Impact Factor ISSN: 1449-2288
Int J Biol Sci 2018; 14(12):1658-1668. doi:10.7150/ijbs.28482 This issue Cite
Research Paper
Epigenetic alterations of a novel antioxidant gene SLC22A3 predispose susceptible individuals to increased risk of esophageal cancer
1. Guangdong Key Laboratory for Genome Stability & Disease Prevention, Department of Pharmacology and Carson International Cancer Center, Shenzhen University School of Medicine, Shenzhen 518039, China;
2. Department of Stomatology, Shenzhen Hospital of Guangzhou University of Chinese Medicine, Shenzhen, 518033, China;
3. Department of Medicine and Therapeutics, Li Ka Shing Institute of Health Sciences, Chinese University of Hong Kong;
4. Shenzhen Huarui Translational Research Institute, Shenzhen, China;
5. Department of Medicine and Oncology, The Johns Hopkins University School of Medicine and Sidney Kimmel Comprehensive Cancer Center, Baltimore, Maryland, USA;
6. Department of Clinical Oncology, the First Affiliated Hospital, Zhengzhou University, Zhengzhou, China;
7. Department of Clinical Oncology, University of Hong Kong, Hong Kong
*These authors contributed equally to this work.
Abstract
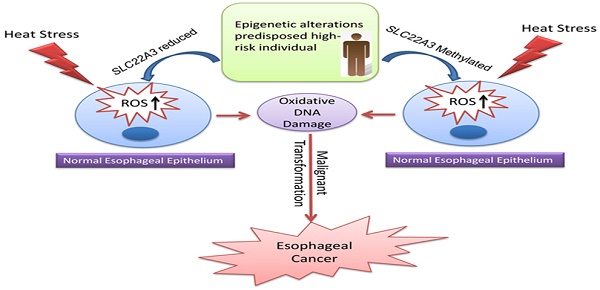
Esophageal squamous cell carcinoma (ESCC) occurs with the highest frequency in China, especially in the high-risk Northern Chinese. Recent studies have reported that SLC22A3 is significantly downregulated in non-tumor (NT) esophageal tissues from familial ESCC patients compared with those from sporadic ESCC. However, the mechanism of how SLC22A3 regulates familial ESCC remains unknown. In this study, post hoc genome-wide association studies (GWAS) in 496 cases with a family history of upper gastrointestinal tract cancers and 1056 controls were performed and the results revealed that SLC22A3 is a novel susceptibility gene for familial ESCC. Reduced expression of SLC22A3 in NT esophageal tissues from familial ESCC patients significantly correlates with its promoter hypermethylation. Moreover, case-control study of Chinese descendants from different risk areas of China revealed that the methylation of the SLC22A3 gene in peripheral blood leukocyte (PBL) DNA samples could be a risk factor for developing ESCC in this high-risk population. Functional studies showed that SLC22A3 is a novel antioxidant gene, and deregulation of SLC22A3 facilitates heat stress-induced oxidative DNA damage and formation of γ-H2AX foci in normal esophageal epithelial cells. Collectively, we show that epigenetic alterations of SLC22A3 predispose susceptible individuals to increased risk of esophageal cancer.
Keywords: Familial ESCC - SLC22A3 - DNA methylation - antioxidant gene -oxidative DNA damage

 Global reach, higher impact
Global reach, higher impact