Impact Factor
ISSN: 1449-2288
Int J Biol Sci 2018; 14(5):577-585. doi:10.7150/ijbs.22220 This issue Cite
Research Paper
Chemical and genetic inhibition of STAT3 sensitizes hepatocellular carcinoma cells to sorafenib induced cell death
1. Department of Biochemistry and Molecular Biology, School of Basic Medical Sciences, Fujian Medical University, Fuzhou, Fujian, 350108, China.
2. Department of Food and Bioengineering, Fujian Vocational College of Bioengineering, Fuzhou, Fujian, 350007, China.
3. Fujian Key Laboratory for Translational Research in Cancer and Neurodegenerative Diseases, Institute for Translational Medicine, Fujian Medical University, Fuzhou, Fujian, 350108, China.
4. Key Laboratory of Ministry of Education for Gastrointestinal Cancer, Fujian Medical University, Fuzhou, Fujian, 350108, China.
*These authors contribute equally to this work.
Abstract
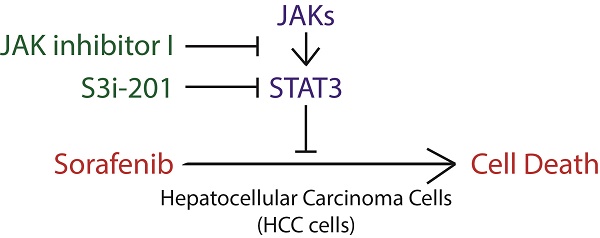
Hepatocellular carcinoma (HCC) has become the second leading cause of cancer related death, with an increasing death rate in recent years. For advanced HCC, sorafenib is the first-line FDA approved drug, with no more than 3 months' overall survival advantage. Recently, a novel strategy has been proposed to improve sorafenib efficacy through enhancing the ability of sorafenib to induce cell death. STAT3 plays a key role in cancer development and recurrence by promoting cell proliferation, survival and immune evasion through its well-established function as a transcription factor in cancer. Notably, STAT3 transcription activity, indicated by its phosphorylation on Y705 is heterogeneous in different liver cancer cell lines. And sorafenib attenuates STAT3 phosphorylation on Y705. However, the role of STAT3 in sorafenib induced cell death is still largely unknown. Here, we show that liver cancer cells also exhibit heterogeneous sensitivities to sorafenib induced cell death, which co-relates with the STAT3-Y705 phosphorylation levels and JAK1/2 expression levels in Hep3B, Huh7 and HepG2 cells. Furthermore, overexpression or knockdown of STAT3 could switch HCC cells between resistant and sensitive to sorafenib induced cell death, which could be partially due to its regulation on Mcl-1, an anti-apoptotic protein. Finally, both inhibitors of STAT3 SH2 domain (S3i-201) or STAT3 upstream kinases JAKs (JAK inhibitor I) could synergistically enhance sorafenib induced cell death. Taken together, these data strongly suggest that STAT3 is not only a downstream effector of sorafenib, but also a key regulator of cellular sensitivity to sorafenib induced cell death, which provide support for the notion to develop STAT3-targeting drugs to improve clinical efficacy of sorafenib in liver cancer.
Keywords: STAT3, HCC, Sorafenib, Cell death

 Global reach, higher impact
Global reach, higher impact