Basel). 2023 12
241. Li H, Sun W, Gong W, Han Y. Transfer and fates of damaged mitochondria: role in health and disease. Febs j. 2024;291:5342-64
242. Ng MYW, Wai T, Simonsen A. Quality control of the mitochondrion. Dev Cell. 2021;56:881-905
243. Nicolás-Ávila JA, Lechuga-Vieco AV, Esteban-Martínez L, Sánchez-Díaz M, Díaz-García E, Santiago DJ. et al. A Network of Macrophages Supports Mitochondrial Homeostasis in the Heart. Cell. 2020;183:94-109.e23
244. Rosina M, Ceci V, Turchi R, Chuan L, Borcherding N, Sciarretta F. et al. Ejection of damaged mitochondria and their removal by macrophages ensure efficient thermogenesis in brown adipose tissue. Cell Metab. 2022;34:533-48.e12
245. An H, Zhou B, Ji X. Mitochondrial quality control in acute ischemic stroke. J Cereb Blood Flow Metab. 2021;41:3157-70
246. Ikeda H, Kawase K, Nishi T, Watanabe T, Takenaga K, Inozume T. et al. Immune evasion through mitochondrial transfer in the tumour microenvironment. Nature. 2025;638:225-36
247. Zhang W, Zhou H, Li H, Mou H, Yinwang E, Xue Y. et al. Cancer cells reprogram to metastatic state through the acquisition of platelet mitochondria. Cell Rep. 2023;42:113464
248. He X, Zhong L, Wang N, Zhao B, Wang Y, Wu X. et al. Gastric Cancer Actively Remodels Mechanical Microenvironment to Promote Chemotherapy Resistance via MSCs-Mediated Mitochondrial Transfer. Adv Sci (Weinh). 2024;11:e2404994
249. Frisbie L, Pressimone C, Dyer E, Baruwal R, Garcia G, St Croix C. et al. Carcinoma-associated mesenchymal stem cells promote ovarian cancer heterogeneity and metastasis through mitochondrial transfer. Cell Rep. 2024;43:114551
250. Nakhle J, Khattar K, Özkan T, Boughlita A, Abba Moussa D, Darlix A. et al. Mitochondria Transfer from Mesenchymal Stem Cells Confers Chemoresistance to Glioblastoma Stem Cells through Metabolic Rewiring. Cancer Res Commun. 2023;3:1041-56
251. Lecoutre S, Clément K, Dugail I. Obesity-Related Adipose Tissue Remodeling in the Light of Extracellular Mitochondria Transfer. Int J Mol Sci. 2022 23
252. Levoux J, Prola A, Lafuste P, Gervais M, Chevallier N, Koumaiha Z. et al. Platelets facilitate the wound-healing capability of mesenchymal stem cells by mitochondrial transfer and metabolic reprogramming. Cell Metab. 2021;33:688-90
253. Cai W, Zhang J, Yu Y, Ni Y, Wei Y, Cheng Y. et al. Mitochondrial Transfer Regulates Cell Fate Through Metabolic Remodeling in Osteoporosis. Adv Sci (Weinh). 2023;10:e2204871
254. Gollihue JL, Rabchevsky AG. Prospects for therapeutic mitochondrial transplantation. Mitochondrion. 2017;35:70-9
255. Schapira AH. Mitochondrial diseases. Lancet. 2012;379:1825-34
256. Davis RL, Liang C, Sue CM. Mitochondrial diseases. Handb Clin Neurol. 2018;147:125-41
257. Koga Y, Povalko N, Nishioka J, Katayama K, Kakimoto N, Matsuishi T. MELAS and L-arginine therapy: pathophysiology of stroke-like episodes. Ann N Y Acad Sci. 2010;1201:104-10
258. Rahman S, Hargreaves IP. Allogeneic stem cell transplantation corrects biochemical derangements in MNGIE. Neurology. 2007;68:1872 author reply; discussion -3
259. Garone C, Garcia-Diaz B, Emmanuele V, Lopez LC, Tadesse S, Akman HO. et al. Deoxypyrimidine monophosphate bypass therapy for thymidine kinase 2 deficiency. EMBO Mol Med. 2014;6:1016-27
260. Dai Y, Zheng K, Clark J, Swerdlow RH, Pulst SM, Sutton JP. et al. Rapamycin drives selection against a pathogenic heteroplasmic mitochondrial DNA mutation. Hum Mol Genet. 2014;23:637-47
261. Khan NA, Auranen M, Paetau I, Pirinen E, Euro L, Forsström S. et al. Effective treatment of mitochondrial myopathy by nicotinamide riboside, a vitamin B3. EMBO Mol Med. 2014;6:721-31
262. Ng YS, Bindoff LA, Gorman GS, Klopstock T, Kornblum C, Mancuso M. et al. Mitochondrial disease in adults: recent advances and future promise. Lancet Neurol. 2021;20:573-84
263. Ippolito L, Morandi A, Taddei ML, Parri M, Comito G, Iscaro A. et al. Cancer-associated fibroblasts promote prostate cancer malignancy via metabolic rewiring and mitochondrial transfer. Oncogene. 2019;38:5339-55
264. Burt R, Dey A, Aref S, Aguiar M, Akarca A, Bailey K. et al. Activated stromal cells transfer mitochondria to rescue acute lymphoblastic leukemia cells from oxidative stress. Blood. 2019;134:1415-29
265. Scalcon V, Bindoli A, Rigobello MP. Significance of the mitochondrial thioredoxin reductase in cancer cells: An update on role, targets and inhibitors. Free Radic Biol Med. 2018;127:62-79
266. Piekarska K, Urban-Wójciuk Z, Kurkowiak M, Pelikant-Małecka I, Schumacher A, Sakowska J. et al. Mesenchymal stem cells transfer mitochondria to allogeneic Tregs in an HLA-dependent manner improving their immunosuppressive activity. Nat Commun. 2022;13:856
267. Court AC, Parra-Crisóstomo E, Castro-Córdova P, Abdo L, Aragão EAA, Lorca R. et al. Survival advantage of native and engineered T cells is acquired by mitochondrial transfer from mesenchymal stem cells. J Transl Med. 2024;22:868
268. Zhang H, Yu X, Ye J, Li H, Hu J, Tan Y. et al. Systematic investigation of mitochondrial transfer between cancer cells and T cells at single-cell resolution. Cancer Cell. 2023;41:1788-802.e10
269. Spees JL, Lee RH, Gregory CA. Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res Ther. 2016;7:125
270. Yang J, Xie Y, Xia Z, Ji S, Yang X, Yue D. et al. HucMSC-Exo Induced N2 Polarization of Neutrophils: Implications for Angiogenesis and Tissue Restoration in Wound Healing. Int J Nanomedicine. 2024;19:3555-75
271. Lu W, Li X, Wang Z, Zhao C, Li Q, Zhang L. et al. Mesenchymal stem cell-derived extracellular vesicles accelerate diabetic wound healing by inhibiting NET-induced ferroptosis of endothelial cells. Int J Biol Sci. 2024;20:3515-29
272. Wang M, Yang D, Li L, Wu P, Sun Y, Zhang X. et al. A Dual Role of Mesenchymal Stem Cell Derived Small Extracellular Vesicles on TRPC6 Protein and Mitochondria to Promote Diabetic Wound Healing. ACS Nano. 2024;18:4871-85
273. Dong Z, Liu X, Li S, Fu X. Inhibition and Rescue of Hyperglycemia-Induced Cellular Senescence by Mitochondrial Transfer from Enucleated Mesenchymal Stem Cell-Derived Microvesicles for Chronic Wound Healing. Adv Sci (Weinh). 2025: e01612.
274. He S, Li Z, Xie L, Lin R, Yan B, Li B. et al. Biomimetic gene delivery system coupled with extracellular vesicle-encapsulated AAV for improving diabetic wound through promoting vascularization and remodeling of inflammatory microenvironment. J Nanobiotechnology. 2025;23:242
275. Han S, Lee M, Shin Y, Giovanni R, Chakrabarty RP, Herrerias MM. et al. Mitochondrial integrated stress response controls lung epithelial cell fate. Nature. 2023;620:890-7
276. Jin T, Liu X, Li G, Sun S, Xie L. Intravenous injection of BMSCs modulate tsRNA expression and ameliorate lung remodeling in COPD mice. Stem Cell Res Ther. 2024;15:450
277. Michaeloudes C, Li X, Mak JCW, Bhavsar PK. Study of Mesenchymal Stem Cell-Mediated Mitochondrial Transfer in In Vitro Models of Oxidant-Mediated Airway Epithelial and Smooth Muscle Cell Injury. Methods Mol Biol. 2021;2269:93-105
278. Ye L, Gao Y, Mok SWF, Liao W, Wang Y, Chen C. et al. Modulation of alveolar macrophage and mitochondrial fitness by medicinal plant-derived nanovesicles to mitigate acute lung injury and viral pneumonia. J Nanobiotechnology. 2024;22:190
279. Liu F, Lu J, Manaenko A, Tang J, Hu Q. Mitochondria in Ischemic Stroke: New Insight and Implications. Aging Dis. 2018;9:924-37
280. Lu M, Guo J, Wu B, Zhou Y, Wu M, Farzaneh M. et al. Mesenchymal Stem Cell-Mediated Mitochondrial Transfer: a Therapeutic Approach for Ischemic Stroke. Transl Stroke Res. 2021;12:212-29
281. Hakiminia B, Alikiaii B, Khorvash F, Mousavi S. Oxidative stress and mitochondrial dysfunction following traumatic brain injury: From mechanistic view to targeted therapeutic opportunities. Fundam Clin Pharmacol. 2022;36:612-62
282. Courtes AA, Gonçalves DF, Hartmann DD, da Rosa PC, Cassol G, Royes LFF. et al. Guanosine protects against behavioural and mitochondrial bioenergetic alterations after mild traumatic brain injury. Brain Res Bull. 2020;163:31-9
283. Zhao J, Qu D, Xi Z, Huan Y, Zhang K, Yu C. et al. Mitochondria transplantation protects traumatic brain injury via promoting neuronal survival and astrocytic BDNF. Transl Res. 2021;235:102-14
284. Yao S, Pang M, Wang Y, Wang X, Lin Y, Lv Y. et al. Mesenchymal stem cell attenuates spinal cord injury by inhibiting mitochondrial quality control-associated neuronal ferroptosis. Redox Biol. 2023;67:102871
285. Li H, Wang C, He T, Zhao T, Chen YY, Shen YL. et al. Mitochondrial Transfer from Bone Marrow Mesenchymal Stem Cells to Motor Neurons in Spinal Cord Injury Rats via Gap Junction. Theranostics. 2019;9:2017-35
286. Xiong X, Zhou C, Yu Y, Xie Q, Xia L, Li Q. et al. Mitochondrial Transplantation/Transfer: Promising Therapeutic Strategies for Spinal Cord Injury. J Orthop Translat. 2025;52:441-50
287. Alway SE, Paez HG, Pitzer CR, Ferrandi PJ, Khan MM, Mohamed JS. et al. Mitochondria transplant therapy improves regeneration and restoration of injured skeletal muscle. J Cachexia Sarcopenia Muscle. 2023;14:493-507
288. Court AC, Vega-Letter AM, Parra-Crisóstomo E, Velarde F, García C, Ortloff A. et al. Mitochondrial transfer balances cell redox, energy and metabolic homeostasis in the osteoarthritic chondrocyte preserving cartilage integrity. Theranostics. 2024;14:6471-86
289. Wu B, Zhao TV, Jin K, Hu Z, Abdel MP, Warrington KJ. et al. Mitochondrial aspartate regulates TNF biogenesis and autoimmune tissue inflammation. Nat Immunol. 2021;22:1551-62
290. Guo Y, Chi X, Wang Y, Heng BC, Wei Y, Zhang X. et al. Mitochondria transfer enhances proliferation, migration, and osteogenic differentiation of bone marrow mesenchymal stem cell and promotes bone defect healing. Stem Cell Res Ther. 2020;11:245
291. Hu Z, Wang D, Gong J, Li Y, Ma Z, Luo T. et al. MSCs Deliver Hypoxia-Treated Mitochondria Reprogramming Acinar Metabolism to Alleviate Severe Acute Pancreatitis Injury. Adv Sci (Weinh). 2023;10:e2207691
292. Wei B, Ji M, Lin Y, Wang S, Liu Y, Geng R. et al. Mitochondrial transfer from bone mesenchymal stem cells protects against tendinopathy both in vitro and in vivo. Stem Cell Res Ther. 2023;14:104
293. Walker M, Levitt MR, Federico EM, Miralles FJ, Levy SH, Lynne Prijoles K. et al. Autologous mitochondrial transplant for acute cerebral ischemia: Phase 1 trial results and review. J Cereb Blood Flow Metab. 2024: 271678x241305230.
294. Guariento A, Piekarski BL, Doulamis IP, Blitzer D, Ferraro AM, Harrild DM. et al. Autologous mitochondrial transplantation for cardiogenic shock in pediatric patients following ischemia-reperfusion injury. J Thorac Cardiovasc Surg. 2021;162:992-1001
295. Zhou X, Liu S, Lu Y, Wan M, Cheng J, Liu J. MitoEVs: A new player in multiple disease pathology and treatment. J Extracell Vesicles. 2023;12:e12320
296. Wang ZH, Chen L, Li W, Chen L, Wang YP. Mitochondria transfer and transplantation in human health and diseases. Mitochondrion. 2022;65:80-7
297. Hyslop LA, Blakely EL, Aushev M, Marley J, Takeda Y, Pyle A. et al. Mitochondrial Donation and Preimplantation Genetic Testing for mtDNA Disease. N Engl J Med. 2025;393:438-49
298. Kim HR, Cho HB, Lee S, Park JI, Kim HJ, Park KH. Fusogenic liposomes encapsulating mitochondria as a promising delivery system for osteoarthritis therapy. Biomaterials. 2023;302:122350
299. Ore A, Angelastro JM, Giulivi C. Integrating Mitochondrial Biology into Innovative Cell Therapies for Neurodegenerative Diseases. Brain Sci. 2024 14
300. Nakai R, Varnum S, Field RL, Shi H, Giwa R, Jia W. et al. Mitochondria transfer-based therapies reduce the morbidity and mortality of Leigh syndrome. Nat Metab. 2024;6:1886-96
301. Li C, Cheung MKH, Han S, Zhang Z, Chen L, Chen J. et al. Mesenchymal stem cells and their mitochondrial transfer: a double-edged sword. Biosci Rep. 2019 39
302. Liu Y, Wang L, Ai J, Li K. Mitochondria in Mesenchymal Stem Cells: Key to Fate Determination and Therapeutic Potential. Stem Cell Rev Rep. 2024;20:617-36
303. Scatena R, Bottoni P, Botta G, Martorana GE, Giardina B. The role of mitochondria in pharmacotoxicology: a reevaluation of an old, newly emerging topic. Am J Physiol Cell Physiol. 2007;293:C12-21
304. Ljubojevic N, Henderson JM, Zurzolo C. The Ways of Actin: Why Tunneling Nanotubes Are Unique Cell Protrusions. Trends Cell Biol. 2021;31:130-42
305. Huang T, Zhang T, Jiang X, Li A, Su Y, Bian Q. et al. Iron oxide nanoparticles augment the intercellular mitochondrial transfer-mediated therapy. Sci Adv. 2021;7:eabj0534
306. Novak J, Neuzil J. To transfer mitochondria or not to transfer mitochondria: ADP does the trick. PLOS Biology. 2024;22:e3002754
307. Gunawan AL, Liparulo I, Stahl A. Mammalian mitohormesis: from mitochondrial stressors to organismal benefits. Embo j. 2025;44:5640-61
308. Scheiblich H, Eikens F, Wischhof L, Opitz S, Jüngling K, Cserép C. et al. Microglia rescue neurons from aggregate-induced neuronal dysfunction and death through tunneling nanotubes. Neuron. 2024;112:3106-25.e8
309. Watson DC, Bayik D, Storevik S, Moreino SS, Sprowls SA, Han J. et al. GAP43-dependent mitochondria transfer from astrocytes enhances glioblastoma tumorigenicity. Nat Cancer. 2023;4:648-64
310. Liu Y, Wu M, Zhong C, Xu B, Kang L. M2-like macrophages transplantation protects against the doxorubicin-induced heart failure via mitochondrial transfer. Biomater Res. 2022;26:14
311. Konari N, Nagaishi K, Kikuchi S, Fujimiya M. Mitochondria transfer from mesenchymal stem cells structurally and functionally repairs renal proximal tubular epithelial cells in diabetic nephropathy in vivo. Sci Rep. 2019;9:5184
312. Fahey M, Bennett M, Thomas M, Montney K, Vivancos-Koopman I, Pugliese B. et al. Mesenchymal stromal cells donate mitochondria to articular chondrocytes exposed to mitochondrial, environmental, and mechanical stress. Sci Rep. 2022;12:21525
Author contact
Corresponding authors: G.X. (xiaogzedu.cn), W.Y. (szyiwh01com).

 Global reach, higher impact
Global reach, higher impact